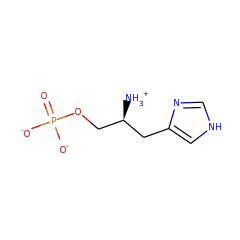
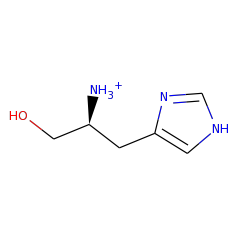
Top Level Name
⌊ Superfamily (core) Haloacid Dehalogenase
⌊ Subgroup C1.5: HAD, Beta-PGM, Phosphatase Like
⌊ C1.5.5: Heptose Bisphosphate Phosphatase Like
⌊ Family histidinol-phosphatase
⌊ FunctionalDomain histidinol-phosphatase (ID 2264)
No Notes.
| Superfamily Assignment Evidence Code(s) | FSM PubMed:7966317 |
| Family Assignment Evidence Code | CFM PubMed:3007936 |
| This entry was last updated on | Nov. 22, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Escherichia coli str. K-12 substr. MG1655 Taxon ID: 511145 | 90111373 | NP_416526.4 (RefSeq) | URP |
| Taxon ID: 543 | 446002250 | WP_000080105.1 (RefSeq) | |
| Escherichia coli Taxon ID: 562 | 857141086 | AKO57553.1 (Genbank) | URP |
| Escherichia coli GM4792 Taxon ID: 1411691 | 827622289 | AKK18023.1 (Genbank) | |
| Escherichia coli GM4792 Taxon ID: 1411691 | 827616396 | AKK12379.1 (Genbank) | |
| Escherichia coli Taxon ID: 562 | 817591852 | AKF72431.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817587712 | AKF68292.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817583571 | AKF64152.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817579432 | AKF60014.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817575290 | AKF55874.1 (Genbank) | URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808735357 | AKD92192.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808730940 | AKD87776.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808722622 | AKD83418.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808718252 | AKD79049.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808713840 | AKD74638.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808709481 | AKD70280.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808705119 | AKD65919.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808700743 | AKD61544.1 (Genbank) | PRP URP |
| Escherichia coli VR50 Taxon ID: 941323 | 801068557 | AKA91054.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. MG1655 Taxon ID: 511145 | 732684095 | AIZ90900.1 (Genbank) | URP |
| Escherichia coli K-12 Taxon ID: 83333 | 731470950 | AIZ51832.1 (Genbank) | PRP URP |
| Escherichia coli ER2796 Taxon ID: 1245474 | 730583221 | AIZ28507.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729878888 | KHH71801.1 (Genbank) | URP |
| Escherichia coli NCTC 50110 Taxon ID: 1333537 | 697407407 | KGL68511.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 685224121 | KGA86173.1 (Genbank) | URP |
| Escherichia coli BW25113 Taxon ID: 679895 | 682119515 | AIN32441.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672845465 | KFH88986.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672838083 | KFH82272.1 (Genbank) | URP |
| Escherichia coli KLY Taxon ID: 1435461 | 664684332 | AIF37283.1 (Genbank) | URP |
| Escherichia coli 3-373-03_S4_C3 Taxon ID: 1444279 | 660030577 | KEL28706.1 (Genbank) | URP |
| Escherichia coli 3-373-03_S4_C1 Taxon ID: 1444224 | 650136094 | KDU55778.1 (Genbank) | URP |
| Escherichia coli 3-373-03_S4_C2 Taxon ID: 1444252 | 650112847 | KDU33254.1 (Genbank) | URP |
| Escherichia coli 907446 Taxon ID: 1268981 | 553576475 | ESC98086.1 (Genbank) | URP |
| Escherichia coli C321.deltaA Taxon ID: 1385755 | 549813466 | AGX34084.1 (Genbank) | URP |
