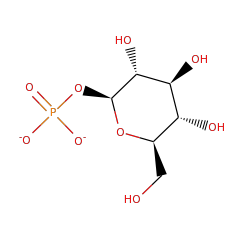
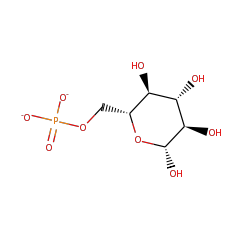
Top Level Name
⌊ Superfamily (core) Haloacid Dehalogenase
⌊ Subgroup C1.5: HAD, Beta-PGM, Phosphatase Like
⌊ C1.5.6: HAD, Beta-PGM, Phosphatase Like
⌊ Family beta-phosphoglucomutase
⌊ FunctionalDomain beta-phosphoglucomutase (ID 2262)
No Notes.
| Superfamily Assignment Evidence Code(s) | FSM PubMed:9649311 |
| Family Assignment Evidence Code | CFM PubMed:12081483 |
| This entry was last updated on | Nov. 22, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Lactococcus lactis Taxon ID: 1358 | 502661210 | WP_012897250.1 (RefSeq) | URP |
| Lactococcus lactis subsp. lactis Taxon ID: 1360 | 736044825 | AJA56351.1 (Genbank) | URP |
| Lactococcus lactis Taxon ID: 1358 | 691499754 | KGF76471.1 (Genbank) | URP |
| Lactococcus lactis subsp. lactis NCDO 2118 Taxon ID: 1117941 | 669185531 | AII11986.1 (Genbank) | URP |
| Lactococcus lactis subsp. lactis KF147 Taxon ID: 684738 | 281374730 | ADA64250.1 (Genbank) | URP |
| Lactococcus lactis subsp. lactis Taxon ID: 1360 | 751242629 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 665764210 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 665764209 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 665764208 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 574451042 | URP | |
| Lactococcus lactis subsp. lactis Dephy 1 Taxon ID: 1390360 | 552527087 | URP | |
| Lactococcus lactis subsp. lactis IO-1 Taxon ID: 1046624 | 374672505 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 296863327 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 296863326 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 296863325 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 296863324 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 296278280 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 258588196 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 258588195 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 75765829 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 66361316 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 66361315 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 66361314 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 66361313 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 29726863 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 29726862 | URP | |
| Lactococcus lactis Taxon ID: 1358 | 1495997 | URP | |
| obsolete GIs = 459284617, 281490972 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| Beta-phosphoglucomutase | P71447 | 5.4.2.6 | PGMB_LACLA (Swiss-Prot) |
Length of Enzyme (full-length): 221 | Length of Functional Domain: 218
MFKAVLFDLDGVITDTAEYHFRAWKALAEEIGINGVDRQFNEQLKGVSREDSLQKILDLA
DKKVSAEEFKELAKRKNDNYVKMIQDVSPADVYPGILQLLKDLRSNKIKIALASASKNGP
FLLERMNLTGYFDAIADPAEVAASKPAPDIFIAAAHAVGVAPSESIGLEDSQAGIQAIKD
SGALPIGVGRPEDLGDDIVIVPDTSHYTLEFLKEVWLQKQK
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.