Top Level Name
⌊ Superfamily (core) Radical SAM
⌊ Subgroup lipoyl synthase like
⌊ Family lipoyl synthase
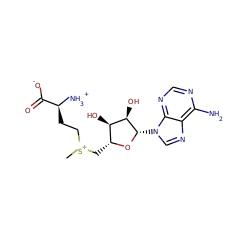
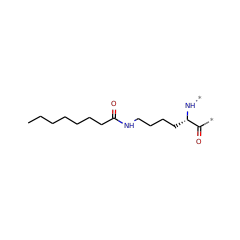
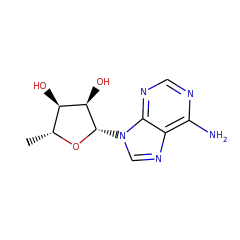
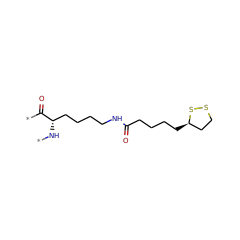
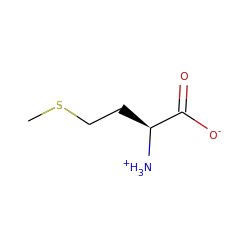
⌊ FunctionalDomain Lipoyl synthase (ID 280037)
No Notes.
| Superfamily Assignment Evidence Code(s) | FSM PubMed:11106496 PubMed:14700636 PubMed:15157071 PubMed:21370834 PubMed:18307109 PubMed:15362861 PubMed:15740115 PubMed:15112987 PubMed:22773649 |
| Family Assignment Evidence Code | CFM PubMed:11106496 PubMed:14700636 PubMed:15157071 |
| This entry was last updated on | June 10, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Escherichia coli O157:H7 str. Sakai Taxon ID: 386585 | 15829920 | NP_308693.1 (RefSeq) | |
| Escherichia coli str. K-12 substr. MG1655 Taxon ID: 511145 | 16128611 | NP_415161.1 (RefSeq) | URP |
| Shigella flexneri 2a str. 301 Taxon ID: 198214 | 24112072 | NP_706582.1 (RefSeq) | |
| Taxon ID: 2 | 445964777 | WP_000042632.1 (RefSeq) | |
| Escherichia coli IAI39 Taxon ID: 585057 | 218699000 | YP_002406629.1 (RefSeq) | URP |
| Escherichia coli O104:H4 str. 2011C-3493 Taxon ID: 1133852 | 407483214 | YP_006780363.1 (RefSeq) | URP |
| Escherichia coli O83:H1 str. NRG 857C Taxon ID: 685038 | 387615914 | YP_006118936.1 (RefSeq) | URP |
| Escherichia coli UMN026 Taxon ID: 585056 | 218703961 | YP_002411480.1 (RefSeq) | URP |
| Shigella dysenteriae Sd197 Taxon ID: 300267 | 82775897 | YP_402244.1 (RefSeq) | URP |
| Escherichia coli Taxon ID: 562 | 1778545 | AAB40828.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 209777188 | ACI86906.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 209777190 | ACI86907.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 209777192 | ACI86908.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 209777194 | ACI86909.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 209777196 | ACI86910.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 595584202 | AHM28544.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 595591575 | AHM35471.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 595596173 | AHM40068.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 723229741 | AIX62384.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 732674853 | AIZ81662.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 732679374 | AIZ86181.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 742950122 | AJB50791.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748268519 | AJE54953.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 751399879 | AJF75964.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 753775091 | AJH09520.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 758861554 | AJO82512.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 806959502 | AKC12807.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817147039 | AKF19627.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817573966 | AKF54550.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817578108 | AKF58690.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817582247 | AKF62828.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817586388 | AKF66968.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817590529 | AKF71108.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 821332507 | AKH25610.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 850013970 | AKN46739.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 857139675 | AKO56142.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 899721653 | AKR19580.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 899726009 | AKR23935.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 899730381 | AKR28306.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 597592401 | EYB38839.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 597594021 | EYB40418.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 597599648 | EYB45915.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 597600583 | EYB46813.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 597605286 | EYB51408.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 597613854 | EYB59556.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 629841577 | KCW95595.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 636539503 | KDM71165.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 636549866 | KDM81285.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 636555619 | KDM86874.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 636874571 | KDN07550.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 641941909 | KDO91656.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 643344915 | KDP17221.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 660654895 | KEO94568.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 660665119 | KEP04647.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 660667263 | KEP06788.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 660672025 | KEP11510.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 660677733 | KEP17132.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 660680765 | KEP20072.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 669336767 | KFD76739.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 671393525 | KFF40148.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 671700077 | KFF52037.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672833802 | KFH78082.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672839978 | KFH84103.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672849615 | KFH92927.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672851091 | KFH94315.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 672857858 | KFI00866.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 678154067 | KFV22637.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 678158874 | KFV27311.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 678163530 | KFV31873.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 678168239 | KFV36379.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 685225327 | KGA87361.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 693354776 | KGI51230.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 699399360 | KGM77930.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 699402255 | KGM80638.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 700819105 | KGP12556.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 700820211 | KGP13611.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 700828647 | KGP21190.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 701201446 | KGP40968.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 701213630 | KGP52601.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 701215905 | KGP54863.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 705993980 | KGT07369.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 706042283 | KGT12057.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 706051474 | KGT20732.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 706054666 | KGT23800.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 706058193 | KGT26920.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 706061753 | KGT30163.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 724480092 | KHD39914.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 724487787 | KHD47351.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 724497777 | KHD57192.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 724500205 | KHD59592.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729777303 | KHG73240.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729781465 | KHG77289.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729785596 | KHG81301.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729792672 | KHG88195.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729795920 | KHG91259.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729800726 | KHG95837.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729804296 | KHG99330.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729813277 | KHH07880.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729819424 | KHH13776.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729821798 | KHH16087.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729822414 | KHH16692.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729833104 | KHH27132.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729834505 | KHH28499.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729843392 | KHH37248.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729848449 | KHH42149.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729853498 | KHH47056.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729856562 | KHH50005.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729863352 | KHH56618.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729867678 | KHH60800.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729872289 | KHH65331.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729874555 | KHH67550.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729882271 | KHH75043.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729884451 | KHH77186.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729891338 | KHH83997.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729900704 | KHH92934.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729904300 | KHH96436.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729908229 | KHI00221.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729920578 | KHI12193.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729922522 | KHI14019.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729927415 | KHI18792.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729931629 | KHI22876.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729942732 | KHI33486.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729944912 | KHI35615.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729945336 | KHI36031.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729952973 | KHI43440.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729959972 | KHI50262.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729964259 | KHI54341.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729964477 | KHI54555.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729970517 | KHI60457.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729976815 | KHI66556.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729981916 | KHI71540.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729988469 | KHI77835.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729993020 | KHI82257.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729996396 | KHI85461.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 729998966 | KHI87937.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730007799 | KHI96525.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730011159 | KHI99844.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730019442 | KHJ07799.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730022840 | KHJ11079.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730029044 | KHJ16921.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730035476 | KHJ23072.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 730041374 | KHJ28879.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 735080607 | KHO57647.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 738559652 | KHQ63137.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 746677758 | KIE69024.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 746683516 | KIE74699.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 746689829 | KIE80955.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748017344 | KIG29598.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748032002 | KIG43547.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748032816 | KIG44347.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748044619 | KIG55535.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748046629 | KIG57294.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748052945 | KIG63214.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748055003 | KIG65032.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748060396 | KIG70220.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748065710 | KIG74732.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748073549 | KIG81199.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748083304 | KIG90573.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748085083 | KIG92294.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748094472 | KIH01371.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748099984 | KIH06816.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748101215 | KIH08021.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748107808 | KIH14297.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748112281 | KIH18330.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748119527 | KIH24930.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748119981 | KIH25371.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748127900 | KIH33090.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 748129478 | KIH34521.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 749029625 | KII10543.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 752194836 | KIN86570.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 755719141 | KIQ40791.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 755816678 | KIQ45636.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 761660594 | KIY29311.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 762042981 | KIZ08321.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763493975 | KIZ63332.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763498153 | KIZ67310.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763507518 | KIZ76048.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763511636 | KIZ79891.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763518031 | KIZ85861.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763518972 | KIZ86734.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763527089 | KIZ94428.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763532934 | KIZ99925.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763533305 | KJA00283.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 763538804 | KJA05525.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765101558 | KJD61328.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765106885 | KJD66486.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765174707 | KJD73137.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765176219 | KJD74624.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765183737 | KJD82036.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765190262 | KJD88312.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 765191896 | KJD89931.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 767818046 | KJG98715.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 767823051 | KJH03535.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 767826130 | KJH06410.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 768568299 | KJI02531.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 768576364 | KJI10373.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 768578621 | KJI12519.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 769024768 | KJJ45674.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788869464 | KJW21544.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788872240 | KJW24057.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788879307 | KJW30870.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788891242 | KJW42218.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788896587 | KJW47175.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788898225 | KJW48791.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788905336 | KJW55499.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788909805 | KJW59595.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788916200 | KJW65744.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 788925628 | KJW74401.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 796819323 | KJY09977.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 803475741 | KKB17488.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 803480263 | KKB21529.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 816355621 | KKK29401.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817016838 | KKO25468.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817021725 | KKO30227.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 817029627 | KKO37763.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822075764 | KLD47885.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822079459 | KLD51520.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822901692 | KLG32757.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822911154 | KLG38414.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822918386 | KLG43896.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822919011 | KLG44506.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822923704 | KLG49081.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822930074 | KLG55280.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822934722 | KLG59781.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822943481 | KLG68295.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822949419 | KLG74071.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822952861 | KLG77370.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822959521 | KLG83760.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822968618 | KLG92502.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822970012 | KLG93876.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822978876 | KLH02437.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822983551 | KLH07008.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822985526 | KLH08921.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822987602 | KLH10940.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 822994381 | KLH17571.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823002402 | KLH25355.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823005691 | KLH28532.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823012819 | KLH35421.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823014949 | KLH37500.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823021933 | KLH44151.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823026600 | KLH48599.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823026815 | KLH48807.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823040141 | KLH61610.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823043685 | KLH65001.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823044549 | KLH65848.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823048571 | KLH69814.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823058596 | KLH79487.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823061988 | KLH82780.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823062506 | KLH83281.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 823073652 | KLH94156.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 836409768 | KLU95184.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838724593 | KLX07900.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838725240 | KLX08545.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838725392 | KLX08696.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838736405 | KLX19579.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838741641 | KLX24748.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838748690 | KLX31749.