| Escherichia coli UMEA 3174-1 Taxon ID: 1281204 | 535614834 | EQX51646.1 (Genbank) | URP |
| Escherichia coli KTE198 Taxon ID: 1181744 | 508347855 | EOV19441.1 (Genbank) | URP |
| Escherichia coli S17 Taxon ID: 1227813 | 449320886 | EMD10908.1 (Genbank) | URP |
| Escherichia coli KTE148 Taxon ID: 1169391 | 431627349 | ELI95757.1 (Genbank) | URP |
| Escherichia coli KTE139 Taxon ID: 1169389 | 431615636 | ELI84761.1 (Genbank) | URP |
| Escherichia coli KTE120 Taxon ID: 1169379 | 431565036 | ELI38180.1 (Genbank) | URP |
| Escherichia coli KTE211 Taxon ID: 1181757 | 431492213 | ELH71816.1 (Genbank) | URP |
| Escherichia coli KTE175 Taxon ID: 1181725 | 431445510 | ELH26437.1 (Genbank) | URP |
| Escherichia coli KTE173 Taxon ID: 1181724 | 431444789 | ELH25811.1 (Genbank) | URP |
| Escherichia coli KTE162 Taxon ID: 1182734 | 431233920 | ELF29498.1 (Genbank) | URP |
| Escherichia coli KTE216 Taxon ID: 1181762 | 431028094 | ELD41138.1 (Genbank) | URP |
| Escherichia coli H730 Taxon ID: 656413 | 385710798 | EIG47774.1 (Genbank) | URP |
| Escherichia coli J53 Taxon ID: 1144303 | 384379226 | EIE37094.1 (Genbank) | URP |
| Escherichia coli XH001 Taxon ID: 1069495 | 344191730 | EGV45839.1 (Genbank) | URP |
| Escherichia coli XH140A Taxon ID: 1068608 | 342361207 | EGU25352.1 (Genbank) | URP |
| Escherichia coli DH1 Taxon ID: 536056 | 260448877 | ACX39299.1 (Genbank) | URP |
| Escherichia coli BW2952 Taxon ID: 595496 | 238861198 | ACR63196.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. DH10B Taxon ID: 316385 | 169889488 | ACB03195.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. MG1655 Taxon ID: 511145 | 87082027 | AAC75083.2 (Genbank) | URP |
| Escherichia coli K-12 Taxon ID: 83333 | 802135473 | PRP URP | |
| Escherichia coli Taxon ID: 562 | 687677165 | URP | |
| Escherichia coli Taxon ID: 562 | 687672873 | URP | |
| Escherichia coli Taxon ID: 562 | 678292973 | URP | |
| Escherichia coli str. K-12 substr. MC4100 Taxon ID: 1403831 | 557272324 | URP | |
| Escherichia coli str. K-12 substr. MDS42 Taxon ID: 1110693 | 359332420 | URP | |
| Escherichia coli DH1 Taxon ID: 536056 | 315136655 | URP | |
| Escherichia coli SE15 Taxon ID: 431946 | 281179122 | URP | |
| Escherichia coli str. K-12 substr. W3110 Taxon ID: 316407 | 1736700 | ||
| Escherichia coli K-12 Taxon ID: 83333 | 123158 | PRP URP | |
| Escherichia coli Taxon ID: 562 | 41711 | URP | |
| Escherichia coli Taxon ID: 562 | 41696 | URP | |
| obsolete GIs = 89108843, 458915953, 457977057, 450244849, 433106322, 433096901, 433048377, 432981458, 432927283, 432919475, 432694886, 432500517, 423702236, 418957617, 417978641, 417947079, 471333783, 387829965, 387621732, 386595188, 238901213, 388478077, 170081653 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| Histidine biosynthesis bifunctional protein HisB {ECO:0000255|HAMAP-Rule:MF_01022} | P06987 | HIS7_ECOLI (Swiss-Prot) |
Length of Enzyme (full-length): 355 | Length of Functional Domain: 195
MSQKYLFIDRDGTLISEPPSDFQVDRFDKLAFEPGVIPELLKLQKAGYKLVMITNQDGLG
TQSFPQADFDGPHNLMMQIFTSQGVQFDEVLICPHLPADECDCRKPKVKLVERYLAEQAM
DRANSYVIGDRATDIQLAENMGITGLRYDRETLNWPMIGEQLTRRDRYAHVVRNTKETQI
DVQVWLDREGGSKINTGVGFFDHMLDQIATHGGFRMEINVKGDLYIDDHHTVEDTGLALG
EALKIALGDKRGICRFGFVLPMDECLARCALDISGRPHLEYKAEFTYQRVGDLSTEMIEH
FFRSLSYTMGVTLHLKTKGKNDHHRVESLFKAFGRTLRQAIRVEGDTLPSSKGVL
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.