Top Level Name
⌊ Superfamily (core) Enolase
⌊ Subgroup galactarate dehydratase
⌊ Family galactarate dehydratase 2
⌊ FunctionalDomain galactarate dehydratase (ID 366)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS |
| Family Assignment Evidence Code | CFM PubMed:19883118 |
| This entry was last updated on | Nov. 21, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Oceanobacillus iheyensis Taxon ID: 182710 | 499379662 | WP_011067240.1 (RefSeq) | |
| Oceanobacillus iheyensis HTE831 Taxon ID: 221109 | 224510961 | URP | |
| Oceanobacillus iheyensis HTE831 Taxon ID: 221109 | 222447155 | URP | |
| Oceanobacillus iheyensis HTE831 Taxon ID: 221109 | 222447154 | URP | |
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870597 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870596 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870595 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870594 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870593 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870592 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870591 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870590 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870589 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 209870588 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105357 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105356 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105355 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105354 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105353 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105352 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105351 | ||
| Oceanobacillus iheyensis Taxon ID: 182710 | 134105350 | ||
| Oceanobacillus iheyensis HTE831 Taxon ID: 221109 | 81745467 | URP | |
| Oceanobacillus iheyensis HTE831 Taxon ID: 221109 | 22778530 | URP | |
| obsolete GI = 23100298 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
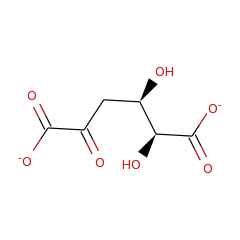
| Galactarate dehydratase (D-threo-forming) | Q8EMJ9 | GALRD_OCEIH (Swiss-Prot) |
Length of Enzyme (full-length): 391 | Length of Functional Domain: 391
MKITDLELHAVGIPRHTGFVNKHVIVKIHTDEGLTGIGEMSDFSHLPLYSVDLHDLKQGL
LSILLGQNPFDLMKINKELTDNFPETMYYYEKGSFIRNGIDNALHDLCAKYLDISVSDFL
GGRVKEKIKVCYPIFRHRFSEEVESNLDVVRQKLEQGFDVFRLYVGKNLDADEEFLSRVK
EEFGSRVRIKSYDFSHLLNWKDAHRAIKRLTKYDLGLEMIESPAPRNDFDGLYQLRLKTD
YPISEHVWSFKQQQEMIKKDAIDIFNISPVFIGGLTSAKKAAYAAEVASKDVVLGTTQEL
SVGTAAMAHLGCSLTNINHTSDPTGPELYVGDVVKNRVTYKDGYLYAPDRSVKGLGIELD
ESLLAKYQVPDLSWDNVTVHQLQDRTADTKS
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.