Top Level Name
⌊ Superfamily (core) Enolase
⌊ Subgroup muconate cycloisomerase
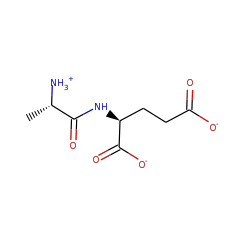
⌊ Family dipeptide epimerase
⌊ FunctionalDomain dipeptide epimerase (ID 457)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS |
| Family Assignment Evidence Code | CFM PubMed:11747448 |
| This entry was last updated on | Nov. 22, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Bacillus subtilis subsp. subtilis str. 168 Taxon ID: 224308 | 16078363 | NP_389181.1 (RefSeq) | PRP URP |
| Bacillus subtilis Taxon ID: 1423 | 489337771 | WP_003244980.1 (RefSeq) | URP |
| Bacillus subtilis KCTC 1028 Taxon ID: 1136873 | 807073643 | AKC46849.1 (Genbank) | URP |
| Bacillus subtilis Taxon ID: 1423 | 760456464 | KIX82539.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis Taxon ID: 135461 | 749185265 | AJE93975.1 (Genbank) | URP |
| Bacillus subtilis Taxon ID: 1423 | 728888513 | AIY96907.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis str. 168 Taxon ID: 224308 | 728884200 | AIY92595.1 (Genbank) | PRP URP |
| Bacillus subtilis subsp. subtilis Taxon ID: 135461 | 672774657 | KFH34232.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis Taxon ID: 135461 | 672773760 | KFH33336.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis str. OH 131.1 Taxon ID: 1404258 | 655530402 | AIC97843.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis str. AG1839 Taxon ID: 1221328 | 649015011 | AIC43933.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis str. JH642 substr. AG174 Taxon ID: 1232554 | 649010647 | AIC39701.1 (Genbank) | URP |
| Bacillus subtilis PY79 Taxon ID: 1415167 | 558567895 | AHA77301.1 (Genbank) | URP |
| Bacillus subtilis subsp. subtilis 6051-HGW Taxon ID: 1147161 | 459389075 | AGG60661.1 (Genbank) | URP |
| Bacillus subtilis MB73/2 Taxon ID: 1267547 | 452116500 | EME06895.1 (Genbank) | URP |
| Bacillus subtilis QB928 Taxon ID: 1220533 | 402480716 | AFQ57225.1 (Genbank) | URP |
| Bacillus sp. Taxon ID: 1409 | 724426954 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 723797760 | URP | |
| Bacillus subtilis BEST7003 Taxon ID: 1204342 | 407964263 | URP | |
| Bacillus subtilis BEST7613 Taxon ID: 1204343 | 407956994 | URP | |
| Bacillus subtilis subsp. subtilis str. 168 Taxon ID: 224308 | 81342334 | PRP URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695821 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695820 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695819 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695818 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695817 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695816 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695815 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 52695814 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 18158853 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 18158852 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 18158851 | URP | |
| Bacillus subtilis Taxon ID: 1423 | 18158850 | URP | |
| Bacillus subtilis subsp. subtilis str. 168 Taxon ID: 224308 | 2633652 | PRP URP | |
| Bacillus subtilis subsp. subtilis str. 168 Taxon ID: 224308 | 2632019 | PRP URP | |
| obsolete GIs = 433618236, 452914080, 221322680, 221318406, 221313484, 221309158, 560128369, 470161469, 402775533 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| L-Ala-D/L-Glu epimerase | O34508 | AEEP_BACSU (Swiss-Prot) |
Length of Enzyme (full-length): 366 | Length of Functional Domain: 360
MKIIRIETSRIAVPLTKPFKTALRTVYTAESVIVRITYDSGAVGWGEAPPTLVITGDSMD
SIESAIHHVLKPALLGKSLAGYEAILHDIQHLLTGNMSAKAAVEMALYDGWAQMCGLPLY
QMLGGYRDTLETDYTVSVNSPEEMAADAENYLKQGFQTLKIKVGKDDIATDIARIQEIRK
RVGSAVKLRLDANQGWRPKEAVTAIRKMEDAGLGIELVEQPVHKDDLAGLKKVTDATDTP
IMADESVFTPRQAFEVLQTRSADLINIKLMKAGGISGAEKINAMAEACGVECMVGSMIET
KLGITAAAHFAASKRNITRFDFDAPLMLKTDVFNGGITYSGSTISMPGKPGLGIIGAALL
KGEKEQ
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.