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838754335 | KLX37362.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838756181 | KLX39166.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838760715 | KLX43599.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838767048 | KLX49853.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838773872 | KLX56552.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838779535 | KLX62140.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838785791 | KLX68309.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838796093 | KLX78505.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838797596 | KLX79985.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838804518 | KLX86824.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838811656 | KLX93896.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838819432 | KLY01611.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838821608 | KLY03771.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 838825368 | KLY07502.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 839395320 | KME72269.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848509015 | KML79305.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848510331 | KML80379.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848511807 | KML81787.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848528053 | KML96985.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848530409 | KML99244.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848531747 | KMM00531.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848541385 | KMM09344.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 848545208 | KMM12584.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 885178283 | KMV41398.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 885189376 | KMV51049.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 885192230 | KMV53459.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 885194537 | KMV55147.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 885201651 | KMV61135.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 885206147 | KMV64222.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909852405 | KNF15303.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909854543 | KNF17414.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909860439 | KNF23242.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909864098 | KNF26674.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909870117 | KNF32571.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909874867 | KNF37124.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909877243 | KNF39430.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909888118 | KNF49788.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909892800 | KNF54240.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909897315 | KNF58542.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909901996 | KNF63016.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909903118 | KNF64084.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909911021 | KNF71779.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909922213 | KNF82531.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909923211 | KNF83469.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909934710 | KNF94383.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909940643 | KNG00194.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909944504 | KNG03882.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909948465 | KNG07568.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909955290 | KNG14264.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909957149 | KNG16070.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909963728 | KNG22400.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909965665 | KNG24310.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909975176 | KNG33486.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909982132 | KNG40284.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 909983717 | KNG41843.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913110731 | KNY01842.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913527048 | KNY55090.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913531891 | KNY59850.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913534727 | KNY62633.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913547946 | KNY75528.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913551191 | KNY78716.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913551673 | KNY79183.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913563423 | KNY90653.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913563754 | KNY90974.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913572947 | KNY99699.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913574558 | KNZ01360.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913583799 | KNZ10266.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913585347 | KNZ11789.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913589713 | KNZ16059.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913595088 | KNZ21166.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 913602236 | KNZ28001.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 914351178 | KOA22436.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 914351897 | KOA23139.1 (Genbank) | URP |
| Escherichia coli Taxon ID: 562 | 914363167 | KOA34350.1 (Genbank) | URP |
| Escherichia coli 'BL21-Gold(DE3)pLysS AG' Taxon ID: 866768 | 253325430 | ACT30032.1 (Genbank) | |
| Escherichia coli 0.1288 Taxon ID: 1005442 | 408347476 | EKJ61682.1 (Genbank) | URP |
| Escherichia coli 07798 Taxon ID: 1005566 | 408221801 | EKI45734.1 (Genbank) | URP |
| Escherichia coli 08BKT77219 Taxon ID: 1081895 | 537039320 | ERE10491.1 (Genbank) | URP |
| Escherichia coli 09BKT076207 Taxon ID: 1081889 | 536798552 | ERB83680.1 (Genbank) | URP |
| Escherichia coli 09BKT078844 Taxon ID: 1081887 | 444550425 | ELV28524.1 (Genbank) | URP |
| Escherichia coli 1-110-08_S1_C1 Taxon ID: 1444049 | 598736885 | EYE39769.1 (Genbank) | URP |
| Escherichia coli 1-110-08_S1_C3 Taxon ID: 1444104 | 598724097 | EYE28059.1 (Genbank) | URP |
| Escherichia coli 1-110-08_S3_C1 Taxon ID: 1444132 | 598726156 | EYE30003.1 (Genbank) | URP |
| Escherichia coli 1-110-08_S4_C1 Taxon ID: 1444215 | 598701375 | EYE07383.1 (Genbank) | URP |
| Escherichia coli 1-110-08_S4_C2 Taxon ID: 1444243 | 598699914 | EYE06006.1 (Genbank) | URP |
| Escherichia coli 1-176-05_S1_C1 Taxon ID: 1444050 | 598682740 | EYD90412.1 (Genbank) | URP |
| Escherichia coli 1-176-05_S3_C1 Taxon ID: 1444133 | 598684554 | EYD92048.1 (Genbank) | URP |
| Escherichia coli 1-176-05_S4_C1 Taxon ID: 1444216 | 611071733 | EZJ74053.1 (Genbank) | URP |
| Escherichia coli 1-176-05_S4_C2 Taxon ID: 1444244 | 632199127 | KDA91284.1 (Genbank) | URP |
| Escherichia coli 1-176-05_S4_C3 Taxon ID: 1444273 | 611024389 | EZJ28919.1 (Genbank) | URP |
| Escherichia coli 1-182-04_S1_C3 Taxon ID: 1444106 | 611097903 | EZJ98856.1 (Genbank) | URP |
| Escherichia coli 1-182-04_S3_C3 Taxon ID: 1444191 | 611076988 | EZJ78925.1 (Genbank) | URP |
| Escherichia coli 1-182-04_S4_C3 Taxon ID: 1444274 | 611019835 | EZJ24528.1 (Genbank) | URP |
| Escherichia coli 1-250-04_S1_C1 Taxon ID: 1444052 | 652007846 | KDX27623.1 (Genbank) | URP |
| Escherichia coli 1-250-04_S3_C2 Taxon ID: 1444163 | 660350966 | KEO37867.1 (Genbank) | URP |
| Escherichia coli 1-392-07_S3_C1 Taxon ID: 1444136 | 651829714 | KDX04223.1 (Genbank) | URP |
| Escherichia coli 1-392-07_S3_C2 Taxon ID: 1444164 | 651783706 | KDW60426.1 (Genbank) | URP |
| Escherichia coli 1-392-07_S4_C3 Taxon ID: 1444275 | 660283424 | KEN73472.1 (Genbank) | URP |
| Escherichia coli 1.2264 Taxon ID: 869675 | 386164447 | EIH26233.1 (Genbank) | URP |
| Escherichia coli 1.2741 Taxon ID: 869669 | 386137942 | EIG79102.1 (Genbank) | URP |
| Escherichia coli 10.0821 Taxon ID: 1005469 | 408616909 | EKK90048.1 (Genbank) | URP |
| Escherichia coli 10.0833 Taxon ID: 1005466 | 408591818 | EKK66236.1 (Genbank) | URP |
| Escherichia coli 10.0869 Taxon ID: 1005467 | 408604011 | EKK77613.1 (Genbank) | URP |
| Escherichia coli 101-1 Taxon ID: 358709 | 194423615 | EDX39605.1 (Genbank) | URP |
| Escherichia coli 110957 Taxon ID: 1268975 | 553333393 | ESA61278.1 (Genbank) | URP |
| Escherichia coli 113302 Taxon ID: 1268977 | 553570371 | ESC92229.1 (Genbank) | URP |
| Escherichia coli 113303 Taxon ID: 1268978 | 553335659 | ESA63463.1 (Genbank) | URP |
| Escherichia coli 1303 Taxon ID: 745156 | 751379541 | AJF55368.1 (Genbank) | URP |
| Escherichia coli 14A Taxon ID: 1207035 | 536911125 | ERC91105.1 (Genbank) | URP |
| Escherichia coli 174750 Taxon ID: 1125634 | 476293445 | EMX11454.1 (Genbank) | URP |
| Escherichia coli 178850 Taxon ID: 1116106 | 477443049 | ENH06955.1 (Genbank) | URP |
| Escherichia coli 178900 Taxon ID: 1116108 | 476763103 | ENA66473.1 (Genbank) | URP |
| Escherichia coli 179100 Taxon ID: 1125652 | 477052213 | END35166.1 (Genbank) | URP |
| Escherichia coli 180200 Taxon ID: 1116104 | 476768405 | ENA71395.1 (Genbank) | URP |
| Escherichia coli 180600 Taxon ID: 1116105 | 476265658 | EMW84201.1 (Genbank) | URP |
| Escherichia coli 1827-70 Taxon ID: 670888 | 310337357 | EFQ02495.1 (Genbank) | URP |
| Escherichia coli 2-005-03_S1_C3 Taxon ID: 1444092 | 611118630 | EZK18362.1 (Genbank) | URP |
| Escherichia coli 2-005-03_S3_C1 Taxon ID: 1444117 | 651779709 | KDW56517.1 (Genbank) | URP |
| Escherichia coli 2-005-03_S3_C3 Taxon ID: 1444175 | 651789112 | KDW65451.1 (Genbank) | URP |
| Escherichia coli 2-011-08_S1_C3 Taxon ID: 1444093 | 649980079 | KDT05678.1 (Genbank) | URP |
| Escherichia coli 2-011-08_S3_C1 Taxon ID: 1444118 | 649977710 | KDT03354.1 (Genbank) | URP |
| Escherichia coli 2-052-05_S4_C1 Taxon ID: 1444202 | 650005069 | KDT29681.1 (Genbank) | URP |
| Escherichia coli 2-052-05_S4_C2 Taxon ID: 1444230 | 651708688 | KDV88501.1 (Genbank) | URP |
| Escherichia coli 2-156-04_S3_C2 Taxon ID: 1444150 | 651756041 | KDW34042.1 (Genbank) | URP |
| Escherichia coli 2-156-04_S3_C3 Taxon ID: 1444178 | 651732425 | KDW11457.1 (Genbank) | URP |
| Escherichia coli 2-177-06_S1_C1 Taxon ID: 1444040 | 651746551 | KDW24997.1 (Genbank) | URP |
| Escherichia coli 2-177-06_S1_C3 Taxon ID: 1444096 | 651766950 | KDW44329.1 (Genbank) | URP |
| Escherichia coli 2-177-06_S3_C1 Taxon ID: 1444121 | 651742902 | KDW21498.1 (Genbank) | URP |
| Escherichia coli 2-177-06_S3_C3 Taxon ID: 1444179 | 660327891 | KEO15642.1 (Genbank) | URP |
| Escherichia coli 2-177-06_S4_C2 Taxon ID: 1444232 | 651766357 | KDW43903.1 (Genbank) | URP |
| Escherichia coli 2-210-07_S1_C2 Taxon ID: 1444069 | 651817983 | KDW93120.1 (Genbank) | URP |
| Escherichia coli 2-210-07_S4_C2 Taxon ID: 1444233 | 652042660 | KDX60460.1 (Genbank) | URP |
| Escherichia coli 2-222-05_S3_C1 Taxon ID: 1444123 | 660303256 | KEN92191.1 (Genbank) | URP |
| Escherichia coli 2-222-05_S3_C2 Taxon ID: 1444153 | 660313101 | KEO01276.1 (Genbank) | URP |
| Escherichia coli 2-222-05_S4_C1 Taxon ID: 1444206 | 660201775 | KEM93851.1 (Genbank) | URP |
| Escherichia coli 2-222-05_S4_C2 Taxon ID: 1444234 | 652071530 | KDX87788.1 (Genbank) | URP |
| Escherichia coli 2-222-05_S4_C3 Taxon ID: 1444264 | 660331802 | KEO19384.1 (Genbank) | URP |
| Escherichia coli 2-316-03_S3_C1 Taxon ID: 1444124 | 652076029 | KDX92122.1 (Genbank) | URP |
| Escherichia coli 2-316-03_S3_C2 Taxon ID: 1444154 | 652084400 | KDY00077.1 (Genbank) | URP |
| Escherichia coli 2-316-03_S4_C1 Taxon ID: 1444207 | 652094475 | KDY09595.1 (Genbank) | URP |
| Escherichia coli 2-316-03_S4_C2 Taxon ID: 1444235 | 652100334 | KDY15042.1 (Genbank) | URP |
| Escherichia coli 2-316-03_S4_C3 Taxon ID: 1444265 | 652101800 | KDY16427.1 (Genbank) | URP |
| Escherichia coli 2-427-07_S1_C1 Taxon ID: 1444043 | 652290740 | KDZ94023.1 (Genbank) | URP |
| Escherichia coli 2-427-07_S1_C3 Taxon ID: 1444099 | 652180092 | KDY89759.1 (Genbank) | URP |
| Escherichia coli 2-427-07_S3_C1 Taxon ID: 1444125 | 652117416 | KDY31407.1 (Genbank) | URP |
| Escherichia coli 2-427-07_S3_C3 Taxon ID: 1444183 | 652118300 | KDY32267.1 (Genbank) | URP |
| Escherichia coli 2-427-07_S4_C1 Taxon ID: 1444208 | 652128380 | KDY41682.1 (Genbank) | URP |
| Escherichia coli 2-460-02_S1_C1 Taxon ID: 1444044 | 660347034 | KEO34064.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S1_C1 Taxon ID: 1444045 | 652168933 | KDY79489.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S1_C2 Taxon ID: 1444074 | 652192239 | KDZ01174.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S3_C1 Taxon ID: 1444127 | 652179213 | KDY88971.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S3_C2 Taxon ID: 1444156 | 652187651 | KDY97039.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S4_C1 Taxon ID: 1444210 | 660298126 | KEN87559.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S4_C2 Taxon ID: 1444238 | 652197031 | KDZ05826.1 (Genbank) | URP |
| Escherichia coli 2-474-04_S4_C3 Taxon ID: 1444268 | 652204038 | KDZ12397.1 (Genbank) | URP |
| Escherichia coli 2.3916 Taxon ID: 869688 | 386222644 | EII45063.1 (Genbank) | URP |
| Escherichia coli 2.4168 Taxon ID: 869690 | 386232550 | EII64535.1 (Genbank) | URP |
| Escherichia coli 201600.1 Taxon ID: 1116129 | 476720147 | ENA25062.1 (Genbank) | URP |
| Escherichia coli 2362-75 Taxon ID: 670897 | 312290165 | EFR18048.1 (Genbank) | URP |
| Escherichia coli 2719100 Taxon ID: 1116119 | 476373099 | EMX90229.1 (Genbank) | URP |
| Escherichia coli 2720900 Taxon ID: 1116120 | 476379241 | EMX96305.1 (Genbank) | URP |
| Escherichia coli 2726800 Taxon ID: 1116122 | 476363360 | EMX80560.1 (Genbank) | URP |
| Escherichia coli 2726950 Taxon ID: 1116123 | 476752667 | ENA56702.1 (Genbank) | URP |
| Escherichia coli 2729250 Taxon ID: 1116004 | 476751708 | ENA55774.1 (Genbank) | URP |
| Escherichia coli 2730450 Taxon ID: 1116006 | 476781523 | ENA83626.1 (Genbank) | URP |
| Escherichia coli 2731150 Taxon ID: 1116007 | 476268065 | EMW86551.1 (Genbank) | URP |
| Escherichia coli 2735000 Taxon ID: 1116008 | 476665981 | EMZ71887.1 (Genbank) | URP |
| Escherichia coli 2747800 Taxon ID: 1116010 | 476261351 | EMW79985.1 (Genbank) | URP |
| Escherichia coli 2756500 Taxon ID: 1116012 | 476245159 | EMW64286.1 (Genbank) | URP |
| Escherichia coli 2770900 Taxon ID: 1116014 | 476237240 | EMW56603.1 (Genbank) | URP |
| Escherichia coli 2780750 Taxon ID: 1116015 | 476233171 | EMW52620.1 (Genbank) | URP |
| Escherichia coli 2785200 Taxon ID: 1116016 | 476217378 | EMW37364.1 (Genbank) | URP |
| Escherichia coli 2848050 Taxon ID: 1116127 | 476208135 | EMW28402.1 (Genbank) | URP |
| Escherichia coli 2860650 Taxon ID: 1116022 | 476798601 | ENB00033.1 (Genbank) | URP |
| Escherichia coli 2862600 Taxon ID: 1116024 | 476801306 | ENB02608.1 (Genbank) | URP |
| Escherichia coli 2864350 Taxon ID: 1116025 | 476798072 | ENA99529.1 (Genbank) | URP |
| Escherichia coli 2866550 Taxon ID: 1116029 | 476156466 | EMV78162.1 (Genbank) | URP |
| Escherichia coli 2866750 Taxon ID: 1116030 | 476158726 | EMV80375.1 (Genbank) | URP |
| Escherichia coli 2871950 Taxon ID: 1116032 | 476142912 | EMV64857.1 (Genbank) | URP |
| Escherichia coli 2875150 Taxon ID: 1116036 | 476815091 | ENB15575.1 (Genbank) | URP |
| Escherichia coli 2886-75 Taxon ID: 1207038 | 536927707 | ERD06963.1 (Genbank) | URP |
| Escherichia coli 3-020-07_S1_C1 Taxon ID: 1444054 | 652213800 | KDZ21622.1 (Genbank) | URP |
| Escherichia coli 3-020-07_S3_C2 Taxon ID: 1444165 | 658729598 | KEJ70641.1 (Genbank) | URP |
| Escherichia coli 3-020-07_S4_C1 Taxon ID: 1444220 | 658720333 | KEJ61893.1 (Genbank) | URP |
| Escherichia coli 3-020-07_S4_C2 Taxon ID: 1444248 | 652236763 | KDZ43306.1 (Genbank) | URP |
| Escherichia coli 3-020-07_S4_C3 Taxon ID: 1444276 | 652242590 | KDZ48845.1 (Genbank) | URP |
| Escherichia coli 3-073-06_S1_C1 Taxon ID: 1444055 | 652248013 | KDZ54165.1 (Genbank) | URP |
| Escherichia coli 3-073-06_S4_C2 Taxon ID: 1444249 | 652268434 | KDZ73058.1 (Genbank) | URP |
| Escherichia coli 3-073-06_S4_C3 Taxon ID: 1444277 | 652281058 | KDZ84910.1 (Genbank) | URP |
| Escherichia coli 3-105-05_S1_C3 Taxon ID: 1444110 | 652286789 | KDZ90278.1 (Genbank) | URP |
| Escherichia coli 3-105-05_S3_C1 Taxon ID: 1444139 | 650012265 | KDT36520.1 (Genbank) | URP |
| Escherichia coli 3-267-03_S3_C1 Taxon ID: 1444140 | 650037272 | KDT60350.1 (Genbank) | URP |
| Escherichia coli 3-373-03_S3_C1 Taxon ID: 1444141 | 650046163 | KDT69038.1 (Genbank) | URP |
| Escherichia coli 3-373-03_S3_C2 Taxon ID: 1444169 | 650087305 | KDU08963.1 (Genbank) | URP |
| Escherichia coli 3-373-03_S3_C3 Taxon ID: 1444194 | 650050038 | KDT72736.1 (Genbank) | URP |
| Escherichia coli 3-475-03_S1_C1 Taxon ID: 1444059 | 650067285 | KDT89419.1 (Genbank) | URP |
| Escherichia coli 3-475-03_S1_C2 Taxon ID: 1444087 | 659981822 | KEK81371.1 (Genbank) | URP |
| Escherichia coli 3-475-03_S3_C2 Taxon ID: 1444170 | 659987871 | KEK87279.1 (Genbank) | URP |
| Escherichia coli 3-475-03_S4_C2 Taxon ID: 1444253 | 650136909 | KDU56559.1 (Genbank) | URP |
| Escherichia coli 3.2303 Taxon ID: 869691 | 386241899 | EII78812.1 (Genbank) | URP |
| Escherichia coli 3.2608 Taxon ID: 869679 | 386178784 | EIH56263.1 (Genbank) | URP |
| Escherichia coli 3.3884 Taxon ID: 869689 | 386229478 | EII56833.1 (Genbank) | URP |
| Escherichia coli 3.4870 Taxon ID: 1005450 | 408558892 | EKK35242.1 (Genbank) | URP |
| Escherichia coli 3.4880 Taxon ID: 1051347 | 444666781 | ELW38839.1 (Genbank) | URP |
| Escherichia coli 3006 Taxon ID: 1005565 | 408218662 | EKI42864.1 (Genbank) | URP |
| Escherichia coli 3030-1 Taxon ID: 754080 | 345360082 | EGW92253.1 (Genbank) | URP |
| Escherichia coli 3431 Taxon ID: 670892 | 315616454 | EFU97071.1 (Genbank) | URP |
| Escherichia coli 4-203-08_S4_C3 Taxon ID: 1444280 | 650147543 | KDU66927.1 (Genbank) | URP |
| Escherichia coli 4.0522 Taxon ID: 869681 | 386190392 | EIH79140.1 (Genbank) | URP |
| Escherichia coli 4.0967 Taxon ID: 869687 | 386217460 | EII33949.1 (Genbank) | URP |
| Escherichia coli 4_1_47FAA Taxon ID: 1127356 | 373248145 | EHP67577.1 (Genbank) | URP |
| Escherichia coli 5-172-05_S4_C3 Taxon ID: 1444269 | 660067621 | KEL63372.1 (Genbank) | URP |
| Escherichia coli 5-366-08_S1_C1 Taxon ID: 1444047 | 660073685 | KEL69230.1 (Genbank) | URP |
| Escherichia coli 5-366-08_S3_C1 Taxon ID: 1444129 | 660098822 | KEL93805.1 (Genbank) | URP |
| Escherichia coli 5.0588 Taxon ID: 869671 | 386152516 | EIH03805.1 (Genbank) | URP |
| Escherichia coli 5.0959 Taxon ID: 869684 | 386205963 | EII10469.1 (Genbank) | URP |
| Escherichia coli 5.2239 Taxon ID: 1005452 | 408558436 | EKK34807.1 (Genbank) | URP |
| Escherichia coli 536 Taxon ID: 362663 | 110342449 | ABG68686.1 (Genbank) | URP |
| Escherichia coli 53638 Taxon ID: 344610 | 188488367 | EDU63470.1 (Genbank) | URP |
| Escherichia coli 541-1 Taxon ID: 752786 | 388404387 | EIL64850.1 (Genbank) | URP |
| Escherichia coli 541-15 Taxon ID: 752785 | 388390137 | EIL51635.1 (Genbank) | URP |
| Escherichia coli 5412 Taxon ID: 1005561 | 408191833 | EKI17429.1 (Genbank) | URP |
| Escherichia coli 576-1 Taxon ID: 752787 | 388401172 | EIL61834.1 (Genbank) | URP |
| Escherichia coli 5905 Taxon ID: 1005558 | 408174479 | EKI01463.1 (Genbank) | URP |
| Escherichia coli 6-175-07_S1_C1 Taxon ID: 1444061 | 660057288 | KEL53349.1 (Genbank) | URP |
| Escherichia coli 6-175-07_S1_C2 Taxon ID: 1444089 | 658674278 | KEJ18280.1 (Genbank) | URP |
| Escherichia coli 6-175-07_S1_C3 Taxon ID: 1444114 | 660161659 | KEM54763.1 (Genbank) | URP |
| Escherichia coli 6-175-07_S3_C2 Taxon ID: 1444172 | 658740457 | KEJ81166.1 (Genbank) | URP |
| Escherichia coli 6-175-07_S3_C3 Taxon ID: 1444197 | 660111195 | KEM05984.1 (Genbank) | URP |
| Escherichia coli 6-175-07_S4_C2 Taxon ID: 1444255 | 660116907 | KEM11599.1 (Genbank) | URP |
| Escherichia coli 6-319-05_S1_C3 Taxon ID: 1444115 | 660141100 | KEM35127.1 (Genbank) | URP |
| Escherichia coli 6-319-05_S3_C3 Taxon ID: 1444198 | 660172558 | KEM65415.1 (Genbank) | URP |
