Yang Y, Jao S, Nanduri S, Starke DW, Mieyal JJ, Qin J
Reactivity of the human thioltransferase (glutaredoxin) C7S, C25S, C78S, C82S mutant and NMR solution structure of its glutathionyl mixed disulfide intermediate reflect catalytic specificity
▸ Abstract
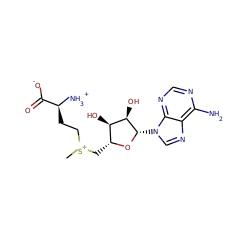
Human thioltransferase (TTase) is a 12 kDa thiol-disulfide oxidoreductase that appears to play a critical role in maintaining the redox environment of the cell. TTase acts as a potent and specific reducing agent for protein-S-S-glutathione mixed disulfides (protein-SSG) likely formed during oxidative stress or as redox intermediates in signal transduction pathways. Accordingly, the catalytic cycle of thioltransferase itself involves a covalent glutathionyl enzyme disulfide intermediate (TTase-C22-SSG). To understand the molecular basis of TTase specificity for the glutathione moiety, we engineered a quadruple Cys to Ser mutant of human TTase (C7S, C25S, C78S, and C82S) which retains only the active site cysteine residue (C22), and we solved its high-resolution NMR solution structure in the mixed disulfide intermediate with glutathione (QM-TTase-SSG). This mutant which cannot form a C22-S-S-C25 intramolecular disulfide displays the same catalytic efficiency (Vmax/KM) and specificity for glutathionyl mixed disulfide substrates as wild-type TTase, indicating that the Cys-25-SH moiety is not required for catalysis or glutathionyl specificity. The structure of human thioltransferase is characterized by a thioredoxin-like fold which comprises a four-stranded central beta-sheet flanked on each side by alpha-helices. The disulfide-adducted glutathione in the TTase-SSG complex has an extended conformation and is localized in a cleft near the protein surface encompassing the residues from helices-alpha2,alpha3, the active site loop, and the loop connecting helix-alpha3 and strand-beta3. Numerous van der Waals and electrostatic interactions between the protein and the glutathione moiety are identified as contributing to stabilization of the complex and confering the substrate specificity. Comparison of the human thioltransferase with other thiol-disulfide oxidoreductases reveals structural and functional differences.
Biochemistry
1998;37(49):17145-17156
| PubMed ID:
9860827
Kriek M, Martins F, Challand MR, Croft A, Roach PL
Thiamine biosynthesis in Escherichia coli: identification of the intermediate and by-product derived from tyrosine
▸ Abstract
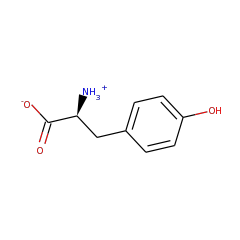
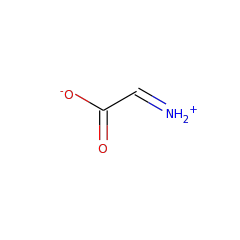
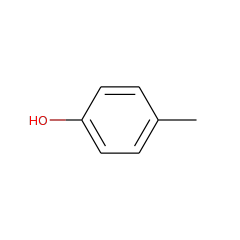
In anaerobic organisms such as E. coli the tyrosine lyase ThiH is essential for the biosynthesis of the thiazole moiety of the vitamin thiamine. ThiH is a member of the “radical AdoMet” family. The products formed by cleavage of tyrosine in vitro have been identified and suggest a radical-mediated cleavage resulting in p-cresol and dehydroglycine which is hydrolyzed to glyoxylate.
Angew Chem Int Ed Engl.
2007;46(48):9223-9226
| PubMed ID:
17969213
Kriek M, Martins F, Leonardi R, Fairhurst SA, Lowe DJ, Roach PL
Thiazole synthase from Escherichia coli: an investigation of the substrates and purified proteins required for activity in vitro
▸ Abstract
Thiamine is biosynthesized by combining two heterocyclic precursors. In Escherichia coli and other anaerobes, one of the heterocycles, 4-methyl-5-(beta-hydroxyethyl) thiazole phosphate, is biosynthesized from 1-deoxyxylulose-5-phosphate, tyrosine, and cysteine. Genetic evidence has identified thiH, thiG, thiS, and thiF as essential for thiazole biosynthesis in E. coli. In this paper, we describe the measurement of the thiazole phosphate-forming reaction using purified protein components. The activity is shown to require four proteins isolated as heterodimers: ThiGH and ThiFS. Reconstitution of the [4Fe-4S] cluster in ThiH was essential for activity, as was the use of ThiS in the thiocarboxylate form. Spectroscopic studies with ThiGH strongly suggested that S-adenosylmethionine (AdoMet) bound to the [4Fe-4S] cluster, which became more susceptible to reduction to the +1 state. Assays of thiazole phosphate formation showed that, in addition to the proteins, Dxp, tyrosine, AdoMet, and a reductant were required. The analysis showed that no more than 1 mol eq of thiazole phosphate was formed per ThiGH. Furthermore, for each mole of thiazole-P formed, 1 eq of AdoMet and 1 eq of tyrosine were utilized, and 1 eq of 5'-deoxyadenosine was produced. These results demonstrate that ThiH is a member of the "radical-AdoMet" family and support a mechanistic hypothesis in which AdoMet is reductively cleaved to yield a highly reactive 5'-deoxyadenosyl radical. This radical is proposed to abstract the phenolic hydrogen atom from tyrosine, and the resultant substrate radical cleaves to yield dehydroglycine, which is required by ThiG for the thiazole cyclization reaction.