| Escherichia coli 6-537-08_S1_C3 Taxon ID: 1444116 | 660269157 | KEN59537.1 (Genbank) | URP |
| Escherichia coli 6-537-08_S3_C3 Taxon ID: 1444199 | 660193050 | KEM85415.1 (Genbank) | URP |
| Escherichia coli 6-537-08_S4_C1 Taxon ID: 1444227 | 660203001 | KEM95036.1 (Genbank) | URP |
| Escherichia coli 6.0172 Taxon ID: 1005455 | 408559950 | EKK36240.1 (Genbank) | URP |
| Escherichia coli 7-233-03_S3_C1 Taxon ID: 1444130 | 660185506 | KEM78089.1 (Genbank) | URP |
| Escherichia coli 8-415-05_S1_C1 Taxon ID: 1444048 | 660236876 | KEN28120.1 (Genbank) | URP |
| Escherichia coli 8-415-05_S3_C2 Taxon ID: 1444159 | 660286301 | KEN76313.1 (Genbank) | URP |
| Escherichia coli 8-415-05_S3_C3 Taxon ID: 1444189 | 660244590 | KEN35524.1 (Genbank) | URP |
| Escherichia coli 8.0566 Taxon ID: 1005462 | 408572769 | EKK48654.1 (Genbank) | URP |
| Escherichia coli 8.0569 Taxon ID: 1005464 | 408573328 | EKK49184.1 (Genbank) | URP |
| Escherichia coli 8.0586 Taxon ID: 1005461 | 408585124 | EKK60026.1 (Genbank) | URP |
| Escherichia coli 8.2524 Taxon ID: 1005460 | 408590082 | EKK64580.1 (Genbank) | URP |
| Escherichia coli 83972 Taxon ID: 525281 | 227838479 | EEJ48945.1 (Genbank) | URP |
| Escherichia coli 88.0221 Taxon ID: 1005397 | 408610079 | EKK83454.1 (Genbank) | URP |
| Escherichia coli 88.1042 Taxon ID: 1005398 | 427214323 | EKV83652.1 (Genbank) | URP |
| Escherichia coli 88.1467 Taxon ID: 1005396 | 427216750 | EKV85847.1 (Genbank) | URP |
| Escherichia coli 89.0511 Taxon ID: 1005399 | 427216383 | EKV85504.1 (Genbank) | URP |
| Escherichia coli 9.0111 Taxon ID: 869686 | 386209756 | EII20243.1 (Genbank) | URP |
| Escherichia coli 9.1649 Taxon ID: 869685 | 802473112 | KKA61117.1 (Genbank) | URP |
| Escherichia coli 90.0039 Taxon ID: 1005401 | 427233792 | EKW01517.1 (Genbank) | URP |
| Escherichia coli 90.0091 Taxon ID: 1005400 | 427235945 | EKW03548.1 (Genbank) | URP |
| Escherichia coli 90.2281 Taxon ID: 1005402 | 427233678 | EKW01407.1 (Genbank) | URP |
| Escherichia coli 900105 (10e) Taxon ID: 869695 | 386262106 | EIJ17553.1 (Genbank) | URP |
| Escherichia coli 907357 Taxon ID: 1268979 | 553343162 | ESA70621.1 (Genbank) | URP |
| Escherichia coli 907391 Taxon ID: 1268980 | 553575508 | ESC97142.1 (Genbank) | URP |
| Escherichia coli 907446 Taxon ID: 1268981 | 553569382 | ESC91284.1 (Genbank) | URP |
| Escherichia coli 907672 Taxon ID: 1268982 | 553583823 | ESD05188.1 (Genbank) | URP |
| Escherichia coli 907700 Taxon ID: 1268983 | 553584934 | ESD06272.1 (Genbank) | URP |
| Escherichia coli 907701 Taxon ID: 1268984 | 553601741 | ESD22397.1 (Genbank) | URP |
| Escherichia coli 907710 Taxon ID: 1268985 | 553596688 | ESD17561.1 (Genbank) | URP |
| Escherichia coli 907713 Taxon ID: 1268986 | 553355313 | ESA82332.1 (Genbank) | URP |
| Escherichia coli 907715 Taxon ID: 1268987 | 553601204 | ESD21881.1 (Genbank) | URP |
| Escherichia coli 907779 Taxon ID: 1268988 | 553367424 | ESA93912.1 (Genbank) | URP |
| Escherichia coli 907889 Taxon ID: 1268989 | 553613979 | ESD34033.1 (Genbank) | URP |
| Escherichia coli 907892 Taxon ID: 1268990 | 553619060 | ESD38831.1 (Genbank) | URP |
| Escherichia coli 908519 Taxon ID: 1268991 | 553612376 | ESD32504.1 (Genbank) | URP |
| Escherichia coli 908521 Taxon ID: 1268992 | 553628461 | ESD47813.1 (Genbank) | URP |
| Escherichia coli 908522 Taxon ID: 1268993 | 553633474 | ESD52621.1 (Genbank) | URP |
| Escherichia coli 908525 Taxon ID: 1268995 | 553652406 | ESD70716.1 (Genbank) | URP |
| Escherichia coli 908555 Taxon ID: 1268997 | 553649148 | ESD67609.1 (Genbank) | URP |
| Escherichia coli 908573 Taxon ID: 1268998 | 553660442 | ESD78275.1 (Genbank) | URP |
| Escherichia coli 908585 Taxon ID: 1268999 | 553665589 | ESD83031.1 (Genbank) | URP |
| Escherichia coli 908616 Taxon ID: 1269000 | 553665082 | ESD82551.1 (Genbank) | URP |
| Escherichia coli 908624 Taxon ID: 1269001 | 553677956 | ESD94809.1 (Genbank) | URP |
| Escherichia coli 908632 Taxon ID: 1269002 | 553689484 | ESE05696.1 (Genbank) | URP |
| Escherichia coli 908675 Taxon ID: 1269004 | 553694344 | ESE10332.1 (Genbank) | URP |
| Escherichia coli 908691 Taxon ID: 1269005 | 553701232 | ESE16907.1 (Genbank) | URP |
| Escherichia coli 909945-2 Taxon ID: 1269007 | 553370165 | ESA96532.1 (Genbank) | URP |
| Escherichia coli 910096-2 Taxon ID: 1269010 | 553706759 | ESE22212.1 (Genbank) | URP |
| Escherichia coli 93-001 Taxon ID: 1005481 | 390669611 | EIN46231.1 (Genbank) | URP |
| Escherichia coli 93.0055 Taxon ID: 1005403 | 427252792 | EKW19262.1 (Genbank) | URP |
| Escherichia coli 93.0056 Taxon ID: 1005404 | 427251286 | EKW17872.1 (Genbank) | URP |
| Escherichia coli 93.0624 Taxon ID: 869680 | 386186080 | EIH68797.1 (Genbank) | URP |
| Escherichia coli 94.0618 Taxon ID: 1005405 | 427253940 | EKW20322.1 (Genbank) | URP |
| Escherichia coli 95.0083 Taxon ID: 1051346 | 444672529 | ELW44239.1 (Genbank) | URP |
| Escherichia coli 95.0183 Taxon ID: 1005406 | 427270373 | EKW35252.1 (Genbank) | URP |
| Escherichia coli 95.0943 Taxon ID: 1005408 | 427270137 | EKW35034.1 (Genbank) | URP |
| Escherichia coli 95.1288 Taxon ID: 1005407 | 427275526 | EKW40139.1 (Genbank) | URP |
| Escherichia coli 95JB1 Taxon ID: 1333535 | 537032714 | ERE04483.1 (Genbank) | URP |
| Escherichia coli 95NR1 Taxon ID: 1333536 | 535932385 | ERA62179.1 (Genbank) | URP |
| Escherichia coli 96.0107 Taxon ID: 1005415 | 427310413 | EKW72663.1 (Genbank) | URP |
| Escherichia coli 96.0109 Taxon ID: 1005414 | 429259900 | EKY43533.1 (Genbank) | URP |
| Escherichia coli 96.0428 Taxon ID: 1005410 | 427286134 | EKW50000.1 (Genbank) | URP |
| Escherichia coli 96.0497 Taxon ID: 869677 | 386169999 | EIH36507.1 (Genbank) | URP |
| Escherichia coli 96.0932 Taxon ID: 1005413 | 427306129 | EKW68675.1 (Genbank) | URP |
| Escherichia coli 96.0939 Taxon ID: 1005412 | 427293565 | EKW56815.1 (Genbank) | URP |
| Escherichia coli 96.154 Taxon ID: 869683 | 386200167 | EIH99158.1 (Genbank) | URP |
| Escherichia coli 97.0003 Taxon ID: 1005416 | 427304565 | EKW67203.1 (Genbank) | URP |
| Escherichia coli 97.0007 Taxon ID: 1005420 | 427322055 | EKW83705.1 (Genbank) | URP |
| Escherichia coli 97.0246 Taxon ID: 869670 | 386143616 | EIG90092.1 (Genbank) | URP |
| Escherichia coli 97.0259 Taxon ID: 869672 | 386157458 | EIH13800.1 (Genbank) | URP |
| Escherichia coli 97.0264 Taxon ID: 869676 | 753924617 | KIO86441.1 (Genbank) | URP |
| Escherichia coli 99.0670 Taxon ID: 1005423 | 444674286 | ELW45841.1 (Genbank) | URP |
| Escherichia coli 99.0678 Taxon ID: 1005422 | 427334043 | EKW95124.1 (Genbank) | URP |
| Escherichia coli 99.0713 Taxon ID: 1005424 | 427333875 | EKW94962.1 (Genbank) | URP |
| Escherichia coli 99.0741 Taxon ID: 869678 | 386171318 | EIH43363.1 (Genbank) | URP |
| Escherichia coli 99.0814 Taxon ID: 1005434 | 444542243 | ELV21627.1 (Genbank) | URP |
| Escherichia coli 99.0815 Taxon ID: 1005433 | 444551562 | ELV29475.1 (Genbank) | URP |
| Escherichia coli 99.0816 Taxon ID: 1005430 | 444566812 | ELV43606.1 (Genbank) | URP |
| Escherichia coli 99.0839 Taxon ID: 1005432 | 444564682 | ELV41610.1 (Genbank) | URP |
| Escherichia coli 99.0848 Taxon ID: 1005431 | 444571036 | ELV47537.1 (Genbank) | URP |
| Escherichia coli 99.1753 Taxon ID: 1005440 | 444582012 | ELV57837.1 (Genbank) | URP |
| Escherichia coli 99.1762 Taxon ID: 1005435 | 444653315 | ELW26038.1 (Genbank) | URP |
| Escherichia coli 99.1775 Taxon ID: 1005438 | 444584872 | ELV60476.1 (Genbank) | URP |
| Escherichia coli 99.1781 Taxon ID: 1005439 | 444649741 | ELW22614.1 (Genbank) | URP |
| Escherichia coli 99.1793 Taxon ID: 1005437 | 444585829 | ELV61369.1 (Genbank) | URP |
| Escherichia coli 99.1805 Taxon ID: 1005436 | 444607895 | ELV82451.1 (Genbank) | URP |
| Escherichia coli A25922R Taxon ID: 1269008 | 553720493 | ESE35411.1 (Genbank) | URP |
| Escherichia coli A35218R Taxon ID: 1269009 | 553712673 | ESE27862.1 (Genbank) | URP |
| Escherichia coli AA86 Taxon ID: 1004153 | 330910389 | EGH38899.1 (Genbank) | URP |
| Escherichia coli ABU 83972 Taxon ID: 655817 | 307552498 | ADN45273.1 (Genbank) | URP |
| Escherichia coli ACN001 Taxon ID: 1311757 | 874075986 | AKP83348.1 (Genbank) | URP |
| Escherichia coli AD30 Taxon ID: 1169664 | 408457913 | EKJ81704.1 (Genbank) | URP |
| Escherichia coli AI27 Taxon ID: 1089445 | 384472012 | EIE56076.1 (Genbank) | URP |
| Escherichia coli APEC IMT5155 Taxon ID: 1329907 | 742676235 | AJB38422.1 (Genbank) | URP |
| Escherichia coli APEC O1 Taxon ID: 405955 | 115511968 | ABJ00043.1 (Genbank) | URP |
| Escherichia coli APEC O78 Taxon ID: 1274814 | 443421172 | AGC86076.1 (Genbank) | URP |
| Escherichia coli ATCC 25922 Taxon ID: 1322345 | 674302543 | AIL17435.1 (Genbank) | URP |
| Escherichia coli ATCC 25922 Taxon ID: 1322345 | 507476686 | EOR51365.1 (Genbank) | URP |
| Escherichia coli ATCC 35150 Taxon ID: 1389955 | 567035705 | ETJ67661.1 (Genbank) | URP |
| Escherichia coli ATCC 700728 Taxon ID: 1051353 | 444599389 | ELV74276.1 (Genbank) | URP |
| Escherichia coli ATCC 8739 Taxon ID: 481805 | 169755943 | ACA78642.1 (Genbank) | URP |
| Escherichia coli ATCC BAA-2192 Taxon ID: 1404261 | 567044345 | ETJ80880.1 (Genbank) | URP |
| Escherichia coli ATCC BAA-2193 Taxon ID: 1404262 | 566653922 | ETJ56729.1 (Genbank) | URP |
| Escherichia coli ATCC BAA-2196 Taxon ID: 1405292 | 566115408 | ETI74178.1 (Genbank) | URP |
| Escherichia coli ATCC BAA-2209 Taxon ID: 1405293 | 564855288 | ETD64676.1 (Genbank) | URP |
| Escherichia coli ATCC BAA-2215 Taxon ID: 1405294 | 564835124 | ETD46867.1 (Genbank) | URP |
| Escherichia coli ATCC BAA-2219 Taxon ID: 1405295 | 566115066 | ETI73875.1 (Genbank) | URP |
| Escherichia coli B str. REL606 Taxon ID: 413997 | 253972611 | ACT38282.1 (Genbank) | URP |
| Escherichia coli B088 Taxon ID: 550672 | 291325267 | EFE64682.1 (Genbank) | URP |
| Escherichia coli B093 Taxon ID: 550674 | 371616258 | EHO04624.1 (Genbank) | URP |
| Escherichia coli B102 Taxon ID: 1151213 | 536803151 | ERB88096.1 (Genbank) | URP |
| Escherichia coli B103 Taxon ID: 1151234 | 536926981 | ERD06122.1 (Genbank) | URP |
| Escherichia coli B104 Taxon ID: 1151243 | 536924955 | ERD04216.1 (Genbank) | URP |
| Escherichia coli B105 Taxon ID: 1151247 | 536941196 | ERD19477.1 (Genbank) | URP |
| Escherichia coli B107 Taxon ID: 1151214 | 536793027 | ERB78311.1 (Genbank) | URP |
| Escherichia coli B108 Taxon ID: 550673 | 536943961 | ERD22092.1 (Genbank) | URP |
| Escherichia coli B109 Taxon ID: 1151244 | 536956273 | ERD33789.1 (Genbank) | URP |
| Escherichia coli B112 Taxon ID: 1151249 | 536958438 | ERD35848.1 (Genbank) | URP |
| Escherichia coli B113 Taxon ID: 1151250 | 536965972 | ERD42964.1 (Genbank) | URP |
| Escherichia coli B114 Taxon ID: 1151251 | 536974264 | ERD50746.1 (Genbank) | URP |
| Escherichia coli B15 Taxon ID: 1151237 | 536976566 | ERD52833.1 (Genbank) | URP |
| Escherichia coli B17 Taxon ID: 1151238 | 536978569 | ERD54699.1 (Genbank) | URP |
| Escherichia coli B171 Taxon ID: 344601 | 194413963 | EDX30240.1 (Genbank) | URP |
| Escherichia coli B185 Taxon ID: 550676 | 291433982 | EFF06955.1 (Genbank) | URP |
| Escherichia coli B26-1 Taxon ID: 1151225 | 536806116 | ERB90973.1 (Genbank) | URP |
| Escherichia coli B26-2 Taxon ID: 1151226 | 536810132 | ERB94882.1 (Genbank) | URP |
| Escherichia coli B28-1 Taxon ID: 1151217 | 536822657 | ERC06934.1 (Genbank) | URP |
| Escherichia coli B28-2 Taxon ID: 1151218 | 536821328 | ERC05668.1 (Genbank) | URP |
| Escherichia coli B29-2 Taxon ID: 1151220 | 536838865 | ERC22217.1 (Genbank) | URP |
| Escherichia coli B354 Taxon ID: 550677 | 291471931 | EFF14414.1 (Genbank) | URP |
| Escherichia coli B36-1 Taxon ID: 1151221 | 536841838 | ERC24983.1 (Genbank) | URP |
| Escherichia coli B36-2 Taxon ID: 1151222 | 536845800 | ERC28829.1 (Genbank) | URP |
| Escherichia coli B40-1 Taxon ID: 1151227 | 536998183 | ERD71733.1 (Genbank) | URP |
| Escherichia coli B41 Taxon ID: 869694 | 386255977 | EIJ05665.1 (Genbank) | URP |
| Escherichia coli B49-2 Taxon ID: 1151246 | 536997483 | ERD71144.1 (Genbank) | URP |
| Escherichia coli B5-2 Taxon ID: 1151224 | 537008595 | ERD81664.1 (Genbank) | URP |
| Escherichia coli B7-1 Taxon ID: 1151215 | 536854148 | ERC36965.1 (Genbank) | URP |
| Escherichia coli B7-2 Taxon ID: 1151216 | 536863022 | ERC45565.1 (Genbank) | URP |
| Escherichia coli B799 Taxon ID: 550682 | 385707903 | EIG44928.1 (Genbank) | URP |
| Escherichia coli B7A Taxon ID: 340184 | 664694887 | AIF61540.1 (Genbank) | URP |
| Escherichia coli B7A Taxon ID: 340184 | 190902593 | EDV62326.1 (Genbank) | URP |
| Escherichia coli B83 Taxon ID: 1151239 | 537012109 | ERD84979.1 (Genbank) | URP |
| Escherichia coli B84 Taxon ID: 1151240 | 537017137 | ERD89732.1 (Genbank) | URP |
| Escherichia coli B85 Taxon ID: 1151241 | 537024356 | ERD96502.1 (Genbank) | URP |
| Escherichia coli B86 Taxon ID: 1151242 | 537024092 | ERD96253.1 (Genbank) | URP |
| Escherichia coli B90 Taxon ID: 1151236 | 537068396 | ERE37746.1 (Genbank) | URP |
| Escherichia coli B93 Taxon ID: 1151231 | 536864356 | ERC46876.1 (Genbank) | URP |
| Escherichia coli B94 Taxon ID: 1151232 | 536869983 | ERC52336.1 (Genbank) | URP |
| Escherichia coli B95 Taxon ID: 1151233 | 536878035 | ERC60123.1 (Genbank) | URP |
| Escherichia coli BCE001_MS16 Taxon ID: 1116037 | 476376332 | EMX93445.1 (Genbank) | URP |
| Escherichia coli BCE006_MS-23 Taxon ID: 1125637 | 477078828 | END60202.1 (Genbank) | URP |
| Escherichia coli BCE007_MS-11 Taxon ID: 1116113 | 476729876 | ENA34638.1 (Genbank) | URP |
| Escherichia coli BCE008_MS-01 Taxon ID: 1116110 | 476820896 | ENB20749.1 (Genbank) | URP |
| Escherichia coli BCE008_MS-13 Taxon ID: 1116471 | 476713987 | ENA18991.1 (Genbank) | URP |
| Escherichia coli BCE032_MS-12 Taxon ID: 1116117 | 476841661 | ENB40451.1 (Genbank) | URP |
| Escherichia coli BIDMC 15 Taxon ID: 1328440 | 575623501 | ETY22453.1 (Genbank) | URP |
| Escherichia coli BIDMC 17A Taxon ID: 1328441 | 575615586 | ETY14620.1 (Genbank) | URP |
| Escherichia coli BIDMC 17B Taxon ID: 1328442 | 575611456 | ETY10563.1 (Genbank) | URP |
| Escherichia coli BIDMC 19A Taxon ID: 1328443 | 575605230 | ETY04387.1 (Genbank) | URP |
| Escherichia coli BIDMC 19B Taxon ID: 1328444 | 575602041 | ETY01221.1 (Genbank) | URP |
| Escherichia coli BIDMC 19C Taxon ID: 1328445 | 550205002 | ERO95464.1 (Genbank) | URP |
| Escherichia coli BIDMC 20A Taxon ID: 1328446 | 575598550 | ETX97769.1 (Genbank) | URP |
| Escherichia coli BIDMC 20B Taxon ID: 1328447 | 575594375 | ETX93640.1 (Genbank) | URP |
| Escherichia coli BIDMC 2B Taxon ID: 1328436 | 575641628 | ETY40399.1 (Genbank) | URP |
| Escherichia coli BIDMC 3 Taxon ID: 1328437 | 575635519 | ETY34331.1 (Genbank) | URP |
| Escherichia coli BIDMC 37 Taxon ID: 1328448 | 554998504 | ESL43043.1 (Genbank) | URP |
| Escherichia coli BIDMC 38 Taxon ID: 1328449 | 554999560 | ESL44093.1 (Genbank) | URP |
| Escherichia coli BIDMC 39 Taxon ID: 1328450 | 554981231 | ESL25901.1 (Genbank) | URP |
| Escherichia coli BIDMC 43a Taxon ID: 1400156 | 575584457 | ETX83822.1 (Genbank) | URP |
| Escherichia coli BIDMC 43b Taxon ID: 1400157 | 575575246 | ETX74735.1 (Genbank) | URP |
| Escherichia coli BIDMC 49a Taxon ID: 1400158 | 575655121 | ETY53711.1 (Genbank) | URP |
| Escherichia coli BIDMC 49b Taxon ID: 1400159 | 575653279 | ETY51902.1 (Genbank) | URP |
| Escherichia coli BIDMC 58 Taxon ID: 1438668 | 635797189 | KDF78542.1 (Genbank) | URP |
| Escherichia coli BIDMC 59 Taxon ID: 1438669 | 635789714 | KDF71082.1 (Genbank) | URP |
| Escherichia coli BIDMC 6 Taxon ID: 1328438 | 575665077 | ETY63512.1 (Genbank) | URP |
| Escherichia coli BIDMC 62 Taxon ID: 1438670 | 635801067 | KDF82378.1 (Genbank) | URP |
| Escherichia coli BIDMC 63 Taxon ID: 1438671 | 635801709 | KDF83019.1 (Genbank) | URP |
| Escherichia coli BIDMC 64 Taxon ID: 1438672 | 635811666 | KDF92953.1 (Genbank) | URP |
| Escherichia coli BIDMC 65 Taxon ID: 1438673 | 635826222 | KDG07402.1 (Genbank) | URP |
| Escherichia coli BIDMC 70 Taxon ID: 1438674 | 635819755 | KDG00984.1 (Genbank) | URP |
| Escherichia coli BIDMC 71 Taxon ID: 1438675 | 635827668 | KDG08822.1 (Genbank) | URP |
| Escherichia coli BIDMC 72 Taxon ID: 1438676 | 635830317 | KDG11446.1 (Genbank) | URP |
| Escherichia coli BIDMC 73 Taxon ID: 1438677 | 635830813 | KDG11940.1 (Genbank) | URP |
| Escherichia coli BIDMC 74 Taxon ID: 1438678 | 635834413 | KDG15506.1 (Genbank) | URP |
| Escherichia coli BIDMC 75 Taxon ID: 1438679 | 635851786 | KDG32746.1 (Genbank) | URP |
| Escherichia coli BIDMC 76 Taxon ID: 1438680 | 635850206 | KDG31174.1 (Genbank) | URP |
| Escherichia coli BIDMC 77 Taxon ID: 1438681 | 635856233 | KDG37141.1 (Genbank) | URP |
| Escherichia coli BIDMC 78 Taxon ID: 1438682 | 635854732 | KDG35648.1 (Genbank) | URP |
| Escherichia coli BIDMC 79 Taxon ID: 1438683 | 635865530 | KDG46376.1 (Genbank) | URP |
| Escherichia coli BIDMC 82 Taxon ID: 1445862 | 612274352 | EZQ67788.1 (Genbank) | URP |
| Escherichia coli BIDMC 83 Taxon ID: 1445863 | 612268671 | EZQ62202.1 (Genbank) | URP |
| Escherichia coli BIDMC 9 Taxon ID: 1328439 | 575628835 | ETY27730.1 (Genbank) | URP |
| Escherichia coli BL21(DE3) Taxon ID: 469008 | 253976805 | ACT42475.1 (Genbank) | URP |
| Escherichia coli BW25113 Taxon ID: 679895 | 682118191 | AIN31117.1 (Genbank) | URP |
| Escherichia coli BW2952 Taxon ID: 595496 | 238861658 | ACR63656.1 (Genbank) | URP |
| Escherichia coli BWH 24 Taxon ID: 1328435 | 550198933 | ERO89518.1 (Genbank) | URP |
| Escherichia coli BWH 32 Taxon ID: 1328432 | 555097641 | ESM40755.1 (Genbank) | URP |
| Escherichia coli BWH 34 Taxon ID: 1328433 | 575645200 | ETY43878.1 (Genbank) | URP |
| Escherichia coli BWH 40 Taxon ID: 1328434 | 575651373 | ETY50024.1 (Genbank) | URP |
| Escherichia coli C-34666 Taxon ID: 1116002 | 476100703 | EMV24058.1 (Genbank) | URP |
| Escherichia coli C691-71 (14b) Taxon ID: 1010817 | 747266904 | KIG28206.1 (Genbank) | URP |
| Escherichia coli CB7326 Taxon ID: 1005559 | 408186852 | EKI12863.1 (Genbank) | URP |
| Escherichia coli CE418 Taxon ID: 1331063 | 557937852 | EST00824.1 (Genbank) | URP |
| Escherichia coli CE516 Taxon ID: 1331064 | 557932682 | ESS96273.1 (Genbank) | URP |
| Escherichia coli CE549 Taxon ID: 1331065 | 557924430 | ESS88919.1 (Genbank) | URP |
| Escherichia coli CFT073 Taxon ID: 199310 | 26107007 | AAN79191.1 (Genbank) | URP |
| Escherichia coli CHS 68 Taxon ID: 1438684 | 635872829 | KDG53615.1 (Genbank) | URP |
| Escherichia coli CHS 69 Taxon ID: 1438685 | 635872013 | KDG52805.1 (Genbank) | URP |
| Escherichia coli CHS 77 Taxon ID: 1438686 | 635879388 | KDG60114.1 (Genbank) | URP |
| Escherichia coli MT8 Taxon ID: 752790 | 388420819 | EIL80486.1 (Genbank) | URP |
| Shigella sp. D9 Taxon ID: 556266 | 332104116 | EGJ07462.1 (Genbank) | URP |
| Escherichia coli DEC10A Taxon ID: 868180 | 378106972 | EHW68598.1 (Genbank) | URP |
| Escherichia coli DEC10B Taxon ID: 868181 | 378115672 | EHW77207.1 (Genbank) | URP |
| Escherichia coli DEC10C Taxon ID: 868182 | 378118358 | EHW79861.1 (Genbank) | URP |
| Escherichia coli DEC10D Taxon ID: 868183 | 378121334 | EHW82788.1 (Genbank) | URP |
| Escherichia coli DEC10E Taxon ID: 868184 | 378133131 | EHW94478.1 (Genbank) | URP |
| Escherichia coli DEC10F Taxon ID: 868185 | 378138908 | EHX00158.1 (Genbank) | URP |
| Escherichia coli DEC11A Taxon ID: 868186 | 378136059 | EHW97359.1 (Genbank) | URP |
| Escherichia coli DEC11B Taxon ID: 868187 | 378146326 | EHX07478.1 (Genbank) | URP |
| Escherichia coli DEC11C Taxon ID: 868188 | 378156428 | EHX17478.1 (Genbank) | URP |
| Escherichia coli DEC11D Taxon ID: 868189 | 378152624 | EHX13717.1 (Genbank) | URP |
| Escherichia coli DEC11E Taxon ID: 868190 | 378160861 | EHX21847.1 (Genbank) | URP |
| Escherichia coli DEC12A Taxon ID: 868191 | 378174707 | EHX35530.1 (Genbank) | URP |
| Escherichia coli DEC12B Taxon ID: 868192 | 378174225 | EHX35052.1 (Genbank) | URP |
| Escherichia coli DEC12C Taxon ID: 868193 | 378176145 | EHX36952.1 (Genbank) | URP |
| Escherichia coli DEC12D Taxon ID: 868194 | 378190202 | EHX50787.1 (Genbank) | URP |
| Escherichia coli DEC12E Taxon ID: 868195 | 378193601 | EHX54133.1 (Genbank) | URP |
| Escherichia coli DEC13A Taxon ID: 868196 | 378189823 | EHX50412.1 (Genbank) | URP |
| Escherichia coli DEC13B Taxon ID: 868197 | 378204358 | EHX64774.1 (Genbank) | URP |
| Escherichia coli DEC13C Taxon ID: 868198 | 378208760 | EHX69140.1 (Genbank) | URP |
| Escherichia coli DEC13D Taxon ID: 868199 | 378208505 | EHX68889.1 (Genbank) | URP |
| Escherichia coli DEC14A Taxon ID: 868201 | 378222104 | EHX82346.1 (Genbank) | URP |
| Escherichia coli DEC14B Taxon ID: 868202 | 378225475 | EHX85673.1 (Genbank) | URP |
| Escherichia coli DEC14C Taxon ID: 868203 | 378233331 | EHX93420.1 (Genbank) | URP |
| Escherichia coli DEC14D Taxon ID: 868204 | 378236645 | EHX96685.1 (Genbank) | URP |
| Escherichia coli DEC15A Taxon ID: 868206 | 378244830 | EHY04772.1 (Genbank) | URP |
| Escherichia coli DEC15B Taxon ID: 868207 | 378250681 | EHY10584.1 (Genbank) | URP |
| Escherichia coli DEC15D Taxon ID: 868209 | 378257457 | EHY17296.1 (Genbank) | URP |
| Escherichia coli DEC15E Taxon ID: 868210 | 378261140 | EHY20936.1 (Genbank) | URP |
| Escherichia coli DEC1A Taxon ID: 868133 | 377848821 | EHU13797.1 (Genbank) | URP |
| Escherichia coli DEC1B Taxon ID: 868134 | 377853432 | EHU18331.1 (Genbank) | URP |
| Escherichia coli DEC1C Taxon ID: 868135 | 377851097 | EHU16052.1 (Genbank) | URP |
| Escherichia coli DEC1D Taxon ID: 868136 | 377862930 | EHU27737.1 (Genbank) | URP |
| Escherichia coli DEC1E Taxon ID: 868137 | 377867042 | EHU31806.1 (Genbank) | URP |
| Escherichia coli DEC2A Taxon ID: 868138 | 377868399 | EHU33143.1 (Genbank) | URP |
| Escherichia coli DEC2B Taxon ID: 868139 | 377879343 | EHU43916.1 (Genbank) | URP |
| Escherichia coli DEC2C Taxon ID: 868140 | 377885451 | EHU49946.1 (Genbank) | URP |
| Escherichia coli DEC2D Taxon ID: 868141 | 377883904 | EHU48422.1 (Genbank) | URP |
| Escherichia coli DEC2E Taxon ID: 868142 | 377898592 | EHU62952.1 (Genbank) | URP |
| Escherichia coli DEC3A Taxon ID: 868143 | 377900000 | EHU64338.1 (Genbank) | URP |
| Escherichia coli DEC3B Taxon ID: 868144 | 377902058 | EHU66367.1 (Genbank) | URP |
| Escherichia coli DEC3C Taxon ID: 868145 | 377913787 | EHU77919.1 (Genbank) | URP |
| Escherichia coli DEC3D Taxon ID: 868146 | 377918013 | EHU82067.1 (Genbank) | URP |
| Escherichia coli DEC3E Taxon ID: 868147 | 377920438 | EHU84459.1 (Genbank) | URP |
| Escherichia coli DEC3F Taxon ID: 868148 | 377931688 | EHU95547.1 (Genbank) | URP |
| Escherichia coli DEC4A Taxon ID: 868149 | 377934066 | EHU97906.1 (Genbank) | URP |
| Escherichia coli DEC4B Taxon ID: 868150 | 377939309 | EHV03065.1 (Genbank) | URP |
| Escherichia coli DEC4D Taxon ID: 868152 | 377951412 | EHV15031.1 (Genbank) | URP |
| Escherichia coli DEC4E Taxon ID: 868153 | 377954186 | EHV17746.1 (Genbank) | URP |
| Escherichia coli DEC4F Taxon ID: 868154 | 377967589 | EHV30995.1 (Genbank) | URP |
| Escherichia coli DEC5A Taxon ID: 868155 | 377965503 | EHV28922.1 (Genbank) | URP |
| Escherichia coli DEC5B Taxon ID: 868156 | 377972529 | EHV35878.1 (Genbank) | URP |
| Escherichia coli DEC5C Taxon ID: 868157 | 377980269 | EHV43534.1 (Genbank) | URP |
| Escherichia coli DEC5D Taxon ID: 868158 | 377980526 | EHV43790.1 (Genbank) | URP |
| Escherichia coli DEC5E Taxon ID: 868159 | 377987414 | EHV50600.1 (Genbank) | URP |
| Escherichia coli DEC6A Taxon ID: 868160 | 377999366 | EHV62449.1 (Genbank) | URP |
| Escherichia coli DEC6B Taxon ID: 868161 | 378001335 | EHV64394.1 (Genbank) | URP |
| Escherichia coli DEC6E Taxon ID: 868164 | 378016465 | EHV79346.1 (Genbank) | URP |
| Escherichia coli DEC7A Taxon ID: 868165 | 378018472 | EHV81329.1 (Genbank) | URP |
| Escherichia coli DEC7B Taxon ID: 868166 | 378037305 | EHV99833.1 (Genbank) | URP |
| Escherichia coli DEC7C Taxon ID: 868167 | 378028062 | EHV90687.1 (Genbank) | URP |
| Escherichia coli DEC7D Taxon ID: 868168 | 378032581 | EHV95162.1 (Genbank) | URP |
| Escherichia coli DEC7E Taxon ID: 868169 | 378042513 | EHW04962.1 (Genbank) | URP |
| Escherichia coli DEC8A Taxon ID: 868170 | 378052373 | EHW14681.1 (Genbank) | URP |
| Escherichia coli DEC8B Taxon ID: 868171 | 378056528 | EHW18768.1 (Genbank) | URP |
| Escherichia coli DEC8D Taxon ID: 868173 | 378068164 | EHW30268.1 (Genbank) | URP |
| Escherichia coli DEC8E Taxon ID: 868174 | 378072943 | EHW35000.1 (Genbank) | URP |
| Escherichia coli DEC9A Taxon ID: 868175 | 378080941 | EHW42897.1 (Genbank) | URP |
| Escherichia coli DEC9B Taxon ID: 868176 | 378082042 | EHW43988.1 (Genbank) | URP |
| Escherichia coli DEC9C Taxon ID: 868177 | 378089742 | EHW51583.1 (Genbank) | URP |
| Escherichia coli DEC9D Taxon ID: 868178 | 378094359 | EHW56157.1 (Genbank) | URP |
| Escherichia coli DEC9E Taxon ID: 868179 | 378101435 | EHW63121.1 (Genbank) | URP |
| Escherichia coli DH1 Taxon ID: 536056 | 260450205 | ACX40627.1 (Genbank) | URP |
| Escherichia coli DORA_A_5_14_21 Taxon ID: 1403943 | 566287321 | ETJ28063.1 (Genbank) | URP |
| Escherichia coli DSM 30083 Taxon ID: 866789 | 668706163 | KFB92055.1 (Genbank) | URP |
| Escherichia coli E101 Taxon ID: 550685 | 371613876 | EHO02364.1 (Genbank) | URP |
| Escherichia coli E110019 Taxon ID: 340186 | 192957596 | EDV88041.1 (Genbank) | URP |
| Escherichia coli 1140 Taxon ID: 1453472 | 662007237 | KEP83807.1 (Genbank) | URP |
| Escherichia coli E1167 Taxon ID: 656372 | 324116702 | EGC10617.1 (Genbank) | URP |
| Escherichia coli E128010 Taxon ID: 670894 | 323158919 | EFZ44930.1 (Genbank) | URP |
| Escherichia coli E1520 Taxon ID: 656374 | 323938390 | EGB34644.1 (Genbank) | URP |
| Escherichia coli 1728 Taxon ID: 1457300 | 608092197 | EZG31268.1 (Genbank) | URP |
| Escherichia coli E1777 Taxon ID: 1331061 | 559785960 | ESV03118.1 (Genbank) | URP |
| Escherichia coli E22 Taxon ID: 340185 | 192931042 | EDV83646.1 (Genbank) | URP |
| Escherichia coli E2265 Taxon ID: 1331062 | 514335278 | EPH47774.1 (Genbank) | URP |
| Escherichia coli E482 Taxon ID: 550687 | 323943043 | EGB39202.1 (Genbank) | URP |
| Escherichia coli EC096/10 Taxon ID: 1280937 | 582996442 | EWC57108.1 (Genbank) | URP |
| Escherichia coli EC1734 Taxon ID: 1005536 | 390909536 | EIP68310.1 (Genbank) | URP |
| Escherichia coli EC1735 Taxon ID: 1005537 | 408238872 | EKI61653.1 (Genbank) | URP |
| Escherichia coli EC1736 Taxon ID: 1005538 | 408248921 | EKI70903.1 (Genbank) | URP |
| Escherichia coli EC1737 Taxon ID: 1005539 | 408252716 | EKI74346.1 (Genbank) | URP |
| Escherichia coli EC1738 Taxon ID: 1005535 | 390897237 | EIP56577.1 (Genbank) | URP |
| Escherichia coli EC1846 Taxon ID: 1005541 | 408259075 | EKI80277.1 (Genbank) | URP |
| Escherichia coli EC1847 Taxon ID: 1005542 | 408268251 | EKI88645.1 (Genbank) | URP |
| Escherichia coli EC1848 Taxon ID: 1005543 | 408269868 | EKI90102.1 (Genbank) | URP |
| Escherichia coli EC1849 Taxon ID: 1005544 | 408278780 | EKI98470.1 (Genbank) | URP |
| Escherichia coli EC1850 Taxon ID: 1005545 | 408284974 | EKJ04026.1 (Genbank) | URP |
| Escherichia coli EC1856 Taxon ID: 1005546 | 408287525 | EKJ06390.1 (Genbank) | URP |
| Escherichia coli EC1862 Taxon ID: 1005547 | 408300314 | EKJ18017.1 (Genbank) | URP |
| Escherichia coli EC1863 Taxon ID: 1005548 | 390924732 | EIP82480.1 (Genbank) | URP |
| Escherichia coli EC1865 Taxon ID: 1005550 | 408309627 | EKJ26778.1 (Genbank) | URP |
| Escherichia coli EC1866 Taxon ID: 1005551 | 408317541 | EKJ33773.1 (Genbank) | URP |
| Escherichia coli EC1868 Taxon ID: 1005552 | 408317271 | EKJ33509.1 (Genbank) | URP |
| Escherichia coli EC1869 Taxon ID: 1005553 | 408331269 | EKJ46460.1 (Genbank) | URP |
| Escherichia coli EC1870 Taxon ID: 1005554 | 408338001 | EKJ52675.1 (Genbank) | URP |
| Escherichia coli EC4100B Taxon ID: 945434 | 320198246 | EFW72850.1 (Genbank) | URP |
| Escherichia coli EC4196 Taxon ID: 1005524 | 390839852 | EIP03931.1 (Genbank) | URP |
| Escherichia coli EC4203 Taxon ID: 1005523 | 390836958 | EIP01423.1 (Genbank) | URP |
| Escherichia coli EC4402 Taxon ID: 1005530 | 390885895 | EIP46067.1 (Genbank) | URP |
| Escherichia coli EC4421 Taxon ID: 1005527 | 390859356 | EIP21707.1 (Genbank) | URP |
| Escherichia coli EC4422 Taxon ID: 1005528 | 390871434 | EIP32845.1 (Genbank) | URP |
| Escherichia coli EC4436 Taxon ID: 1005532 | 390894338 | EIP53856.1 (Genbank) | URP |
| Escherichia coli EC4437 Taxon ID: 1005533 | 390910260 | EIP69014.1 (Genbank) | URP |
| Escherichia coli EC4439 Taxon ID: 1005531 | 390887862 | EIP47783.1 (Genbank) | URP |
| Escherichia coli EC96038 Taxon ID: 1005560 | 408191612 | EKI17212.1 (Genbank) | URP |
| Escherichia coli ECA-0157 Taxon ID: 758829 | 558723056 | EST74141.1 (Genbank) | URP |
| Escherichia coli ECA-727 Taxon ID: 758830 | 558728254 | EST78586.1 (Genbank) | URP |
| Escherichia coli ECC-1470 Taxon ID: 758831 | 752305036 | AJG07655.1 (Genbank) | URP |
| Escherichia coli ECC-1470 Taxon ID: 758831 | 558725344 | EST76118.1 (Genbank) | URP |
| Escherichia coli ECC-Z Taxon ID: 758832 | 558709086 | EST60465.1 (Genbank) | URP |
| Escherichia coli EPECa12 Taxon ID: 670896 | 391309352 | EIQ67025.1 (Genbank) | URP |
| Escherichia coli EPECa14 Taxon ID: 670893 | 323153651 | EFZ39899.1 (Genbank) | URP |
| Escherichia coli ER2796 Taxon ID: 1245474 | 730585866 | AIZ31152.1 (Genbank) | URP |
| Escherichia coli Envira 10/1 Taxon ID: 1115998 | 476354892 | EMX72184.1 (Genbank) | URP |
| Escherichia coli Envira 8/11 Taxon ID: 1115997 | 476360484 | EMX77737.1 (Genbank) | URP |
| Escherichia coli F11 Taxon ID: 340197 | 190907428 | EDV67025.1 (Genbank) | URP |
| Escherichia coli FAP1 Taxon ID: 1412834 | 695937594 | AIT33825.1 (Genbank) | URP |
| Escherichia coli FDA504 Taxon ID: 1005470 | 408102215 | EKH34631.1 (Genbank) | URP |
| Escherichia coli FDA506 Taxon ID: 1005474 | 408089947 | EKH23239.1 (Genbank) | URP |
| Escherichia coli FDA507 Taxon ID: 1005472 | 408095370 | EKH28354.1 (Genbank) | URP |
| Escherichia coli FDA517 Taxon ID: 1005473 | 390652615 | EIN30817.1 (Genbank) | URP |
| Escherichia coli FRIK1985 Taxon ID: 1005476 | 390673480 | EIN49722.1 (Genbank) | URP |
| Escherichia coli FRIK1990 Taxon ID: 1005478 | 390672694 | EIN48971.1 (Genbank) | URP |
| Escherichia coli FRIK1996 Taxon ID: 1005475 | 390650634 | EIN29034.1 (Genbank) | URP |
| Escherichia coli FRIK1999 Taxon ID: 1005477 | 408109885 | EKH41743.1 (Genbank) | URP |
| Escherichia coli FRIK2001 Taxon ID: 1005479 | 408132179 | EKH62175.1 (Genbank) | URP |
| Escherichia coli FRIK920 Taxon ID: 1005483 | 408075514 | EKH09746.1 (Genbank) | URP |
| Escherichia coli FVEC1302 Taxon ID: 656379 | 298279364 | EFI20872.1 (Genbank) | URP |
| Escherichia coli FVEC1412 Taxon ID: 656380 | 291428476 | EFF01501.1 (Genbank) | URP |
| Escherichia coli G3/10 Taxon ID: 1455601 | 699380023 | KGM59856.1 (Genbank) | URP |
| Escherichia coli G58-1 Taxon ID: 754078 | 345380896 | EGX12788.1 (Genbank) | URP |
| Escherichia coli GM4792 Taxon ID: 1411691 | 827617495 | AKK13478.1 (Genbank) | |
| Escherichia coli GM4792 Taxon ID: 1411691 | 827621192 | AKK16926.1 (Genbank) | |
| Escherichia coli H120 Taxon ID: 656383 | 323945103 | EGB41165.1 (Genbank) | URP |
| Escherichia coli H252 Taxon ID: 656388 | 323952794 | EGB48662.1 (Genbank) | URP |
| Escherichia coli H263 Taxon ID: 656390 | 323958386 | EGB54092.1 (Genbank) | URP |
| Escherichia coli H299 Taxon ID: 656393 | 331080730 | EGI51904.1 (Genbank) | URP |
| Escherichia coli H397 Taxon ID: 656398 | 371611141 | EHN99667.1 (Genbank) | URP |
| Escherichia coli H489 Taxon ID: 656404 | 323963197 | EGB58765.1 (Genbank) | URP |
| Escherichia coli H494 Taxon ID: 656405 | 371593756 | EHN82632.1 (Genbank) | URP |
| Escherichia coli H591 Taxon ID: 656408 | 331076344 | EGI47626.1 (Genbank) | URP |
| Escherichia coli H730 Taxon ID: 656413 | 385712735 | EIG49677.1 (Genbank) | URP |
| Escherichia coli H736 Taxon ID: 656414 | 331037928 | EGI10148.1 (Genbank) | URP |
| Escherichia coli HM605 Taxon ID: 752789 | 388415825 | EIL75736.1 (Genbank) | URP |
| Escherichia coli HS Taxon ID: 331112 | 157065803 | ABV05058.1 (Genbank) | URP |
| Escherichia coli HVH 1 (4-6876161) Taxon ID: 1280956 | 534546898 | EQN06516.1 (Genbank) | URP |
| Escherichia coli HVH 10 (4-6832164) Taxon ID: 1280964 | 534580910 | EQN39492.1 (Genbank) | URP |
| Escherichia coli HVH 100 (4-2850729) Taxon ID: 1281036 | 534885489 | EQQ35537.1 (Genbank) | URP |
| Escherichia coli HVH 102 (4-6906788) Taxon ID: 1281038 | 534893233 | EQQ43154.1 (Genbank) | URP |
| Escherichia coli HVH 103 (4-5904188) Taxon ID: 1281039 | 534906339 | EQQ55933.1 (Genbank) | URP |
| Escherichia coli HVH 106 (4-6881831) Taxon ID: 1281042 | 534910659 | EQQ60220.1 (Genbank) | URP |
| Escherichia coli HVH 107 (4-5860571) Taxon ID: 1281043 | 534931664 | EQQ80819.1 (Genbank) | URP |
| Escherichia coli HVH 108 (4-6924867) Taxon ID: 1281044 | 556160239 | ESP48316.1 (Genbank) | URP |
| Escherichia coli HVH 109 (4-6977162) Taxon ID: 1281045 | 534930840 | EQQ80038.1 (Genbank) | URP |
| Escherichia coli HVH 110 (4-6978754) Taxon ID: 1281046 | 534918605 | EQQ67974.1 (Genbank) | URP |
| Escherichia coli HVH 111 (4-7039018) Taxon ID: 1281047 | 534929465 | EQQ78675.1 (Genbank) | URP |
| Escherichia coli HVH 112 (4-5987253) Taxon ID: 1281048 | 534942992 | EQQ91976.1 (Genbank) | URP |
| Escherichia coli HVH 113 (4-7535473) Taxon ID: 1281049 | 534946940 | EQQ95841.1 (Genbank) | URP |
| Escherichia coli HVH 114 (4-7037740) Taxon ID: 1281050 | 534944362 | EQQ93335.1 (Genbank) | URP |
| Escherichia coli HVH 116 (4-6879942) Taxon ID: 1281053 | 534960845 | EQR09490.1 (Genbank) | URP |
| Escherichia coli HVH 117 (4-6857191) Taxon ID: 1281054 | 534972075 | EQR20554.1 (Genbank) | URP |
| Escherichia coli HVH 118 (4-7345399) Taxon ID: 1281055 | 534978352 | EQR26751.1 (Genbank) | URP |
| Escherichia coli HVH 119 (4-6879578) Taxon ID: 1281056 | 534975107 | EQR23551.1 (Genbank) | URP |
| Escherichia coli HVH 12 (4-7653042) Taxon ID: 1280965 | 556135321 | ESP23755.1 (Genbank) | URP |
| Escherichia coli HVH 120 (4-6978681) Taxon ID: 1281057 | 534985058 | EQR33355.1 (Genbank) | URP |
| Escherichia coli HVH 121 (4-6877826) Taxon ID: 1281058 | 534996443 | EQR44554.1 (Genbank) | URP |
| Escherichia coli HVH 122 (4-6851606) Taxon ID: 1281059 | 534987790 | EQR36065.1 (Genbank) | URP |
| Escherichia coli HVH 125 (4-2634716) Taxon ID: 1281060 | 534998588 | EQR46669.1 (Genbank) | URP |
| Escherichia coli HVH 126 (4-6034225) Taxon ID: 1281061 | 535002524 | EQR50491.1 (Genbank) | URP |
| Escherichia coli HVH 127 (4-7303629) Taxon ID: 1281062 | 535007497 | EQR55321.1 (Genbank) | URP |
| Escherichia coli HVH 128 (4-7030436) Taxon ID: 1281063 | 535015486 | EQR63002.1 (Genbank) | URP |
| Escherichia coli HVH 13 (4-7634056) Taxon ID: 1280966 | 534596236 | EQN54125.1 (Genbank) | URP |
| Escherichia coli HVH 130 (4-7036876) Taxon ID: 1281064 | 535021813 | EQR69250.1 (Genbank) | URP |
| Escherichia coli HVH 132 (4-6876862) Taxon ID: 1281065 | 535021512 | EQR68952.1 (Genbank) | URP |
| Escherichia coli HVH 133 (4-4466519) Taxon ID: 1281066 | 535027982 | EQR75159.1 (Genbank) | URP |
| Escherichia coli HVH 134 (4-6073441) Taxon ID: 1281067 | 535024679 | EQR71878.1 (Genbank) | URP |
| Escherichia coli HVH 135 (4-4449320) Taxon ID: 1281068 | 535029657 | EQR76831.1 (Genbank) | URP |
| Escherichia coli HVH 136 (4-5970458) Taxon ID: 1281069 | 556133029 | ESP21484.1 (Genbank) | URP |
| Escherichia coli HVH 137 (4-2124971) Taxon ID: 1281070 | 535042794 | EQR89836.1 (Genbank) | URP |
| Escherichia coli HVH 138 (4-6066704) Taxon ID: 1281071 | 535049515 | EQR96471.1 (Genbank) | URP |
| Escherichia coli HVH 139 (4-3192644) Taxon ID: 1281072 | 535054521 | EQS01382.1 (Genbank) | URP |
| Escherichia coli HVH 140 (4-5894387) Taxon ID: 1281073 | 535060218 | EQS07035.1 (Genbank) | URP |
| Escherichia coli HVH 141 (4-5995973) Taxon ID: 1281074 | 535061827 | EQS08619.1 (Genbank) | URP |
| Escherichia coli HVH 142 (4-5627451) Taxon ID: 1281075 | 535077211 | EQS23779.1 (Genbank) | URP |
| Escherichia coli HVH 143 (4-5674999) Taxon ID: 1281076 | 535070754 | EQS17396.1 (Genbank) | URP |
| Escherichia coli HVH 144 (4-4451937) Taxon ID: 1281077 | 535081783 | EQS28317.1 (Genbank) | URP |
| Escherichia coli HVH 145 (4-5672112) Taxon ID: 1281078 | 535076895 | EQS23464.1 (Genbank) | URP |
| Escherichia coli HVH 146 (4-3189767) Taxon ID: 1281079 | 535096423 | EQS42759.1 (Genbank) | URP |
| Escherichia coli HVH 147 (4-5893887) Taxon ID: 1281080 | 535090422 | EQS36824.1 (Genbank) | URP |
| Escherichia coli HVH 148 (4-3192490) Taxon ID: 1281081 | 556144367 | ESP32647.1 (Genbank) | URP |
| Escherichia coli HVH 149 (4-4451880) Taxon ID: 1281082 | 535095952 | EQS42298.1 (Genbank) | URP |
| Escherichia coli HVH 150 (4-3258106) Taxon ID: 1281083 | 535112154 | EQS58372.1 (Genbank) | URP |
| Escherichia coli HVH 151 (4-5755573) Taxon ID: 1281084 | 535104386 | EQS50415.1 (Genbank) | URP |
| Escherichia coli HVH 152 (4-3447545) Taxon ID: 1281085 | 556159782 | ESP47861.1 (Genbank) | URP |
| Escherichia coli HVH 153 (3-9344314) Taxon ID: 1281086 | 535109403 | EQS55482.1 (Genbank) | URP |
| Escherichia coli HVH 154 (4-5636698) Taxon ID: 1281087 | 535125817 | EQS71622.1 (Genbank) | URP |
| Escherichia coli HVH 155 (4-4509048) Taxon ID: 1281088 | 535968738 | ERA68695.1 (Genbank) | URP |
| Escherichia coli HVH 156 (4-3206505) Taxon ID: 1281089 | 535977624 | ERA77473.1 (Genbank) | URP |
| Escherichia coli HVH 157 (4-3406229) Taxon ID: 1281090 | 535979048 | ERA78869.1 (Genbank) | URP |
| Escherichia coli HVH 158 (4-3224287) Taxon ID: 1281091 | 535115717 | EQS61720.1 (Genbank) | URP |
| Escherichia coli HVH 159 (4-5818141) Taxon ID: 1281092 | 535983563 | ERA83328.1 (Genbank) | URP |
| Escherichia coli HVH 16 (4-7649002) Taxon ID: 1280967 | 534597188 | EQN55063.1 (Genbank) | URP |
| Escherichia coli HVH 160 (4-5695937) Taxon ID: 1281093 | 535992243 | ERA91921.1 (Genbank) | URP |
| Escherichia coli HVH 161 (4-3119890) Taxon ID: 1281094 | 535123653 | EQS69502.1 (Genbank) | URP |
| Escherichia coli HVH 162 (4-5627982) Taxon ID: 1281095 | 535140846 | EQS85822.1 (Genbank) | URP |
| Escherichia coli HVH 163 (4-4697553) Taxon ID: 1281096 | 535128491 | EQS73847.1 (Genbank) | URP |
| Escherichia coli HVH 164 (4-5953081) Taxon ID: 1281097 | 535140027 | EQS85010.1 (Genbank) | URP |
| Escherichia coli HVH 167 (4-6073565) Taxon ID: 1281098 | 535145514 | EQS90449.1 (Genbank) | URP |
| Escherichia coli HVH 169 (4-1075578) Taxon ID: 1281099 | 535156737 | EQT01511.1 (Genbank) | URP |
| Escherichia coli HVH 17 (4-7473087) Taxon ID: 1280968 | 534598419 | EQN56268.1 (Genbank) | URP |
| Escherichia coli HVH 170 (4-3026949) Taxon ID: 1281100 | 535160170 | EQT04880.1 (Genbank) | URP |
| Escherichia coli HVH 171 (4-3191958) Taxon ID: 1281101 | 535159738 | EQT04464.1 (Genbank) | URP |
| Escherichia coli HVH 172 (4-3248542) Taxon ID: 1281102 | 535172591 | EQT17172.1 (Genbank) | URP |
| Escherichia coli HVH 173 (3-9175482) Taxon ID: 1281103 | 535168587 | EQT13231.1 (Genbank) | URP |
| Escherichia coli HVH 175 (4-3405184) Taxon ID: 1281104 | 535185445 | EQT29871.1 (Genbank) | URP |
| Escherichia coli HVH 176 (4-3428664) Taxon ID: 1281105 | 535182622 | EQT27066.1 (Genbank) | URP |
| Escherichia coli HVH 177 (4-2876612) Taxon ID: 1281106 | 565510064 | ETF21949.1 (Genbank) | URP |
| Escherichia coli HVH 178 (4-3189163) Taxon ID: 1281107 | 556145851 | ESP34109.1 (Genbank) | URP |
| Escherichia coli HVH 18 (4-8589585) Taxon ID: 1280969 | 534614126 | EQN71044.1 (Genbank) | URP |
| Escherichia coli HVH 182 (4-0985554) Taxon ID: 1281109 | 535198902 | EQT43113.1 (Genbank) | URP |
| Escherichia coli HVH 183 (4-3205932) Taxon ID: 1281110 | 535195614 | EQT39856.1 (Genbank) | URP |
| Escherichia coli HVH 184 (4-3343286) Taxon ID: 1281111 | 535204654 | EQT48751.1 (Genbank) | URP |
| Escherichia coli HVH 185 (4-2876639) Taxon ID: 1281112 | 535210324 | EQT54299.1 (Genbank) | URP |
| Escherichia coli HVH 186 (4-3405044) Taxon ID: 1281113 | 535218504 | EQT62348.1 (Genbank) | URP |
| Escherichia coli HVH 187 (4-4471660) Taxon ID: 1281114 | 535212524 | EQT56456.1 (Genbank) | URP |
| Escherichia coli HVH 188 (4-2356988) Taxon ID: 1281115 | 535219726 | EQT63530.1 (Genbank) | URP |