J Biol Chem.
2007;282(24):17413-17423
| PubMed ID:
17403671
Challand MR, Martins FT, Roach PL
Catalytic activity of the anaerobic tyrosine lyase required for thiamine biosynthesis in Escherichia coli
▸ Abstract
Thiazole synthase in Escherichia coli is an alphabeta heterodimer of ThiG and ThiH. ThiH is a tyrosine lyase that cleaves the C alpha-C beta bond of tyrosine, generating p-cresol as a by-product, to form dehydroglycine. This reactive intermediate acts as one of three substrates for the thiazole cyclization reaction catalyzed by ThiG. ThiH is a radical S-adenosylmethionine (AdoMet) enzyme that utilizes a [4Fe-4S](+) cluster to reductively cleave AdoMet, forming methionine and a 5'-deoxyadenosyl radical. Analysis of the time-dependent formation of the reaction products 5'-deoxyadenosine (DOA) and p-cresol has demonstrated catalytic behavior of the tyrosine lyase. The kinetics of product formation showed a pre-steady state burst phase, and the involvement of DOA in product inhibition was identified by the addition of 5'-methylthioadenosine/S-adenosylhomocysteine nucleosidase to activity assays. This hydrolyzed the DOA and changed the rate-determining step but, in addition, substantially increased the uncoupled turnover of AdoMet. Addition of glyoxylate and ammonium inhibited the tyrosine cleavage reaction, but the reductive cleavage of AdoMet continued in an uncoupled manner. Tyrosine analogues were incubated with ThiGH, which showed a strong preference for phenolic substrates. 4-Hydroxyphenylpropionic acid analogues allowed uncoupled AdoMet cleavage but did not result in further reaction (C alpha-C beta bond cleavage). The results of the substrate analogue studies and the product inhibition can be explained by a mechanistic hypothesis involving two reaction pathways, a product-forming pathway and a futile cycle.
J Biol Chem
2010;285(8):5240-5248
| PubMed ID:
19923213
Leonardi R, Roach PL
Thiamine biosynthesis in Escherichia coli: in vitro reconstitution of the thiazole synthase activity
▸ Abstract
The biosynthesis of thiamine in Escherichia coli requires the formation of an intermediate thiazole from tyrosine, 1-deoxy-d-xylulose-5-phosphate (Dxp), and cysteine using at least six structural proteins, ThiFSGH, IscS, and ThiI. We describe for the first time the reconstitution of thiazole synthase activity using cell-free extracts and proteins derived from adenosine-treated E. coli 83-1 cells. The addition of adenosine or adenine to growing cultures of Aerobacter aerogenes, Salmonella typhimurium, and E. coli has been shown previously to relieve the repression by thiamine of its own biosynthesis and increase the expression levels of the thiamine biosynthetic enzymes. By exploiting this effect, we show that the in vitro thiazole synthase activity of cleared lysates or desalted proteins from E. coli 83-1 cells is dependent upon the addition of purified ThiGH-His complex, tyrosine (but not cysteine or 1-deoxy-d-xylulose-5-phosphate), and an as yet unidentified intermediate present in the protein fraction from these cells. The activity is strongly stimulated by the addition of S-adenosylmethionine and NADPH.
J Biol Chem
2004;279(17):17054-17062
| PubMed ID:
14757766
Martinez-Gomez NC, Robers M, Downs DM
Mutational analysis of ThiH, a member of the radical S-adenosylmethionine (AdoMet) protein superfamily
▸ Abstract
Thiamine pyrophosphate (TPP) is an essential cofactor for all forms of life. In Salmonella enterica, the thiH gene product is required for the synthesis of the 4-methyl-5-beta hydroxyethyl-thiazole monophosphate moiety of TPP. ThiH is a member of the radical S-adenosylmethionine (AdoMet) superfamily of proteins that is characterized by the presence of oxygen labile [Fe-S] clusters. Lack of an in vitro activity assay for ThiH has hampered the analysis of this interesting enzyme. We circumvented this problem by using an in vivo activity assay for ThiH. Random and directed mutagenesis of the thiH gene was performed. Analysis of auxotrophic thiH mutants defined two classes, those that required thiazole to make TPP (null mutants) and those with thiamine auxotrophy that was corrected by either L-tyrosine or thiazole (ThiH* mutants). Increased levels of AdoMet also corrected the thiamine requirement of members of the latter class. Residues required for in vivo function were identified and are discussed in the context of structures available for AdoMet enzymes.
J Biol Chem
2004;279(39):40505-40510
| PubMed ID:
15271986