| Escherichia coli HVH 189 (4-3220125) Taxon ID: 1281116 | 535235372 | EQT79181.1 (Genbank) | URP |
| Escherichia coli HVH 19 (4-7154984) Taxon ID: 1280970 | 534616870 | EQN73733.1 (Genbank) | URP |
| Escherichia coli HVH 190 (4-3255514) Taxon ID: 1281117 | 535225556 | EQT69227.1 (Genbank) | URP |
| Escherichia coli HVH 191 (3-9341900) Taxon ID: 1281118 | 535241161 | EQT84852.1 (Genbank) | URP |
| Escherichia coli HVH 192 (4-3054470) Taxon ID: 1281119 | 535243541 | EQT87185.1 (Genbank) | URP |
| Escherichia coli HVH 193 (4-3331423) Taxon ID: 1281120 | 535249456 | EQT93028.1 (Genbank) | URP |
| Escherichia coli HVH 194 (4-2356805) Taxon ID: 1281121 | 535255275 | EQT98780.1 (Genbank) | URP |
| Escherichia coli HVH 195 (3-7155360) Taxon ID: 1281122 | 535257354 | EQU00821.1 (Genbank) | URP |
| Escherichia coli HVH 196 (4-4530470) Taxon ID: 1281123 | 535257513 | EQU00977.1 (Genbank) | URP |
| Escherichia coli HVH 197 (4-4466217) Taxon ID: 1281124 | 535282862 | EQU25990.1 (Genbank) | URP |
| Escherichia coli HVH 198 (4-3206106) Taxon ID: 1281125 | 535272972 | EQU16237.1 (Genbank) | URP |
| Escherichia coli HVH 199 (4-5670322) Taxon ID: 1281126 | 535271296 | EQU14573.1 (Genbank) | URP |
| Escherichia coli HVH 2 (4-6943160) Taxon ID: 1280957 | 534549023 | EQN08638.1 (Genbank) | URP |
| Escherichia coli HVH 20 (4-5865042) Taxon ID: 1280971 | 534607986 | EQN64733.1 (Genbank) | URP |
| Escherichia coli HVH 200 (4-4449924) Taxon ID: 1281127 | 535287510 | EQU30577.1 (Genbank) | URP |
| Escherichia coli HVH 201 (4-4459431) Taxon ID: 1281128 | 535284222 | EQU27337.1 (Genbank) | URP |
| Escherichia coli HVH 202 (4-3163997) Taxon ID: 1281129 | 535297976 | EQU40955.1 (Genbank) | URP |
| Escherichia coli HVH 203 (4-3126218) Taxon ID: 1281130 | 535295739 | EQU38746.1 (Genbank) | URP |
| Escherichia coli HVH 204 (4-3112802) Taxon ID: 1281131 | 535302447 | EQU45364.1 (Genbank) | URP |
| Escherichia coli HVH 205 (4-3094677) Taxon ID: 1281132 | 535314463 | EQU57283.1 (Genbank) | URP |
| Escherichia coli HVH 206 (4-3128229) Taxon ID: 1281133 | 535306832 | EQU49716.1 (Genbank) | URP |
| Escherichia coli HVH 207 (4-3113221) Taxon ID: 1281134 | 535318435 | EQU61240.1 (Genbank) | URP |
| Escherichia coli HVH 208 (4-3112292) Taxon ID: 1281135 | 535320046 | EQU62842.1 (Genbank) | URP |
| Escherichia coli HVH 209 (4-3062651) Taxon ID: 1281136 | 535321845 | EQU64625.1 (Genbank) | URP |
| Escherichia coli HVH 21 (4-4517873) Taxon ID: 1280972 | 534626394 | EQN83160.1 (Genbank) | URP |
| Escherichia coli HVH 210 (4-3042480) Taxon ID: 1281137 | 535993528 | ERA93181.1 (Genbank) | URP |
| Escherichia coli HVH 211 (4-3041891) Taxon ID: 1281138 | 535332441 | EQU75145.1 (Genbank) | URP |
| Escherichia coli HVH 212 (3-9305343) Taxon ID: 1281139 | 535337854 | EQU80457.1 (Genbank) | URP |
| Escherichia coli HVH 213 (4-3042928) Taxon ID: 1281140 | 535346045 | EQU88524.1 (Genbank) | URP |
| Escherichia coli HVH 214 (4-3062198) Taxon ID: 1281141 | 565514392 | ETF26253.1 (Genbank) | URP |
| Escherichia coli HVH 215 (4-3008371) Taxon ID: 1281142 | 535343918 | EQU86463.1 (Genbank) | URP |
| Escherichia coli HVH 216 (4-3042952) Taxon ID: 1281143 | 535356929 | EQU99289.1 (Genbank) | URP |
| Escherichia coli HVH 217 (4-1022806) Taxon ID: 1281144 | 535355322 | EQU97692.1 (Genbank) | URP |
| Escherichia coli HVH 218 (4-4500903) Taxon ID: 1281145 | 535361840 | EQV04112.1 (Genbank) | URP |
| Escherichia coli HVH 22 (4-2258986) Taxon ID: 1280973 | 534623483 | EQN80286.1 (Genbank) | URP |
| Escherichia coli HVH 220 (4-5876842) Taxon ID: 1281146 | 535370459 | EQV12581.1 (Genbank) | URP |
| Escherichia coli HVH 221 (4-3136817) Taxon ID: 1281147 | 535373358 | EQV15400.1 (Genbank) | URP |
| Escherichia coli HVH 222 (4-2977443) Taxon ID: 1281148 | 535375068 | EQV17070.1 (Genbank) | URP |
| Escherichia coli HVH 223 (4-2976528) Taxon ID: 1281149 | 535383493 | EQV25331.1 (Genbank) | URP |
| Escherichia coli HVH 225 (4-1273116) Taxon ID: 1281150 | 535395537 | EQV37142.1 (Genbank) | URP |
| Escherichia coli HVH 227 (4-2277670) Taxon ID: 1281151 | 535388467 | EQV30235.1 (Genbank) | URP |
| Escherichia coli HVH 228 (4-7787030) Taxon ID: 1281152 | 535996879 | ERA96490.1 (Genbank) | URP |
| Escherichia coli HVH 23 (4-6066488) Taxon ID: 1280974 | 565518330 | ETF30179.1 (Genbank) | URP |
| Escherichia coli HVH 24 (4-5985145) Taxon ID: 1280975 | 534629942 | EQN86631.1 (Genbank) | URP |
| Escherichia coli HVH 25 (4-5851939) Taxon ID: 1280976 | 534645631 | EQO02027.1 (Genbank) | URP |
| Escherichia coli HVH 26 (4-5703913) Taxon ID: 1280977 | 534642434 | EQN98915.1 (Genbank) | URP |
| Escherichia coli HVH 27 (4-7449267) Taxon ID: 1280978 | 534642771 | EQN99251.1 (Genbank) | URP |
| Escherichia coli HVH 28 (4-0907367) Taxon ID: 1280979 | 534653544 | EQO09861.1 (Genbank) | URP |
| Escherichia coli HVH 29 (4-3418073) Taxon ID: 1280980 | 534652061 | EQO08401.1 (Genbank) | URP |
| Escherichia coli HVH 3 (4-7276001) Taxon ID: 1280958 | 534550204 | EQN09811.1 (Genbank) | URP |
| Escherichia coli HVH 30 (4-2661829) Taxon ID: 1280981 | 534668048 | EQO24177.1 (Genbank) | URP |
| Escherichia coli HVH 31 (4-2602156) Taxon ID: 1280982 | 534666312 | EQO22453.1 (Genbank) | URP |
| Escherichia coli HVH 32 (4-3773988) Taxon ID: 1280983 | 534670849 | EQO26912.1 (Genbank) | URP |
| Escherichia coli HVH 33 (4-2174936) Taxon ID: 1280984 | 534679068 | EQO35034.1 (Genbank) | URP |
| Escherichia coli HVH 35 (4-2962667) Taxon ID: 1280985 | 534678737 | EQO34706.1 (Genbank) | URP |
| Escherichia coli HVH 36 (4-5675286) Taxon ID: 1280986 | 556121726 | ESP10322.1 (Genbank) | URP |
| Escherichia coli HVH 37 (4-2773848) Taxon ID: 1280987 | 534689145 | EQO44943.1 (Genbank) | URP |
| Escherichia coli HVH 38 (4-2774682) Taxon ID: 1280988 | 534698085 | EQO53746.1 (Genbank) | URP |
| Escherichia coli HVH 39 (4-2679949) Taxon ID: 1280989 | 534692504 | EQO48252.1 (Genbank) | URP |
| Escherichia coli HVH 4 (4-7276109) Taxon ID: 1280959 | 534564590 | EQN23992.1 (Genbank) | URP |
| Escherichia coli HVH 40 (4-1219782) Taxon ID: 1280990 | 534699117 | EQO54756.1 (Genbank) | URP |
| Escherichia coli HVH 41 (4-2677849) Taxon ID: 1280991 | 534719471 | EQO72905.1 (Genbank) | URP |
| Escherichia coli HVH 42 (4-2100061) Taxon ID: 1280992 | 534710681 | EQO64312.1 (Genbank) | URP |
| Escherichia coli HVH 43 (4-2173468) Taxon ID: 1280993 | 534708400 | EQO62056.1 (Genbank) | URP |
| Escherichia coli HVH 44 (4-2298570) Taxon ID: 1280994 | 534720654 | EQO74073.1 (Genbank) | URP |
| Escherichia coli HVH 45 (4-3129918) Taxon ID: 1280995 | 534728283 | EQO81563.1 (Genbank) | URP |
| Escherichia coli HVH 46 (4-2758776) Taxon ID: 1280996 | 534738350 | EQO91487.1 (Genbank) | URP |
| Escherichia coli HVH 48 (4-2658593) Taxon ID: 1280997 | 534733797 | EQO87016.1 (Genbank) | URP |
| Escherichia coli HVH 5 (4-7148410) Taxon ID: 1280960 | 534562747 | EQN22256.1 (Genbank) | URP |
| Escherichia coli HVH 51 (4-2172526) Taxon ID: 1280999 | 534745575 | EQO98593.1 (Genbank) | URP |
| Escherichia coli HVH 53 (4-0631051) Taxon ID: 1281000 | 534744672 | EQO97699.1 (Genbank) | URP |
| Escherichia coli HVH 55 (4-2646161) Taxon ID: 1281002 | 534749746 | EQP02675.1 (Genbank) | URP |
| Escherichia coli HVH 56 (4-2153033) Taxon ID: 1281003 | 534759056 | EQP11870.1 (Genbank) | URP |
| Escherichia coli HVH 58 (4-2839709) Taxon ID: 1281004 | 534761927 | EQP14699.1 (Genbank) | URP |
| Escherichia coli HVH 59 (4-1119338) Taxon ID: 1281005 | 534775824 | EQP28414.1 (Genbank) | URP |
| Escherichia coli HVH 6 (3-8296502) Taxon ID: 1280961 | 534573961 | EQN32826.1 (Genbank) | URP |
| Escherichia coli HVH 61 (4-2736020) Taxon ID: 1281006 | 534770715 | EQP23383.1 (Genbank) | URP |
| Escherichia coli HVH 63 (4-2542528) Taxon ID: 1281007 | 534773590 | EQP26207.1 (Genbank) | URP |
| Escherichia coli HVH 65 (4-2262045) Taxon ID: 1281008 | 534790382 | EQP42808.1 (Genbank) | URP |
| Escherichia coli HVH 68 (4-0888028) Taxon ID: 1281009 | 534789272 | EQP41727.1 (Genbank) | URP |
| Escherichia coli HVH 69 (4-2837072) Taxon ID: 1281010 | 534784712 | EQP37214.1 (Genbank) | URP |
| Escherichia coli HVH 7 (4-7315031) Taxon ID: 1280962 | 534579882 | EQN38474.1 (Genbank) | URP |
| Escherichia coli HVH 70 (4-2963531) Taxon ID: 1281011 | 534807562 | EQP59466.1 (Genbank) | URP |
| Escherichia coli HVH 74 (4-1034782) Taxon ID: 1281013 | 534804236 | EQP56288.1 (Genbank) | URP |
| Escherichia coli HVH 76 (4-2538717) Taxon ID: 1281014 | 534811455 | EQP63203.1 (Genbank) | URP |
| Escherichia coli HVH 77 (4-2605759) Taxon ID: 1281015 | 534825840 | EQP77191.1 (Genbank) | URP |
| Escherichia coli HVH 78 (4-2735946) Taxon ID: 1281016 | 534821163 | EQP72616.1 (Genbank) | URP |
| Escherichia coli HVH 79 (4-2512823) Taxon ID: 1281017 | 534827043 | EQP78302.1 (Genbank) | URP |
| Escherichia coli HVH 80 (4-2428830) Taxon ID: 1281018 | 534829722 | EQP80894.1 (Genbank) | URP |
| Escherichia coli HVH 82 (4-2209276) Taxon ID: 1281019 | 534834450 | EQP85504.1 (Genbank) | URP |
| Escherichia coli HVH 83 (4-2051087) Taxon ID: 1281020 | 565510244 | ETF22127.1 (Genbank) | URP |
| Escherichia coli HVH 84 (4-1021478) Taxon ID: 1281021 | 534846042 | EQP96877.1 (Genbank) | URP |
| Escherichia coli HVH 85 (4-0792144) Taxon ID: 1281022 | 534844246 | EQP95170.1 (Genbank) | URP |
| Escherichia coli HVH 86 (4-7026218) Taxon ID: 1281023 | 556137253 | ESP25663.1 (Genbank) | URP |
| Escherichia coli HVH 87 (4-5977630) Taxon ID: 1281024 | 534864964 | EQQ15495.1 (Genbank) | URP |
| Escherichia coli HVH 88 (4-5854636) Taxon ID: 1281025 | 534856612 | EQQ07256.1 (Genbank) | URP |
| Escherichia coli HVH 89 (4-5885604) Taxon ID: 1281026 | 534860536 | EQQ11136.1 (Genbank) | URP |
| Escherichia coli HVH 9 (4-6942539) Taxon ID: 1280963 | 534575782 | EQN34608.1 (Genbank) | URP |
| Escherichia coli HVH 90 (4-3191362) Taxon ID: 1281027 | 534868011 | EQQ18458.1 (Genbank) | URP |
| Escherichia coli HVH 91 (4-4638751) Taxon ID: 1281028 | 534879267 | EQQ29523.1 (Genbank) | URP |
| Escherichia coli HVH 92 (4-5930790) Taxon ID: 1281029 | 534881248 | EQQ31408.1 (Genbank) | URP |
| Escherichia coli HVH 95 (4-6074464) Taxon ID: 1281031 | 534879167 | EQQ29425.1 (Genbank) | URP |
| Escherichia coli HVH 96 (4-5934869) Taxon ID: 1281032 | 534900263 | EQQ49982.1 (Genbank) | URP |
| Escherichia coli HVH 98 (4-5799287) Taxon ID: 1281034 | 554704272 | ESK09758.1 (Genbank) | URP |
| Escherichia coli IHE3034 Taxon ID: 714962 | 294492652 | ADE91408.1 (Genbank) | URP |
| Escherichia coli J96 Taxon ID: 1206108 | 432348033 | ELL42485.1 (Genbank) | URP |
| Escherichia coli JB1-95 Taxon ID: 869682 | 386196437 | EIH90659.1 (Genbank) | URP |
| Escherichia coli JJ1886 Taxon ID: 1355100 | 554511187 | AGY83547.1 (Genbank) | URP |
| Escherichia coli Jurua 18/11 Taxon ID: 1115999 | 476348337 | EMX65682.1 (Genbank) | URP |
| Escherichia coli Jurua 20/10 Taxon ID: 1116000 | 476338718 | EMX56151.1 (Genbank) | URP |
| Escherichia coli K-12 Taxon ID: 83333 | 731473595 | AIZ54477.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808699406 | AKD60207.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808703777 | AKD64577.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808708155 | AKD68954.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808712522 | AKD73320.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808716932 | AKD77729.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808721302 | AKD82098.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808729623 | AKD86459.1 (Genbank) | PRP URP |
| Escherichia coli K-12 Taxon ID: 83333 | 808734034 | AKD90869.1 (Genbank) | PRP URP |
| Escherichia coli K02 Taxon ID: 1409786 | 601469291 | EYT11431.1 (Genbank) | URP |
| Escherichia coli KD1 Taxon ID: 752783 | 388384108 | EIL45850.1 (Genbank) | URP |
| Escherichia coli KD2 Taxon ID: 752784 | 388394718 | EIL55975.1 (Genbank) | URP |
| Escherichia coli KLY Taxon ID: 1435461 | 664683047 | AIF35998.1 (Genbank) | URP |
| Escherichia coli KO11 Taxon ID: 595495 | 323379553 | ADX51821.1 (Genbank) | URP |
| Escherichia coli KOEGE 10 (25a) Taxon ID: 1281155 | 536012066 | ERB11327.1 (Genbank) | URP |
| Escherichia coli KOEGE 118 (317a) Taxon ID: 1281171 | 535460243 | EQW00089.1 (Genbank) | URP |
| Escherichia coli KOEGE 131 (358a) Taxon ID: 1281172 | 535475268 | EQW15164.1 (Genbank) | URP |
| Escherichia coli KOEGE 3 (4a) Taxon ID: 1281153 | 536005560 | ERB04964.1 (Genbank) | URP |
| Escherichia coli KOEGE 30 (63a) Taxon ID: 1281156 | 535393083 | EQV34744.1 (Genbank) | URP |
| Escherichia coli KOEGE 32 (66a) Taxon ID: 1281157 | 535397144 | EQV38696.1 (Genbank) | URP |
| Escherichia coli KOEGE 40 (102a) Taxon ID: 1281159 | 535404536 | EQV45920.1 (Genbank) | URP |
| Escherichia coli KOEGE 43 (105a) Taxon ID: 1281160 | 535417536 | EQV58616.1 (Genbank) | URP |
| Escherichia coli KOEGE 44 (106a) Taxon ID: 1281161 | 535419419 | EQV60346.1 (Genbank) | URP |
| Escherichia coli KOEGE 56 (169a) Taxon ID: 1281162 | 535433130 | EQV73762.1 (Genbank) | URP |
| Escherichia coli KOEGE 58 (171a) Taxon ID: 1281163 | 535432825 | EQV73460.1 (Genbank) | URP |
| Escherichia coli KOEGE 61 (174a) Taxon ID: 1281164 | 535430855 | EQV71529.1 (Genbank) | URP |
| Escherichia coli KOEGE 62 (175a) Taxon ID: 1281165 | 535445280 | EQV85582.1 (Genbank) | URP |
| Escherichia coli KOEGE 68 (182a) Taxon ID: 1281166 | 535441820 | EQV82239.1 (Genbank) | URP |
| Escherichia coli KOEGE 7 (16a) Taxon ID: 1281154 | 536010230 | ERB09568.1 (Genbank) | URP |
| Escherichia coli KOEGE 70 (185a) Taxon ID: 1281167 | 535450590 | EQV90826.1 (Genbank) | URP |
| Escherichia coli KOEGE 73 (195a) Taxon ID: 1281169 | 535452183 | EQV92384.1 (Genbank) | URP |
| Escherichia coli KOEGE 77 (202a) Taxon ID: 1281170 | 535467219 | EQW07329.1 (Genbank) | URP |
| Escherichia coli KTE1 Taxon ID: 1169315 | 508527676 | EOW96227.1 (Genbank) | URP |
| Escherichia coli KTE100 Taxon ID: 1182699 | 508440005 | EOW10034.1 (Genbank) | URP |
| Escherichia coli KTE101 Taxon ID: 1182700 | 431357072 | ELG43746.1 (Genbank) | URP |
| Escherichia coli KTE103 Taxon ID: 1182702 | 508448194 | EOW18076.1 (Genbank) | URP |
| Escherichia coli KTE104 Taxon ID: 1169372 | 431535098 | ELI11481.1 (Genbank) | URP |
| Escherichia coli KTE105 Taxon ID: 1169373 | 431536725 | ELI12882.1 (Genbank) | URP |
| Escherichia coli KTE106 Taxon ID: 1169374 | 431540134 | ELI15763.1 (Genbank) | URP |
| Escherichia coli KTE107 Taxon ID: 1182703 | 508457357 | EOW27062.1 (Genbank) | URP |
| Escherichia coli KTE108 Taxon ID: 1182704 | 508452980 | EOW22754.1 (Genbank) | URP |
| Escherichia coli KTE109 Taxon ID: 1169375 | 431545706 | ELI20353.1 (Genbank) | URP |
| Escherichia coli KTE111 Taxon ID: 1182706 | 431203229 | ELF01905.1 (Genbank) | URP |
| Escherichia coli KTE112 Taxon ID: 1169376 | 431559144 | ELI32716.1 (Genbank) | URP |
| Escherichia coli KTE113 Taxon ID: 1169377 | 431555123 | ELI28981.1 (Genbank) | URP |
| Escherichia coli KTE115 Taxon ID: 1182707 | 431367680 | ELG54155.1 (Genbank) | URP |
| Escherichia coli KTE116 Taxon ID: 1182708 | 431203890 | ELF02479.1 (Genbank) | URP |
| Escherichia coli KTE117 Taxon ID: 1169378 | 431559760 | ELI33298.1 (Genbank) | URP |
| Escherichia coli KTE118 Taxon ID: 1182709 | 431370916 | ELG56709.1 (Genbank) | URP |
| Escherichia coli KTE119 Taxon ID: 1182710 | 431213276 | ELF11152.1 (Genbank) | URP |
| Escherichia coli KTE12 Taxon ID: 1169332 | 430901121 | ELC23104.1 (Genbank) | URP |
| Escherichia coli KTE120 Taxon ID: 1169379 | 431571897 | ELI44766.1 (Genbank) | URP |
| Escherichia coli KTE121 Taxon ID: 1182711 | 508454203 | EOW23950.1 (Genbank) | URP |
| Escherichia coli KTE122 Taxon ID: 1169380 | 431575148 | ELI47895.1 (Genbank) | URP |
| Escherichia coli KTE123 Taxon ID: 1182712 | 431374324 | ELG59916.1 (Genbank) | URP |
| Escherichia coli KTE124 Taxon ID: 1169381 | 431573935 | ELI46724.1 (Genbank) | URP |
| Escherichia coli KTE125 Taxon ID: 1169382 | 431587549 | ELI58922.1 (Genbank) | URP |
| Escherichia coli KTE126 Taxon ID: 1182713 | 508469549 | EOW39069.1 (Genbank) | URP |
| Escherichia coli KTE127 Taxon ID: 1182714 | 508470508 | EOW40017.1 (Genbank) | URP |
| Escherichia coli KTE128 Taxon ID: 1169383 | 431590243 | ELI61339.1 (Genbank) | URP |
| Escherichia coli KTE129 Taxon ID: 1169384 | 431592794 | ELI63363.1 (Genbank) | URP |
| Escherichia coli KTE13 Taxon ID: 1169318 | 508263241 | EOU36540.1 (Genbank) | URP |
| Escherichia coli KTE130 Taxon ID: 1182715 | 508480919 | EOW50217.1 (Genbank) | URP |
| Escherichia coli KTE131 Taxon ID: 1169385 | 431601366 | ELI70883.1 (Genbank) | URP |
| Escherichia coli KTE132 Taxon ID: 1182716 | 508481403 | EOW50691.1 (Genbank) | URP |
| Escherichia coli KTE133 Taxon ID: 1169386 | 431605374 | ELI74763.1 (Genbank) | URP |
| Escherichia coli KTE134 Taxon ID: 1182717 | 508483725 | EOW52987.1 (Genbank) | URP |
| Escherichia coli KTE135 Taxon ID: 1182718 | 431379843 | ELG64753.1 (Genbank) | URP |
| Escherichia coli KTE136 Taxon ID: 1182719 | 431388159 | ELG71897.1 (Genbank) | URP |
| Escherichia coli KTE137 Taxon ID: 1169387 | 431609297 | ELI78622.1 (Genbank) | URP |
| Escherichia coli KTE138 Taxon ID: 1169388 | 431614355 | ELI83510.1 (Genbank) | URP |
| Escherichia coli KTE139 Taxon ID: 1169389 | 431619589 | ELI88509.1 (Genbank) | URP |
| Escherichia coli KTE14 Taxon ID: 1169333 | 508297738 | EOU70262.1 (Genbank) | URP |
| Escherichia coli KTE140 Taxon ID: 1182720 | 431391455 | ELG75095.1 (Genbank) | URP |
| Escherichia coli KTE141 Taxon ID: 1182721 | 431397403 | ELG80859.1 (Genbank) | URP |
| Escherichia coli KTE142 Taxon ID: 1182722 | 431217936 | ELF15422.1 (Genbank) | URP |
| Escherichia coli KTE143 Taxon ID: 1182723 | 431224597 | ELF21813.1 (Genbank) | URP |
| Escherichia coli KTE144 Taxon ID: 1182724 | 431401694 | ELG85028.1 (Genbank) | URP |
| Escherichia coli KTE145 Taxon ID: 1169390 | 431622288 | ELI91069.1 (Genbank) | URP |
| Escherichia coli KTE147 Taxon ID: 1182726 | 431405364 | ELG88607.1 (Genbank) | URP |
| Escherichia coli KTE148 Taxon ID: 1169391 | 431634274 | ELJ02525.1 (Genbank) | URP |
| Escherichia coli KTE15 Taxon ID: 1169334 | 430911539 | ELC32822.1 (Genbank) | URP |
| Escherichia coli KTE150 Taxon ID: 1169392 | 431631127 | ELI99446.1 (Genbank) | URP |
| Escherichia coli KTE153 Taxon ID: 1169393 | 431637039 | ELJ05158.1 (Genbank) | URP |
| Escherichia coli KTE154 Taxon ID: 1182728 | 431413721 | ELG96486.1 (Genbank) | URP |
| Escherichia coli KTE155 Taxon ID: 1182729 | 508493014 | EOW62168.1 (Genbank) | URP |
| Escherichia coli KTE156 Taxon ID: 1182730 | 431224859 | ELF22069.1 (Genbank) | URP |
| Escherichia coli KTE157 Taxon ID: 1169394 | 431648747 | ELJ16121.1 (Genbank) | URP |
| Escherichia coli KTE158 Taxon ID: 1182731 | 431419388 | ELH01738.1 (Genbank) | URP |
| Escherichia coli KTE16 Taxon ID: 1169335 | 430909556 | ELC30922.1 (Genbank) | URP |
| Escherichia coli KTE160 Taxon ID: 1169395 | 431650259 | ELJ17596.1 (Genbank) | URP |
| Escherichia coli KTE161 Taxon ID: 1182733 | 431231017 | ELF26787.1 (Genbank) | URP |
| Escherichia coli KTE162 Taxon ID: 1182734 | 431236629 | ELF31834.1 (Genbank) | URP |
| Escherichia coli KTE163 Taxon ID: 1169396 | 431651345 | ELJ18606.1 (Genbank) | URP |
| Escherichia coli KTE165 Taxon ID: 1182735 | 431424352 | ELH06448.1 (Genbank) | URP |
| Escherichia coli KTE166 Taxon ID: 1169397 | 431662405 | ELJ29180.1 (Genbank) | URP |
| Escherichia coli KTE167 Taxon ID: 1169398 | 431665186 | ELJ31913.1 (Genbank) | URP |
| Escherichia coli KTE168 Taxon ID: 1169399 | 431665803 | ELJ32513.1 (Genbank) | URP |
| Escherichia coli KTE169 Taxon ID: 1182736 | 431246672 | ELF40930.1 (Genbank) | URP |
| Escherichia coli KTE17 Taxon ID: 1169336 | 431268184 | ELF59665.1 (Genbank) | URP |
| Escherichia coli KTE170 Taxon ID: 1182737 | 508502022 | EOW71060.1 (Genbank) | URP |
| Escherichia coli KTE171 Taxon ID: 1182738 | 431246834 | ELF41082.1 (Genbank) | URP |
| Escherichia coli KTE173 Taxon ID: 1181724 | 431446658 | ELH27402.1 (Genbank) | URP |
| Escherichia coli KTE174 Taxon ID: 1169400 | 431676938 | ELJ43021.1 (Genbank) | URP |
| Escherichia coli KTE175 Taxon ID: 1181725 | 431448562 | ELH29277.1 (Genbank) | URP |
| Escherichia coli KTE176 Taxon ID: 1169401 | 431678325 | ELJ44329.1 (Genbank) | URP |
| Escherichia coli KTE177 Taxon ID: 1169402 | 431681362 | ELJ47151.1 (Genbank) | URP |
| Escherichia coli KTE178 Taxon ID: 1181726 | 430947188 | ELC66895.1 (Genbank) | URP |
| Escherichia coli KTE179 Taxon ID: 1169403 | 431691800 | ELJ57251.1 (Genbank) | URP |
| Escherichia coli KTE18 Taxon ID: 1169337 | 431275413 | ELF66442.1 (Genbank) | URP |
| Escherichia coli KTE180 Taxon ID: 1169404 | 431693456 | ELJ58869.1 (Genbank) | URP |
| Escherichia coli KTE181 Taxon ID: 1181727 | 430958743 | ELC77320.1 (Genbank) | URP |
| Escherichia coli KTE182 Taxon ID: 1181728 | 508533279 | EOX01748.1 (Genbank) | URP |
| Escherichia coli KTE183 Taxon ID: 1181729 | 431453655 | ELH34039.1 (Genbank) | URP |
| Escherichia coli KTE184 Taxon ID: 1181730 | 431457006 | ELH37349.1 (Genbank) | URP |
| Escherichia coli KTE185 Taxon ID: 1181731 | 508557611 | EOX24874.1 (Genbank) | URP |
| Escherichia coli KTE186 Taxon ID: 1181732 | 508558712 | EOX25967.1 (Genbank) | URP |
| Escherichia coli KTE187 Taxon ID: 1181733 | 430956326 | ELC75002.1 (Genbank) | URP |
| Escherichia coli KTE188 Taxon ID: 1181734 | 430966811 | ELC84174.1 (Genbank) | URP |
| Escherichia coli KTE189 Taxon ID: 1181735 | 430969206 | ELC86356.1 (Genbank) | URP |
| Escherichia coli KTE19 Taxon ID: 1169338 | 508283908 | EOU56910.1 (Genbank) | URP |
| Escherichia coli KTE190 Taxon ID: 1181736 | 431444263 | ELH25286.1 (Genbank) | URP |
| Escherichia coli KTE191 Taxon ID: 1181737 | 430975809 | ELC92693.1 (Genbank) | URP |
| Escherichia coli KTE192 Taxon ID: 1181738 | 431429512 | ELH11440.1 (Genbank) | URP |
| Escherichia coli KTE193 Taxon ID: 1181739 | 430974029 | ELC90967.1 (Genbank) | URP |
| Escherichia coli KTE194 Taxon ID: 1181740 | 431436518 | ELH18033.1 (Genbank) | URP |
| Escherichia coli KTE195 Taxon ID: 1181741 | 508339639 | EOV11379.1 (Genbank) | URP |
| Escherichia coli KTE196 Taxon ID: 1181742 | 431462477 | ELH42688.1 (Genbank) | URP |
| Escherichia coli KTE197 Taxon ID: 1181743 | 431470259 | ELH50182.1 (Genbank) | URP |
| Escherichia coli KTE198 Taxon ID: 1181744 | 508357443 | EOV28890.1 (Genbank) | URP |
| Escherichia coli KTE199 Taxon ID: 1181745 | 508345419 | EOV17035.1 (Genbank) | URP |
| Escherichia coli KTE2 Taxon ID: 1169324 | 430878741 | ELC02127.1 (Genbank) | URP |
| Escherichia coli KTE20 Taxon ID: 1169339 | 508300037 | EOU72529.1 (Genbank) | URP |
| Escherichia coli KTE200 Taxon ID: 1181746 | 508354207 | EOV25712.1 (Genbank) | URP |
| Escherichia coli KTE201 Taxon ID: 1181747 | 430985389 | ELD01995.1 (Genbank) | URP |
| Escherichia coli KTE202 Taxon ID: 1181748 | 431478236 | ELH57985.1 (Genbank) | URP |
| Escherichia coli KTE203 Taxon ID: 1181749 | 431473934 | ELH53757.1 (Genbank) | URP |
| Escherichia coli KTE204 Taxon ID: 1181750 | 430991608 | ELD08011.1 (Genbank) | URP |
| Escherichia coli KTE205 Taxon ID: 1181751 | 430997367 | ELD13634.1 (Genbank) | URP |
| Escherichia coli KTE206 Taxon ID: 1181752 | 431000092 | ELD16166.1 (Genbank) | URP |
| Escherichia coli KTE207 Taxon ID: 1181753 | 431485906 | ELH65563.1 (Genbank) | URP |
| Escherichia coli KTE208 Taxon ID: 1181754 | 431009099 | ELD23723.1 (Genbank) | URP |
| Escherichia coli KTE209 Taxon ID: 1181755 | 431482416 | ELH62119.1 (Genbank) | URP |
| Escherichia coli KTE21 Taxon ID: 1169340 | 430922243 | ELC43003.1 (Genbank) | URP |
| Escherichia coli KTE210 Taxon ID: 1181756 | 431009869 | ELD24473.1 (Genbank) | URP |
| Escherichia coli KTE211 Taxon ID: 1181757 | 431494105 | ELH73695.1 (Genbank) | URP |
| Escherichia coli KTE212 Taxon ID: 1181758 | 431018136 | ELD31573.1 (Genbank) | URP |
| Escherichia coli KTE213 Taxon ID: 1181759 | 431024016 | ELD37209.1 (Genbank) | URP |
| Escherichia coli KTE214 Taxon ID: 1181760 | 431027973 | ELD41018.1 (Genbank) | URP |
| Escherichia coli KTE215 Taxon ID: 1181761 | 431506028 | ELH84632.1 (Genbank) | URP |
| Escherichia coli KTE216 Taxon ID: 1181762 | 431032000 | ELD44724.1 (Genbank) | URP |
| Escherichia coli KTE217 Taxon ID: 1181763 | 431498154 | ELH77368.1 (Genbank) | URP |
| Escherichia coli KTE218 Taxon ID: 1181764 | 431509717 | ELH87966.1 (Genbank) | URP |
| Escherichia coli KTE219 Taxon ID: 1181765 | 508369477 | EOV40794.1 (Genbank) | URP |
| Escherichia coli KTE22 Taxon ID: 1169341 | 431305773 | ELF94094.1 (Genbank) | URP |
| Escherichia coli KTE220 Taxon ID: 1181766 | 431042298 | ELD52790.1 (Genbank) | URP |
| Escherichia coli KTE221 Taxon ID: 1181767 | 508372862 | EOV44133.1 (Genbank) | URP |
| Escherichia coli KTE222 Taxon ID: 1181768 | 508367369 | EOV38705.1 (Genbank) | URP |
| Escherichia coli KTE223 Taxon ID: 1181769 | 431514371 | ELH92213.1 (Genbank) | URP |
| Escherichia coli KTE224 Taxon ID: 1181770 | 431044409 | ELD54682.1 (Genbank) | URP |
| Escherichia coli KTE225 Taxon ID: 1181771 | 508541658 | EOX09997.1 (Genbank) | URP |
| Escherichia coli KTE226 Taxon ID: 1181772 | 508544486 | EOX12796.1 (Genbank) | URP |
| Escherichia coli KTE227 Taxon ID: 1181773 | 431517443 | ELH94965.1 (Genbank) | URP |
| Escherichia coli KTE228 Taxon ID: 1181774 | 431044838 | ELD55095.1 (Genbank) | URP |
| Escherichia coli KTE229 Taxon ID: 1181775 | 431519543 | ELH96995.1 (Genbank) | URP |
| Escherichia coli KTE23 Taxon ID: 1169342 | 431286519 | ELF77345.1 (Genbank) | URP |
| Escherichia coli KTE230 Taxon ID: 1181776 | 431054861 | ELD64430.1 (Genbank) | URP |
| Escherichia coli KTE231 Taxon ID: 1169323 | 508280102 | EOU53164.1 (Genbank) | URP |
| Escherichia coli KTE232 Taxon ID: 1169365 | 431695970 | ELJ61168.1 (Genbank) | URP |
| Escherichia coli KTE233 Taxon ID: 1181777 | 431057042 | ELD66520.1 (Genbank) | URP |
| Escherichia coli KTE234 Taxon ID: 1181778 | 431063728 | ELD72945.1 (Genbank) | URP |
| Escherichia coli KTE235 Taxon ID: 1181779 | 431072960 | ELD80697.1 (Genbank) | URP |
| Escherichia coli KTE236 Taxon ID: 1181780 | 431077258 | ELD84519.1 (Genbank) | URP |
| Escherichia coli KTE237 Taxon ID: 1181781 | 431084842 | ELD90966.1 (Genbank) | URP |
| Escherichia coli KTE24 Taxon ID: 1169343 | 508302368 | EOU74837.1 (Genbank) | URP |
| Escherichia coli KTE240 Taxon ID: 1181782 | 508548168 | EOX16423.1 (Genbank) | URP |
| Escherichia coli KTE25 Taxon ID: 1169344 | 430917668 | ELC38708.1 (Genbank) | URP |
| Escherichia coli KTE26 Taxon ID: 1169345 | 430928548 | ELC49096.1 (Genbank) | URP |
| Escherichia coli KTE27 Taxon ID: 1169346 | 508309516 | EOU81797.1 (Genbank) | URP |
| Escherichia coli KTE28 Taxon ID: 1169347 | 430932380 | ELC52803.1 (Genbank) | URP |
| Escherichia coli KTE29 Taxon ID: 1169348 | 431300091 | ELF89654.1 (Genbank) | URP |
| Escherichia coli KTE3 Taxon ID: 1169316 | 508267451 | EOU40691.1 (Genbank) | URP |
| Escherichia coli KTE34 Taxon ID: 1169352 | 508327618 | EOU99600.1 (Genbank) | URP |
| Escherichia coli KTE35 Taxon ID: 1169320 | 508282200 | EOU55224.1 (Genbank) | URP |
| Escherichia coli KTE36 Taxon ID: 1169353 | 508323095 | EOU95157.1 (Genbank) | URP |
| Escherichia coli KTE37 Taxon ID: 1169354 | 508325807 | EOU97835.1 (Genbank) | URP |
| Escherichia coli KTE38 Taxon ID: 1169355 | 508341655 | EOV13366.1 (Genbank) | URP |
| Escherichia coli KTE39 Taxon ID: 1169356 | 430938592 | ELC58833.1 (Genbank) | URP |
| Escherichia coli KTE4 Taxon ID: 1169325 | 430879751 | ELC03082.1 (Genbank) | URP |
| Escherichia coli KTE40 Taxon ID: 1169357 | 508343691 | EOV15349.1 (Genbank) | URP |
| Escherichia coli KTE41 Taxon ID: 1169358 | 508532094 | EOX00585.1 (Genbank) | URP |
| Escherichia coli KTE42 Taxon ID: 1169359 | 431286737 | ELF77561.1 (Genbank) | URP |
| Escherichia coli KTE43 Taxon ID: 1169360 | 431295055 | ELF85226.1 (Genbank) | URP |
| Escherichia coli KTE44 Taxon ID: 1169361 | 430943932 | ELC64038.1 (Genbank) | URP |
| Escherichia coli KTE45 Taxon ID: 1169362 | 431278227 | ELF69221.1 (Genbank) | URP |
| Escherichia coli KTE46 Taxon ID: 1182653 | 431311607 | ELF99765.1 (Genbank) | URP |
| Escherichia coli KTE47 Taxon ID: 1182654 | 431086888 | ELD92904.1 (Genbank) | URP |
| Escherichia coli KTE48 Taxon ID: 1182655 | 431313074 | ELG01053.1 (Genbank) | URP |
| Escherichia coli KTE49 Taxon ID: 1182656 | 431094057 | ELD99707.1 (Genbank) | URP |
| Escherichia coli KTE5 Taxon ID: 1169326 | 430889931 | ELC12578.1 (Genbank) | URP |
| Escherichia coli KTE50 Taxon ID: 1182657 | 431318165 | ELG05932.1 (Genbank) | URP |
| Escherichia coli KTE51 Taxon ID: 1182658 | 431099744 | ELE04764.1 (Genbank) | URP |
| Escherichia coli KTE53 Taxon ID: 1182660 | 431103260 | ELE07930.1 (Genbank) | URP |
| Escherichia coli KTE54 Taxon ID: 1182661 | 431320365 | ELG08007.1 (Genbank) | URP |
| Escherichia coli KTE55 Taxon ID: 1182662 | 431110928 | ELE14845.1 (Genbank) | URP |
| Escherichia coli KTE56 Taxon ID: 1182663 | 431108991 | ELE12962.1 (Genbank) | URP |
| Escherichia coli KTE57 Taxon ID: 1182664 | 431119730 | ELE22729.1 (Genbank) | URP |
| Escherichia coli KTE58 Taxon ID: 1182665 | 431124174 | ELE26828.1 (Genbank) | URP |
| Escherichia coli KTE59 Taxon ID: 1182666 | 431330133 | ELG17415.1 (Genbank) | URP |
| Escherichia coli KTE6 Taxon ID: 1169327 | 431251607 | ELF45614.1 (Genbank) | URP |
| Escherichia coli KTE60 Taxon ID: 1182667 | 431133150 | ELE35148.1 (Genbank) | URP |
| Escherichia coli KTE61 Taxon ID: 1182668 | 508383927 | EOV54993.1 (Genbank) | URP |
| Escherichia coli KTE62 Taxon ID: 1182669 | 431133440 | ELE35430.1 (Genbank) | URP |
| Escherichia coli KTE63 Taxon ID: 1182670 | 431332207 | ELG19450.1 (Genbank) | URP |
| Escherichia coli KTE64 Taxon ID: 1182671 | 508394086 | EOV64972.1 (Genbank) | URP |
| Escherichia coli KTE65 Taxon ID: 1182672 | 431341451 | ELG28458.1 (Genbank) | URP |
| Escherichia coli KTE66 Taxon ID: 1182673 | 431143572 | ELE45295.1 (Genbank) | URP |
| Escherichia coli KTE67 Taxon ID: 1182674 | 431140694 | ELE42460.1 (Genbank) | URP |
| Escherichia coli KTE68 Taxon ID: 1182675 | 508395721 | EOV66564.1 (Genbank) | URP |
| Escherichia coli KTE69 Taxon ID: 1182676 | 508396660 | EOV67480.1 (Genbank) | URP |
| Escherichia coli KTE7 Taxon ID: 1169317 | 508264933 | EOU38215.1 (Genbank) | URP |
| Escherichia coli KTE70 Taxon ID: 1182677 | 508410002 | EOV80510.1 (Genbank) | URP |
| Escherichia coli KTE71 Taxon ID: 1182678 | 508412709 | EOV83153.1 (Genbank) | URP |
| Escherichia coli KTE72 Taxon ID: 1182679 | 431151145 | ELE52180.1 (Genbank) | URP |
| Escherichia coli KTE73 Taxon ID: 1182680 | 508411896 | EOV82366.1 (Genbank) | URP |
| Escherichia coli KTE74 Taxon ID: 1182681 | 508427123 | EOV97340.1 (Genbank) | URP |
| Escherichia coli KTE75 Taxon ID: 1182682 | 431157320 | ELE57964.1 (Genbank) | URP |
| Escherichia coli KTE76 Taxon ID: 1182683 | 431162408 | ELE62857.1 (Genbank) | URP |
| Escherichia coli KTE77 Taxon ID: 1182684 | 431165318 | ELE65676.1 (Genbank) | URP |
| Escherichia coli KTE78 Taxon ID: 1182685 | 431342189 | ELG29179.1 (Genbank) | URP |
| Escherichia coli KTE79 Taxon ID: 1182686 | 431345298 | ELG32220.1 (Genbank) | URP |
| Escherichia coli KTE8 Taxon ID: 1169328 | 431259828 | ELF52191.1 (Genbank) | URP |
| Escherichia coli KTE80 Taxon ID: 1182687 | 431173842 | ELE73912.1 (Genbank) | URP |
| Escherichia coli KTE81 Taxon ID: 1182688 | 431174195 | ELE74256.1 (Genbank) | URP |
| Escherichia coli KTE82 Taxon ID: 1169363 | 431709632 | ELJ74086.1 (Genbank) | URP |
| Escherichia coli KTE83 Taxon ID: 1182689 | 431184758 | ELE84504.1 (Genbank) | URP |
| Escherichia coli KTE85 Taxon ID: 1169364 | 431711279 | ELJ75632.1 (Genbank) | URP |
| Escherichia coli KTE86 Taxon ID: 1182691 | 431183863 | ELE83636.1 (Genbank) | URP |
| Escherichia coli KTE87 Taxon ID: 1182692 | 431193487 | ELE92823.1 (Genbank) | URP |
| Escherichia coli KTE88 Taxon ID: 1169366 | 431709075 | ELJ73572.1 (Genbank) | URP |
| Escherichia coli KTE89 Taxon ID: 1182693 | 508427189 | EOV97405.1 (Genbank) | URP |
| Escherichia coli KTE9 Taxon ID: 1169329 | 431266688 | ELF58228.1 (Genbank) | URP |
| Escherichia coli KTE90 Taxon ID: 1169367 | 431720839 | ELJ84858.1 (Genbank) | URP |
| Escherichia coli KTE91 Taxon ID: 1182694 | 431357180 | ELG43853.1 (Genbank) | URP |
| Escherichia coli KTE93 Taxon ID: 1182696 | 431194277 | ELE93542.1 (Genbank) | URP |
| Escherichia coli KTE94 Taxon ID: 1169368 | 431725407 | ELJ89261.1 (Genbank) | URP |
| Escherichia coli KTE95 Taxon ID: 1169369 | 431725666 | ELJ89506.1 (Genbank) | URP |
| Escherichia coli KTE97 Taxon ID: 1169370 | 431732863 | ELJ96311.1 (Genbank) | URP |
| Escherichia coli KTE98 Taxon ID: 1182698 | 508427364 | EOV97579.1 (Genbank) | URP |
| Escherichia coli KTE99 Taxon ID: 1169371 | 431736172 | ELJ99514.1 (Genbank) | URP |
| Escherichia coli LAU-EC10 Taxon ID: 1417796 | 564930561 | ETE25492.1 (Genbank) | URP |
| Escherichia coli LAU-EC6 Taxon ID: 1389417 | 564923498 | ETE18660.1 (Genbank) | URP |
| Escherichia coli LAU-EC7 Taxon ID: 1389418 | 564937459 | ETE32176.1 (Genbank) | URP |
| Escherichia coli LAU-EC8 Taxon ID: 1389419 | 564917813 | ETE13166.1 (Genbank) | URP |
| Escherichia coli LAU-EC9 Taxon ID: 1389420 | 564944947 | ETE39516.1 (Genbank) | URP |
| Escherichia coli LY180 Taxon ID: 1335916 | 544201346 | AGW08038.1 (Genbank) | URP |
| Escherichia coli M114 Taxon ID: 656416 | 902637304 | KNA41681.1 (Genbank) | URP |
| Escherichia coli M605 Taxon ID: 656417 | 331044537 | EGI16664.1 (Genbank) | URP |
| Escherichia coli M718 Taxon ID: 656419 | 331049914 | EGI21972.1 (Genbank) | URP |
| Escherichia coli M863 Taxon ID: 656420 | 323967575 | EGB62991.1 (Genbank) | URP |
| Escherichia coli M919 Taxon ID: 656421 | 385540086 | EIF86913.1 (Genbank) | URP |
| Escherichia coli MA6 Taxon ID: 1005557 | 408171959 | EKH99047.1 (Genbank) | URP |
| Escherichia coli MGH 57 Taxon ID: 1438687 | 635887014 | KDG67637.1 (Genbank) | URP |
| Escherichia coli MGH 58 Taxon ID: 1438688 | 635884935 | KDG65586.1 (Genbank) | URP |
| Escherichia coli MP020940.1 Taxon ID: 1116140 | 476342646 | EMX60037.1 (Genbank) | URP |
| Escherichia coli MP020980.2 Taxon ID: 1116139 | 476337774 | EMX55211.1 (Genbank) | URP |
| Escherichia coli MP021017.1 Taxon ID: 1116057 | 476324488 | EMX42048.1 (Genbank) | URP |
| Escherichia coli MP021017.12 Taxon ID: 1116111 | 476106039 | EMV29053.1 (Genbank) | URP |
| Escherichia coli MP021552.11 Taxon ID: 1116134 | 476041318 | EMU66660.1 (Genbank) | URP |
| Escherichia coli MP021552.8 Taxon ID: 1116133 | 476326028 | EMX43580.1 (Genbank) | URP |
| Escherichia coli MP021561.2 Taxon ID: 1116136 | 476314834 | EMX32515.1 (Genbank) | URP |
| Escherichia coli MP021561.3 Taxon ID: 1116137 | 476845928 | ENB42731.1 (Genbank) | URP |
| Escherichia coli MP021566.1 Taxon ID: 1116138 | 476308856 | EMX26651.1 (Genbank) | URP |
| Escherichia coli MP1 Taxon ID: 1314835 | 587648230 | EWY55193.1 (Genbank) | URP |
| Escherichia coli MRSN 10204 Taxon ID: 1409953 | 816076767 | KKJ24412.1 (Genbank) | URP |
| Escherichia coli MS 107-1 Taxon ID: 679207 | 300529552 | EFK50614.1 (Genbank) | URP |
| Escherichia coli MS 110-3 Taxon ID: 749536 | 315287080 | EFU46494.1 (Genbank) | URP |
| Escherichia coli MS 115-1 Taxon ID: 749537 | 300412447 | EFJ95757.1 (Genbank) | URP |
| Escherichia coli MS 117-3 Taxon ID: 749539 | 324016098 | EGB85317.1 (Genbank) | URP |
| Escherichia coli MS 119-7 Taxon ID: 679206 | 300524407 | EFK45476.1 (Genbank) | URP |
| Escherichia coli MS 124-1 Taxon ID: 679205 | 300842658 | EFK70418.1 (Genbank) | URP |
| Escherichia coli MS 145-7 Taxon ID: 679204 | 308120880 | EFO58142.1 (Genbank) | URP |
| Escherichia coli MS 146-1 Taxon ID: 749540 | 301077680 | EFK92486.1 (Genbank) | URP |
| Escherichia coli MS 153-1 Taxon ID: 749541 | 315292094 | EFU51446.1 (Genbank) | URP |
| Escherichia coli MS 16-3 Taxon ID: 749542 | 315299113 | EFU58367.1 (Genbank) | URP |
| Escherichia coli MS 182-1 Taxon ID: 749545 | 300416762 | EFK00073.1 (Genbank) | URP |
| Escherichia coli MS 185-1 Taxon ID: 749546 | 300298417 | EFJ54802.1 (Genbank) | URP |
| Escherichia coli MS 196-1 Taxon ID: 749548 | 299878130 | EFI86341.1 (Genbank) | URP |
| Escherichia coli MS 198-1 Taxon ID: 749549 | 300355417 | EFJ71287.1 (Genbank) | URP |
| Escherichia coli MS 200-1 Taxon ID: 749550 | 300304457 | EFJ58977.1 (Genbank) | URP |
| Escherichia coli MS 21-1 Taxon ID: 749527 | 300455515 | EFK19008.1 (Genbank) | URP |
| Escherichia coli MS 45-1 Taxon ID: 749528 | 300407157 | EFJ90695.1 (Genbank) | URP |
| Escherichia coli MS 57-2 Taxon ID: 749529 | 324006321 | EGB75540.1 (Genbank) | URP |
| Escherichia coli MS 60-1 Taxon ID: 749530 | 324010483 | EGB79702.1 (Genbank) | URP |
| Escherichia coli MS 69-1 Taxon ID: 749531 | 300396146 | EFJ79684.1 (Genbank) | URP |
| Escherichia coli MS 79-10 Taxon ID: 796392 | 342928759 | EGU97481.1 (Genbank) | URP |
| Escherichia coli MS 84-1 Taxon ID: 749533 | 300400491 | EFJ84029.1 (Genbank) | URP |
| Escherichia coli MS 85-1 Taxon ID: 679202 | 315255070 | EFU35038.1 (Genbank) | URP |
| Escherichia coli N1 Taxon ID: 1005567 | 408231669 | EKI54933.1 (Genbank) | URP |
| Escherichia coli NA114 Taxon ID: 1033813 | 333968584 | AEG35389.1 (Genbank) | URP |
| Escherichia coli NB8 Taxon ID: 1431454 | 816245552 | KKK03952.1 (Genbank) | URP |
| Escherichia coli NC101 Taxon ID: 753642 | 305853703 | EFM54142.1 (Genbank) | URP |
| Escherichia coli NCTC 50110 Taxon ID: 1333537 | 697406775 | KGL67899.1 (Genbank) | URP |
| Escherichia coli NE037 Taxon ID: 1005484 | 408130111 | EKH60281.1 (Genbank) | URP |
| Escherichia coli NE098 Taxon ID: 1005486 | 408336557 | EKJ51314.1 (Genbank) | URP |
| Escherichia coli NE1487 Taxon ID: 1005485 | 408121878 | EKH52771.1 (Genbank) | URP |
| Escherichia coli Nissle 1917 Taxon ID: 316435 | 660510791 | AID77682.1 (Genbank) | URP |
| Escherichia coli O08 Taxon ID: 1227814 | 449322400 | EMD12392.1 (Genbank) | URP |
| Escherichia coli O103:H11 str. 04-3023 Taxon ID: 1446501 | 607003492 | EZA32143.1 (Genbank) | URP |
| Escherichia coli O103:H11 str. 2010C-3214 Taxon ID: 1446502 | 606507771 | EYV63267.1 (Genbank) | URP |
| Escherichia coli O103:H2 str. 2009C-3279 Taxon ID: 1446503 | 607717918 | EZE00520.1 (Genbank) | URP |
| Escherichia coli O103:H2 str. 2010C-4433 Taxon ID: 1446504 | 606883797 | EYZ18741.1 (Genbank) | URP |
| Escherichia coli O103:H2 str. 2011C-3750 Taxon ID: 1446505 | 606746982 | EYX89491.1 (Genbank) | URP |
| Escherichia coli O103:H2 str. CVM9450 Taxon ID: 1165941 | 388343903 | EIL09802.1 (Genbank) | URP |
| Escherichia coli O104:H21 str. 94-3025 Taxon ID: 1446497 | 607371536 | EZA64432.1 (Genbank) | URP |
| Escherichia coli O104:H21 str. CFSAN002236 Taxon ID: 1335303 | 540145154 | ERF90602.1 (Genbank) | URP |
| Escherichia coli O104:H21 str. CFSAN002237 Taxon ID: 1335304 | 540151718 | ERF96926.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 01-09591 Taxon ID: 1042803 | 340735865 | EGR64921.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 04-8351 Taxon ID: 1048265 | 354858432 | EHF18883.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 09-7901 Taxon ID: 1048266 | 354877337 | EHF37697.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02030 Taxon ID: 1240768 | 429351716 | EKY88436.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02033-1 Taxon ID: 1240769 | 429351849 | EKY88568.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02092 Taxon ID: 1240770 | 429353177 | EKY89886.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02093 Taxon ID: 1240771 | 429367090 | EKZ03691.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02281 Taxon ID: 1240772 | 429368001 | EKZ04593.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02318 Taxon ID: 1240773 | 429370496 | EKZ07062.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-02913 Taxon ID: 1240774 | 429382883 | EKZ19347.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-03439 Taxon ID: 1240775 | 429385639 | EKZ22092.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-03943 Taxon ID: 1240777 | 429385116 | EKZ21570.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-04080 Taxon ID: 1240776 | 429397332 | EKZ33679.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-3677 Taxon ID: 1048334 | 354883545 | EHF43864.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4404 Taxon ID: 1068614 | 354879132 | EHF39473.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4522 Taxon ID: 1068615 | 354885530 | EHF45827.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4623 Taxon ID: 1068616 | 354888216 | EHF48477.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4632 C1 Taxon ID: 1068617 | 354901701 | EHF61828.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4632 C2 Taxon ID: 1068618 | 354904517 | EHF64608.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4632 C3 Taxon ID: 1068619 | 354907174 | EHF67239.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4632 C4 Taxon ID: 1068620 | 354921959 | EHF81878.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 11-4632 C5 Taxon ID: 1068621 | 354917984 | EHF77945.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 2009EL-2050 Taxon ID: 1134782 | 406778929 | AFS58353.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 2009EL-2071 Taxon ID: 1133853 | 407064090 | AFS85137.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 2011C-3493 Taxon ID: 1133852 | 407055511 | AFS75562.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. 2011EL-1675A Taxon ID: 1446507 | 606724319 | EYX68205.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. C227-11 Taxon ID: 1048254 | 816211673 | AKE85479.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. C227-11 Taxon ID: 1048254 | 341917152 | EGT66768.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. C227-11 Taxon ID: 1048254 | 354858540 | EHF18990.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. C236-11 Taxon ID: 1048256 | 354864334 | EHF24763.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. E112/10 Taxon ID: 1090928 | 472425837 | EMS08246.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. E92/11 Taxon ID: 1090927 | 472422270 | EMS04764.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-4984 Taxon ID: 1240761 | 429409612 | EKZ45839.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-4986 Taxon ID: 1240762 | 429411708 | EKZ47914.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-4987 Taxon ID: 1240763 | 429418446 | EKZ54590.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-4988 Taxon ID: 1240764 | 429429587 | EKZ65655.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-5603 Taxon ID: 1240765 | 429428066 | EKZ64146.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-5604 Taxon ID: 1240766 | 429435734 | EKZ71752.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-6006 Taxon ID: 1240767 | 429444875 | EKZ80820.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-9450 Taxon ID: 1240758 | 429398695 | EKZ35028.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-9941 Taxon ID: 1240759 | 429456669 | EKZ92514.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec11-9990 Taxon ID: 1240760 | 429398778 | EKZ35110.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec12-0465 Taxon ID: 1240778 | 429439404 | EKZ75387.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. Ec12-0466 Taxon ID: 1240779 | 429450454 | EKZ86349.1 (Genbank) | URP |
| Escherichia coli O104:H4 str. LB226692 Taxon ID: 1040638 | 340741670 | EGR75816.1 (Genbank) | URP |
| Escherichia coli O111:H11 str. CFSAN001630 Taxon ID: 1232151 | 421943734 | EKU01011.1 (Genbank) | URP |
| Escherichia coli O111:H11 str. CVM9455 Taxon ID: 1165939 | 394422076 | EJE95481.1 (Genbank) | URP |
| Escherichia coli O111:H11 str. CVM9534 Taxon ID: 1165946 | 388347028 | EIL12726.1 (Genbank) | URP |
| Escherichia coli O111:H11 str. CVM9545 Taxon ID: 1163391 | 388369613 | EIL33200.1 (Genbank) | URP |
| Escherichia coli O111:H11 str. CVM9553 Taxon ID: 1165947 | 394408483 | EJE83126.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. 2009C-4126 Taxon ID: 1446508 | 612243717 | EZQ37822.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. 2009EL-2169 Taxon ID: 1446509 | 612235297 | EZQ29767.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. 2011C-3453 Taxon ID: 1446510 | 612249149 | EZQ43088.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. CFSAN001632 Taxon ID: 1232154 | 421946563 | EKU03687.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. CVM9570 Taxon ID: 1165945 | 388357730 | EIL22260.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. CVM9574 Taxon ID: 1165943 | 388359951 | EIL24205.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. CVM9602 Taxon ID: 1165942 | 394391910 | EJE68735.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. CVM9634 Taxon ID: 1165944 | 394399576 | EJE75582.1 (Genbank) | URP |
| Escherichia coli O111:H8 str. F6627 Taxon ID: 1446498 | 607389062 | EZA81333.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 01-3076 Taxon ID: 1446512 | 651346208 | KDV14036.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 03-3484 Taxon ID: 1446513 | 606974335 | EZA04685.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 04-3211 Taxon ID: 1446514 | 606956146 | EYZ87221.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 08-4487 Taxon ID: 1446515 | 606643132 | EYW90998.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2009C-4006 Taxon ID: 1446516 | 607757313 | EZE38529.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2009C-4052 Taxon ID: 1446517 | 607769588 | EZE50415.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-3053 Taxon ID: 1446511 | 612234274 | EZQ28756.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-3977 Taxon ID: 1446518 | 606440918 | EYU99185.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4086 Taxon ID: 1446519 | 606432721 | EYU91177.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4221 Taxon ID: 1446520 | 606418184 | EYU77445.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4592 Taxon ID: 1446521 | 606868730 | EYZ04262.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4622 Taxon ID: 1446522 | 606863300 | EYY99142.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4715 Taxon ID: 1446523 | 606861474 | EYY97389.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4735 Taxon ID: 1446524 | 606849907 | EYY86543.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4746 Taxon ID: 1446525 | 606836715 | EYY74008.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4799 Taxon ID: 1446526 | 606826784 | EYY64821.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2010C-4818 Taxon ID: 1446527 | 606829197 | EYY67132.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2011C-3170 Taxon ID: 1446528 | 606770898 | EYY11953.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2011C-3362 Taxon ID: 1446529 | 606766592 | EYY07842.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2011C-3573 Taxon ID: 1446530 | 606750864 | EYX93060.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2011C-3632 Taxon ID: 1446531 | 606735092 | EYX78111.1 (Genbank) | URP |
| Escherichia coli O111:NM str. 2011C-3679 Taxon ID: 1446532 | 606733579 | EYX76654.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6722 Taxon ID: 1446533 | 607638252 | EZD23874.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6723 Taxon ID: 1446534 | 607635168 | EZD20986.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6728 Taxon ID: 1446535 | 607643871 | EZD29399.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6890 Taxon ID: 1446536 | 607658082 | EZD43013.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6895 Taxon ID: 1446537 | 607662910 | EZD47682.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6897 Taxon ID: 1446538 | 607669694 | EZD54139.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6898 Taxon ID: 1446539 | 607670843 | EZD55260.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6904 Taxon ID: 1446540 | 607677239 | EZD61538.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6908 Taxon ID: 1446541 | 607684810 | EZD68729.1 (Genbank) | URP |
| Escherichia coli O111:NM str. K6915 Taxon ID: 1446542 | 607684316 | EZD68251.1 (Genbank) | URP |
| Escherichia coli O113:H21 str. 07-4224 Taxon ID: 1446543 | 606994519 | EZA23538.1 (Genbank) | URP |
| Escherichia coli O118:H16 str. 06-3256 Taxon ID: 1446545 | 606959618 | EYZ90609.1 (Genbank) | URP |
| Escherichia coli O118:H16 str. 06-3612 Taxon ID: 1446546 | 606935358 | EYZ67758.1 (Genbank) | URP |
| Escherichia coli O118:H16 str. 07-4255 Taxon ID: 1446547 | 651690281 | KDV72391.1 (Genbank) | URP |
| Escherichia coli O118:H16 str. 08-3651 Taxon ID: 1446548 | 606657278 | EYX04619.1 (Genbank) | URP |
| Escherichia coli O118:H16 str. 2009C-4446 Taxon ID: 1446549 | 607767774 | EZE48652.1 (Genbank) | URP |
| Escherichia coli O119:H4 str. 03-3458 Taxon ID: 1446551 | 606965717 | EYZ96388.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 03-3227 Taxon ID: 1446552 | 606979430 | EZA09341.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 06-3003 Taxon ID: 1446553 | 606958080 | EYZ89112.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 06-3822 Taxon ID: 1446554 | 606917420 | EYZ50768.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2009C-4050 Taxon ID: 1446555 | 607759965 | EZE41055.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2009C-4659 Taxon ID: 1446556 | 606521093 | EYV75860.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2009C-4750 Taxon ID: 1446557 | 607792361 | EZE72462.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2009EL1302 Taxon ID: 1446558 | 607803024 | EZE82781.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2009EL1412 Taxon ID: 1446559 | 606522657 | EYV77363.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-3609 Taxon ID: 1446560 | 606454202 | EYV11654.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-3794 Taxon ID: 1446561 | 607816903 | EZE96154.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-3840 Taxon ID: 1446562 | 606442521 | EYV00737.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-4254 Taxon ID: 1446563 | 606417901 | EYU77174.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-4732 Taxon ID: 1446564 | 606850773 | EYY87260.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-4824 Taxon ID: 1446565 | 606821297 | EYY59616.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-4966 Taxon ID: 1446566 | 606813120 | EYY52024.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010C-4989 Taxon ID: 1446567 | 606796592 | EYY36264.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2010EL1058 Taxon ID: 1446568 | 606791224 | EYY31201.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2011C-3072 Taxon ID: 1446569 | 606790343 | EYY30390.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2011C-3108 Taxon ID: 1446570 | 606781945 | EYY22301.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2011C-3216 Taxon ID: 1446571 | 606773149 | EYY14125.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2011C-3500 Taxon ID: 1446572 | 606760372 | EYY02105.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2011C-3537 Taxon ID: 1446573 | 606754855 | EYX96826.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. 2011C-3609 Taxon ID: 1446574 | 651674224 | KDV56441.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. F6714 Taxon ID: 1446499 | 607403934 | EZA95621.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. K5198 Taxon ID: 1446575 | 607567732 | EZC54961.1 (Genbank) | URP |
| Escherichia coli O121:H19 str. K5269 Taxon ID: 1446576 | 607571114 | EZC58297.1 (Genbank) | URP |
| Escherichia coli O121:H7 str. 2009C-3299 Taxon ID: 1446578 | 607727088 | EZE09578.1 (Genbank) | URP |
| Escherichia coli O123:H11 str. 2009C-3307 Taxon ID: 1446580 | 607743768 | EZE25502.1 (Genbank) | URP |
| Escherichia coli O127:H27 str. C43/90 Taxon ID: 1090929 | 472427799 | EMS10088.1 (Genbank) | URP |
| Escherichia coli O128:H2 str. 2011C-3317 Taxon ID: 1446581 | 651679132 | KDV61310.1 (Genbank) | URP |
| Escherichia coli E24377A Taxon ID: 331111 | 157078374 | ABV18082.1 (Genbank) | URP |
| Escherichia coli O145:H25 str. 07-3858 Taxon ID: 1446583 | 651653467 | KDV35797.1 (Genbank) | URP |
| Escherichia coli O145:H28 str. 2009C-3292 Taxon ID: 1446584 | 607716262 | EZD98949.1 (Genbank) | URP |
| Escherichia coli O145:H28 str. 4865/96 Taxon ID: 1078035 | 636542118 | KDM73692.1 (Genbank) | URP |
| Escherichia coli O145:H28 str. RM12581 Taxon ID: 1248823 | 628075651 | AHY69177.1 (Genbank) | URP |
| Escherichia coli O145:H28 str. RM12761 Taxon ID: 1248903 | 628027157 | AHY63624.1 (Genbank) | URP |
| Escherichia coli O145:H28 str. RM13514 Taxon ID: 1248902 | 573967085 | AHG07350.1 (Genbank) | URP |
| Escherichia coli O145:H28 str. RM13516 Taxon ID: 1248915 | 573933478 | AHG13283.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 06-3484 Taxon ID: 1446585 | 606939900 | EYZ72019.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 08-4270 Taxon ID: 1446586 | 606643805 | EYW91645.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3507 Taxon ID: 1446587 | 606493000 | EYV49010.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3508 Taxon ID: 1446588 | 607814250 | EZE93608.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3509 Taxon ID: 1446589 | 606492091 | EYV48125.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3510 Taxon ID: 1446590 | 606486675 | EYV42848.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3511 Taxon ID: 1446591 | 606484770 | EYV41003.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3516 Taxon ID: 1446592 | 606476249 | EYV32924.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3517 Taxon ID: 1446593 | 606469584 | EYV26579.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3518 Taxon ID: 1446594 | 606473219 | EYV30052.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3521 Taxon ID: 1446595 | 606463279 | EYV20498.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-3526 Taxon ID: 1446596 | 606457152 | EYV14542.1 (Genbank) | URP |
| Escherichia coli O145:NM str. 2010C-4557C2 Taxon ID: 1446597 | 606881184 | EYZ16220.1 (Genbank) | URP |
| Escherichia coli O146:H21 str. 2010C-3325 Taxon ID: 1446599 | 651656736 | KDV39063.1 (Genbank) | URP |
| Escherichia coli O153:H2 str. 2010C-5034 Taxon ID: 1446603 | 606802891 | EYY42152.1 (Genbank) | URP |
| Escherichia coli O156:H25 str. 2011C-3602 Taxon ID: 1446605 | 606741562 | EYX84231.1 (Genbank) | URP |
| Escherichia coli O157 str. 2010EL-2044 Taxon ID: 1446606 | 612260530 | EZQ54181.1 (Genbank) | URP |
| Escherichia coli O157 str. 2010EL-2045 Taxon ID: 1446607 | 612244502 | EZQ38582.1 (Genbank) | URP |
| Escherichia coli O157:H- str. 493-89 Taxon ID: 926027 | 320643483 | EFX12653.1 (Genbank) | URP |
| Escherichia coli O157:H- str. H 2687 Taxon ID: 926028 | 320648818 | EFX17445.1 (Genbank) | URP |
| Escherichia coli O157:H16 str. 98-3133 Taxon ID: 1446608 | 607378138 | EZA70669.1 (Genbank) | URP |
| Escherichia coli O157:H43 str. T22 Taxon ID: 1137135 | 478696495 | ENO08659.1 (Genbank) | URP |
| Escherichia coli O157:H7 Taxon ID: 83334 | 814570333 | KKF75244.1 (Genbank) | URP |
| Escherichia coli O157:H7 Taxon ID: 83334 | 814581312 | KKF84872.1 (Genbank) | URP |
| Escherichia coli O157:H7 Taxon ID: 83334 | 821097231 | KKY48905.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 06-3745 Taxon ID: 1446609 | 606918495 | EYZ51817.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 06-4039 Taxon ID: 1446610 | 606905133 | EYZ39142.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 07-3091 Taxon ID: 1446611 | 606901684 | EYZ35783.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 08-3037 Taxon ID: 1446613 | 606665696 | EYX12576.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 08-3527 Taxon ID: 1446614 | 606666642 | EYX13497.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 08-4169 Taxon ID: 1446615 | 606647575 | EYW95274.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 08-4529 Taxon ID: 1446616 | 607701794 | EZD85019.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 1044 Taxon ID: 997824 | 326341379 | EGD65171.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 1125 Taxon ID: 997825 | 326345843 | EGD69582.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2009C-4258 Taxon ID: 1446617 | 607778341 | EZE58810.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2009EL1449 Taxon ID: 1446618 | 607793516 | EZE73576.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2009EL1705 Taxon ID: 1446619 | 606510774 | EYV66204.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2009EL1913 Taxon ID: 1446620 | 607803603 | EZE83350.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2009EL2109 Taxon ID: 1446621 | 606497398 | EYV53268.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2010C-4979C1 Taxon ID: 1446622 | 606811968 | EYY50921.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-1107 Taxon ID: 1446623 | 606722705 | EYX66639.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2090 Taxon ID: 1446624 | 606715267 | EYX59615.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2091 Taxon ID: 1446625 | 606707034 | EYX51687.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2092 Taxon ID: 1446626 | 606702306 | EYX47233.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2093 Taxon ID: 1446627 | 606692083 | EYX37545.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2094 Taxon ID: 1446628 | 606694778 | EYX40082.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2096 Taxon ID: 1446629 | 606686370 | EYX32105.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2097 Taxon ID: 1446630 | 606683388 | EYX29372.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2098 Taxon ID: 1446631 | 606680217 | EYX26314.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2099 Taxon ID: 1446632 | 606633837 | EYW82414.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2101 Taxon ID: 1446633 | 606631163 | EYW79840.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2103 Taxon ID: 1446634 | 606625268 | EYW74201.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2104 Taxon ID: 1446635 | 606620464 | EYW69766.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2105 Taxon ID: 1446636 | 606612922 | EYW62517.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2106 Taxon ID: 1446637 | 606608242 | EYW58173.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2107 Taxon ID: 1446638 | 606600694 | EYW51081.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2108 Taxon ID: 1446639 | 606601912 | EYW52255.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2109 Taxon ID: 1446640 | 606596938 | EYW47434.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2111 Taxon ID: 1446641 | 606589826 | EYW40904.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2112 Taxon ID: 1446642 | 606588402 | EYW39582.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2113 Taxon ID: 1446643 | 606586664 | EYW37886.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2114 Taxon ID: 1446644 | 606568171 | EYW20297.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2286 Taxon ID: 1446645 | 606571436 | EYW23496.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2287 Taxon ID: 1446646 | 606567143 | EYW19292.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2288 Taxon ID: 1446647 | 606557548 | EYW10297.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2289 Taxon ID: 1446648 | 606559897 | EYW12519.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2290 Taxon ID: 1446649 | 607829714 | EZF07528.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2312 Taxon ID: 1446650 | 606549722 | EYW02704.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. 2011EL-2313 Taxon ID: 1446651 | 607824579 | EZF02467.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC1212 Taxon ID: 941435 | 320193034 | EFW67674.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC4042 Taxon ID: 444449 | 208737303 | EDZ84987.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC4045 Taxon ID: 444448 | 208731927 | EDZ80615.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC4076 Taxon ID: 444453 | 189003094 | EDU72080.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC4115 Taxon ID: 444450 | 209160563 | ACI37996.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC4401 Taxon ID: 478004 | 189358108 | EDU76527.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC4486 Taxon ID: 478005 | 189360846 | EDU79265.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. EC869 Taxon ID: 478008 | 189373473 | EDU91889.1 (Genbank) | URP |
| Escherichia coli O157:H7 EDL933 Taxon ID: 155864 | 667690293 | AIG66902.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F6142 Taxon ID: 1446652 | 607394199 | EZA86223.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F6749 Taxon ID: 1446653 | 607411864 | EZB03294.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F6750 Taxon ID: 1446654 | 607413194 | EZB04603.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F6751 Taxon ID: 1446655 | 607417332 | EZB08547.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F7350 Taxon ID: 1446656 | 606537772 | EYV91596.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F7377 Taxon ID: 1446657 | 607423145 | EZB14085.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. F7410 Taxon ID: 1446659 | 607432286 | EZB22876.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. G5101 Taxon ID: 926026 | 320638077 | EFX07841.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. G5303 Taxon ID: 1446660 | 607441151 | EZB31517.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. H2495 Taxon ID: 1446661 | 607445550 | EZB35718.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. H2498 Taxon ID: 1446662 | 607450493 | EZB40509.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1420 Taxon ID: 1446663 | 607453839 | EZB43824.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1792 Taxon ID: 1446664 | 607471226 | EZB60758.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1793 Taxon ID: 1446665 | 607467350 | EZB56917.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1795 Taxon ID: 1446666 | 607483381 | EZB72683.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1796 Taxon ID: 1446667 | 607486936 | EZB76193.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1845 Taxon ID: 1446668 | 607478865 | EZB68286.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1921 Taxon ID: 1446669 | 607498029 | EZB86956.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K1927 Taxon ID: 1446670 | 607488566 | EZB77795.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2188 Taxon ID: 1446671 | 607502670 | EZB91545.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2191 Taxon ID: 1446672 | 607512696 | EZC01318.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2192 Taxon ID: 1446673 | 607519379 | EZC07861.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2324 Taxon ID: 1446674 | 607516313 | EZC04858.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2581 Taxon ID: 1446675 | 607526849 | EZC15151.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2622 Taxon ID: 1446676 | 607528657 | EZC16908.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2845 Taxon ID: 1446677 | 607535222 | EZC23341.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K2854 Taxon ID: 1446678 | 607542526 | EZC30531.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K4396 Taxon ID: 1446679 | 607547476 | EZC35337.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K4405 Taxon ID: 1446680 | 607550976 | EZC38767.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K4406 Taxon ID: 1446681 | 607553814 | EZC41556.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K4527 Taxon ID: 1446682 | 607558623 | EZC46101.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5418 Taxon ID: 1446683 | 607570460 | EZC57652.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5448 Taxon ID: 1446684 | 607580244 | EZC67275.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5449 Taxon ID: 1446685 | 607586183 | EZC73131.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5453 Taxon ID: 1446686 | 607585014 | EZC71977.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5460 Taxon ID: 1446687 | 607593886 | EZC80707.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5467 Taxon ID: 1446688 | 607601812 | EZC88444.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5602 Taxon ID: 1446689 | 607605586 | EZC92127.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5607 Taxon ID: 1446690 | 607609092 | EZC95584.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5609 Taxon ID: 1446691 | 607607890 | EZC94406.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5806 Taxon ID: 1446692 | 606529432 | EYV83922.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K5852 Taxon ID: 1446693 | 607618988 | EZD05252.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K6590 Taxon ID: 1446694 | 607620091 | EZD06339.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K6676 Taxon ID: 1446695 | 607625537 | EZD11636.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K6687 Taxon ID: 1446696 | 607632307 | EZD18256.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. K7140 Taxon ID: 1446697 | 607690981 | EZD74691.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. LSU-61 Taxon ID: 926032 | 320664882 | EFX32017.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. SS17 Taxon ID: 1328859 | 666002403 | AIF92260.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. SS52 Taxon ID: 1330457 | 734587414 | AJA24615.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. TW14359 Taxon ID: 544404 | 254591194 | ACT70555.1 (Genbank) | URP |
| Escherichia coli O157:H7 str. TW14588 Taxon ID: 502346 | 217320612 | EEC29036.1 (Genbank) | URP |
| Escherichia coli O157:NM str. 08-4540 Taxon ID: 1446698 | 607708166 | EZD91195.1 (Genbank) | URP |
| Escherichia coli O15:H18 str. K1516 Taxon ID: 1446601 | 607474586 | EZB64089.1 (Genbank) | URP |
| Escherichia coli O165:H25 str. 2010C-4874 Taxon ID: 1446700 | 606818823 | EYY57366.1 (Genbank) | URP |
| Escherichia coli O169:H41 str. F9792 Taxon ID: 1446702 | 607437260 | EZB27748.1 (Genbank) | URP |
| Escherichia coli O174:H21 str. 03-3269 Taxon ID: 1446703 | 606969172 | EYZ99749.1 (Genbank) | URP |
| Escherichia coli O174:H8 str. 04-3038 Taxon ID: 1446704 | 607008566 | EZA37026.1 (Genbank) | URP |
| Escherichia coli O177:NM str. 2010C-4558 Taxon ID: 1446706 | 606871682 | EYZ07085.1 (Genbank) | URP |
| Escherichia coli O25:NM str. E2539C1 Taxon ID: 1446708 | 607388411 | EZA80707.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2009C-4747 Taxon ID: 1446715 | 612241042 | EZQ35258.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 03-3500 Taxon ID: 1446710 | 608655802 | EZG50801.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. 05-3646 Taxon ID: 1446500 | 607015159 | EZA43358.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 06-3464 Taxon ID: 1446711 | 608657246 | EZG52188.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2009C-3612 Taxon ID: 1446712 | 608720625 | EZH10272.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2009C-3689 Taxon ID: 1446713 | 608718999 | EZH08712.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2009C-3996 Taxon ID: 1446714 | 608736376 | EZH24870.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2009C-4760 Taxon ID: 1446716 | 608739255 | EZH27649.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2009C-4826 Taxon ID: 1446717 | 608735990 | EZH24500.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-3051 Taxon ID: 1446718 | 608750857 | EZH38256.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-3472 Taxon ID: 1446719 | 608755941 | EZH43068.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-3871 Taxon ID: 1446720 | 608756078 | EZH43195.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-3902 Taxon ID: 1446721 | 608769201 | EZH55377.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-4244 Taxon ID: 1446722 | 608769806 | EZH55950.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-4430 Taxon ID: 1446723 | 608666823 | EZG60772.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-4819 Taxon ID: 1446724 | 608664077 | EZG58201.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-4834 Taxon ID: 1446725 | 608681438 | EZG74133.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010C-5028 Taxon ID: 1446726 | 608681561 | EZG74253.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2010EL-1699 Taxon ID: 1446727 | 608692783 | EZG84384.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2011C-3270 Taxon ID: 1446728 | 608692656 | EZG84260.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. 2011C-3274 Taxon ID: 1446729 | 651693624 | KDV75724.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2011C-3282 Taxon ID: 1446730 | 608713914 | EZH04269.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2011C-3387 Taxon ID: 1446731 | 608706123 | EZG96761.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2011C-3506 Taxon ID: 1446732 | 608713221 | EZH03601.1 (Genbank) | URP |
| Escherichia coli O26:H1 str. 2011C-3655 Taxon ID: 1446733 | 608726264 | EZH15745.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CFSAN001629 Taxon ID: 1232150 | 421940946 | EKT98373.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CVM10021 Taxon ID: 1165950 | 394405439 | EJE80620.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CVM10026 Taxon ID: 1165951 | 388379626 | EIL42274.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CVM10030 Taxon ID: 1165952 | 394431662 | EJF03834.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CVM10224 Taxon ID: 1165953 | 394394285 | EJE70894.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CVM9942 Taxon ID: 1165949 | 388378301 | EIL41058.1 (Genbank) | URP |
| Escherichia coli O26:H11 str. CVM9952 Taxon ID: 1163738 | 394433593 | EJF05602.1 (Genbank) | URP |
| Escherichia coli O26:NM str. 2010C-4347 Taxon ID: 1446734 | 606424917 | EYU83928.1 (Genbank) | URP |
| Escherichia coli O26:NM str. 2010C-4788 Taxon ID: 1446735 | 606839294 | EYY76421.1 (Genbank) | URP |
| Escherichia coli O28ac:NM str. 02-3404 Taxon ID: 1446737 | 606984867 | EZA14365.1 (Genbank) | URP |
| Escherichia coli O32:H37 str. P4 Taxon ID: 1167694 | 385154668 | EIF16678.1 (Genbank) | URP |
| Escherichia coli O39:NM str. F8704-2 Taxon ID: 1446739 | 607698000 | EZD81312.1 (Genbank) | URP |
| Escherichia coli O45:H2 str. 01-3147 Taxon ID: 1446740 | 607000306 | EZA29116.1 (Genbank) | URP |
| Escherichia coli O45:H2 str. 2009C-3686 Taxon ID: 1446741 | 607739270 | EZE21094.1 (Genbank) | URP |
| Escherichia coli O45:H2 str. 2009C-4780 Taxon ID: 1446742 | 607782684 | EZE63046.1 (Genbank) | URP |
| Escherichia coli O45:H2 str. 2009C-4780 Taxon ID: 1446742 | 607789111 | EZE69318.1 (Genbank) | URP |
| Escherichia coli O45:H2 str. 2010C-3876 Taxon ID: 1446743 | 606435313 | EYU93692.1 (Genbank) | URP |
| Escherichia coli O45:H2 str. 2010C-4211 Taxon ID: 1446744 | 651673032 | KDV55261.1 (Genbank) | URP |
| Escherichia coli O55:H7 str. 06-3555 Taxon ID: 1446745 | 606929214 | EYZ61911.1 (Genbank) | URP |
| Escherichia coli O55:H7 str. 3256-97 Taxon ID: 926029 | 320654404 | EFX22451.1 (Genbank) | URP |
| Escherichia coli O55:H7 str. CB9615 Taxon ID: 701177 | 290761452 | ADD55413.1 (Genbank) | URP |
| Escherichia coli O55:H7 str. RM12579 Taxon ID: 1048689 | 374357684 | AEZ39391.1 (Genbank) | URP |
| Escherichia coli O55:H7 str. USDA 5905 Taxon ID: 926030 | 320660085 | EFX27615.1 (Genbank) | URP |
| Escherichia coli O69:H11 str. 06-3325 Taxon ID: 1446750 | 606943173 | EYZ75156.1 (Genbank) | URP |
| Escherichia coli O69:H11 str. 07-3763 Taxon ID: 1446751 | 651652901 | KDV35233.1 (Genbank) | URP |
| Escherichia coli O69:H11 str. 07-4281 Taxon ID: 1446752 | 606673324 | EYX19720.1 (Genbank) | URP |
| Escherichia coli O69:H11 str. 08-4661 Taxon ID: 1446753 | 607722964 | EZE05494.1 (Genbank) | URP |
| Escherichia coli O69:H11 str. 2009C-3601 Taxon ID: 1446754 | 607748984 | EZE30610.1 (Genbank) | URP |
| Escherichia coli O6:H16 str. 99-3165 Taxon ID: 1446747 | 606535956 | EYV89833.1 (Genbank) | URP |
| Escherichia coli O6:H16 str. F5656C1 Taxon ID: 1446748 | 607381084 | EZA73559.1 (Genbank) | URP |
| Escherichia coli O6:H16:CFA/II str. B2C Taxon ID: 1370107 | 572725556 | ETS24433.1 (Genbank) | URP |
| Escherichia coli O78:H12 str. 00-3279 Taxon ID: 1446756 | 651348842 | KDV16562.1 (Genbank) | URP |
| Escherichia coli O79:H7 str. 06-3501 Taxon ID: 1446760 | 606924796 | EYZ57632.1 (Genbank) | URP |
| Escherichia coli O7:K1 str. CE10 Taxon ID: 1072459 | 349736759 | AEQ11465.1 (Genbank) | URP |
| Escherichia coli O81:NM str. 02-3012 Taxon ID: 1446761 | 606987984 | EZA17319.1 (Genbank) | URP |
| Escherichia coli O83:H1 str. NRG 857C Taxon ID: 685038 | 312945175 | ADR26002.1 (Genbank) | URP |
| Escherichia coli O86:H34 str. 99-3124 Taxon ID: 1446762 | 606544935 | EYV98519.1 (Genbank) | URP |
| Escherichia coli O91:H14 str. 06-3691 Taxon ID: 1446763 | 606912700 | EYZ46174.1 (Genbank) | URP |
| Escherichia coli O91:H14 str. 2009C-3227 Taxon ID: 1446764 | 607710980 | EZD93869.1 (Genbank) | URP |
| Escherichia coli O91:H21 str. 2009C-3740 Taxon ID: 1446765 | 651664619 | KDV46890.1 (Genbank) | URP |
| Escherichia coli O91:H21 str. 2009C-4646 Taxon ID: 1446766 | 607785680 | EZE65982.1 (Genbank) | URP |
| Escherichia coli O91:NM str. 2009C-3745 Taxon ID: 1446768 | 607747820 | EZE29471.1 (Genbank) | URP |
| Escherichia coli 1180 Taxon ID: 670904 | 323179896 | EFZ65453.1 (Genbank) | URP |
| Escherichia coli 1357 Taxon ID: 670905 | 323185018 | EFZ70385.1 (Genbank) | URP |
| Escherichia coli ONT:H33 str. C48/93 Taxon ID: 1090930 | 472415254 | EMR97926.1 (Genbank) | URP |
| Escherichia coli P0298942.1 Taxon ID: 1116141 | 476717229 | ENA22191.1 (Genbank) | URP |
| Escherichia coli P0298942.10 Taxon ID: 1116147 | 476849771 | ENB45047.1 (Genbank) | URP |
| Escherichia coli P0298942.12 Taxon ID: 1116149 | 477422674 | ENG87684.1 (Genbank) | URP |
| Escherichia coli P0298942.15 Taxon ID: 1116151 | 476873844 | ENB66334.1 (Genbank) | URP |
| Escherichia coli P0298942.2 Taxon ID: 1116142 | 476877781 | ENB69983.1 (Genbank) | URP |
| Escherichia coli P0298942.6 Taxon ID: 1116143 | 476874310 | ENB66775.1 (Genbank) | URP |
| Escherichia coli P0298942.7 Taxon ID: 1116144 | 476890300 | ENB81536.1 (Genbank) | URP |
| Escherichia coli P0298942.9 Taxon ID: 1116146 | 476896383 | ENB87277.1 (Genbank) | URP |
| Escherichia coli P0299438.10 Taxon ID: 1116045 | 476899415 | ENB90139.1 (Genbank) | URP |
| Escherichia coli P0299438.2 Taxon ID: 1116152 | 476702554 | ENA07700.1 (Genbank) | URP |
| Escherichia coli P0299438.4 Taxon ID: 1116039 | 476917578 | ENC07435.1 (Genbank) | URP |
| Escherichia coli P0299438.7 Taxon ID: 1116042 | 476933386 | ENC22873.1 (Genbank) | URP |
| Escherichia coli P0299438.8 Taxon ID: 1116043 | 476938091 | ENC27388.1 (Genbank) | URP |
| Escherichia coli P0299483.2 Taxon ID: 1125649 | 477102269 | END82710.1 (Genbank) | URP |
| Escherichia coli P02997067.6 Taxon ID: 1116131 | 476948123 | ENC37043.1 (Genbank) | URP |
| Escherichia coli P0299917.1 Taxon ID: 1116047 | 476702692 | ENA07835.1 (Genbank) | URP |
| Escherichia coli P0299917.10 Taxon ID: 1116056 | 476953623 | ENC42244.1 (Genbank) | URP |
| Escherichia coli P0299917.3 Taxon ID: 1116049 | 476976925 | ENC64161.1 (Genbank) | URP |
| Escherichia coli P0299917.4 Taxon ID: 1116050 | 476971991 | ENC59442.1 (Genbank) | URP |
| Escherichia coli P0299917.5 Taxon ID: 1116051 | 476976807 | ENC64048.1 (Genbank) | URP |
| Escherichia coli P0299917.7 Taxon ID: 1116053 | 476996205 | ENC82604.1 (Genbank) | URP |
| Escherichia coli P0301867.1 Taxon ID: 1116066 | 476304047 | EMX21905.1 (Genbank) | URP |
| Escherichia coli P0301867.11 Taxon ID: 1116072 | 477007569 | ENC93323.1 (Genbank) | URP |
| Escherichia coli P0301867.4 Taxon ID: 1116068 | 476736856 | ENA41440.1 (Genbank) | URP |
| Escherichia coli P0301867.5 Taxon ID: 1116069 | 477440624 | ENH04625.1 (Genbank) | URP |
| Escherichia coli P0301904.3 Taxon ID: 1125647 | 477119854 | END99418.1 (Genbank) | URP |
| Escherichia coli P0302293.10 Taxon ID: 1116079 | 477150825 | ENE28644.1 (Genbank) | URP |
| Escherichia coli P0302293.2 Taxon ID: 1116073 | 476299631 | EMX17549.1 (Genbank) | URP |
| Escherichia coli P0302293.3 Taxon ID: 1116074 | 477146745 | ENE24848.1 (Genbank) | URP |
| Escherichia coli P0302293.6 Taxon ID: 1116076 | 477161439 | ENE38671.1 (Genbank) | URP |
| Escherichia coli P0302293.7 Taxon ID: 1125643 | 477122737 | ENE02172.1 (Genbank) | URP |
| Escherichia coli P0302308.1 Taxon ID: 1116153 | 476292284 | EMX10300.1 (Genbank) | URP |
| Escherichia coli P0302308.10 Taxon ID: 1116158 | 477017190 | END02474.1 (Genbank) | URP |
| Escherichia coli P0302308.11 Taxon ID: 1116159 | 477020435 | END05532.1 (Genbank) | URP |
| Escherichia coli P0302308.12 Taxon ID: 1116160 | 477461519 | ENH24418.1 (Genbank) | URP |
| Escherichia coli P0302308.13 Taxon ID: 1116161 | 477463057 | ENH25915.1 (Genbank) | URP |
| Escherichia coli P0302308.4 Taxon ID: 1116156 | 477044785 | END28334.1 (Genbank) | URP |
| Escherichia coli P0304777.1 Taxon ID: 1116163 | 476286191 | EMX04287.1 (Genbank) | URP |
| Escherichia coli P0304777.10 Taxon ID: 1116171 | 477173324 | ENE50141.1 (Genbank) | URP |
| Escherichia coli P0304777.12 Taxon ID: 1116173 | 477193076 | ENE69184.1 (Genbank) | URP |
| Escherichia coli P0304777.13 Taxon ID: 1116174 | 477195150 | ENE71222.1 (Genbank) | URP |
| Escherichia coli P0304777.14 Taxon ID: 1116175 | 477199934 | ENE75894.1 (Genbank) | URP |
| Escherichia coli P0304777.15 Taxon ID: 1116176 | 477205288 | ENE81092.1 (Genbank) | URP |
| Escherichia coli P0304777.2 Taxon ID: 1116164 | 477210155 | ENE85786.1 (Genbank) | URP |
| Escherichia coli P0304777.3 Taxon ID: 1116165 | 477213567 | ENE88979.1 (Genbank) | URP |
| Escherichia coli P0304816.1 Taxon ID: 1116177 | 476694395 | EMZ99649.1 (Genbank) | URP |
| Escherichia coli P0304816.10 Taxon ID: 1116186 | 477253380 | ENF26429.1 (Genbank) | URP |
| Escherichia coli P0304816.12 Taxon ID: 1116188 | 477261964 | ENF34644.1 (Genbank) | URP |
| Escherichia coli P0304816.13 Taxon ID: 1116189 | 477270359 | ENF42591.1 (Genbank) | URP |
| Escherichia coli P0304816.14 Taxon ID: 1116190 | 477265673 | ENF38204.1 (Genbank) | URP |
| Escherichia coli P0304816.3 Taxon ID: 1116179 | 477476053 | ENH38302.1 (Genbank) | URP |
| Escherichia coli P0304816.4 Taxon ID: 1116180 | 477474953 | ENH37224.1 (Genbank) | URP |
| Escherichia coli P0304816.7 Taxon ID: 1116183 | 477292987 | ENF64095.1 (Genbank) | URP |
| Escherichia coli P0304816.9 Taxon ID: 1116185 | 477305560 | ENF76255.1 (Genbank) | URP |
| Escherichia coli P0305260.1 Taxon ID: 1125644 | 476689963 | EMZ95285.1 (Genbank) | URP |
| Escherichia coli P0305260.12 Taxon ID: 1116086 | 477320755 | ENF90809.1 (Genbank) | URP |
| Escherichia coli P0305260.2 Taxon ID: 1125645 | 477133387 | ENE12422.1 (Genbank) | URP |
| Escherichia coli P0305260.7 Taxon ID: 1116081 | 477353493 | ENG22287.1 (Genbank) | URP |
| Escherichia coli P0305260.8 Taxon ID: 1116082 | 477360731 | ENG29293.1 (Genbank) | URP |
| Escherichia coli P0305260.9 Taxon ID: 1116083 | 477368963 | ENG37285.1 (Genbank) | URP |
| Escherichia coli P12b Taxon ID: 910348 | 383101978 | AFG39487.1 (Genbank) | URP |
| Escherichia coli P4-96 Taxon ID: 1097579 | 558714943 | EST66203.1 (Genbank) | URP |
| Escherichia coli P4-NR Taxon ID: 1117111 | 558719585 | EST70740.1 (Genbank) | URP |
| Escherichia coli PA10 Taxon ID: 1005494 | 390708228 | EIN81492.1 (Genbank) | URP |
| Escherichia coli PA11 Taxon ID: 1051349 | 444599765 | ELV74623.1 (Genbank) | URP |
| Escherichia coli PA13 Taxon ID: 1051350 | 444614170 | ELV88408.1 (Genbank) | URP |
| Escherichia coli PA15 Taxon ID: 1005496 | 390709931 | EIN82970.1 (Genbank) | URP |
| Escherichia coli PA19 Taxon ID: 1051351 | 444614379 | ELV88607.1 (Genbank) | URP |
| Escherichia coli PA2 Taxon ID: 1005487 | 444622967 | ELV96910.1 (Genbank) | URP |
| Escherichia coli PA22 Taxon ID: 1005497 | 390720335 | EIN93047.1 (Genbank) | URP |
| Escherichia coli PA23 Taxon ID: 1005498 | 408150174 | EKH78782.1 (Genbank) | URP |
| Escherichia coli PA25 Taxon ID: 1005500 | 390733328 | EIO04912.1 (Genbank) | URP |
| Escherichia coli PA28 Taxon ID: 1005501 | 390736436 | EIO07770.1 (Genbank) | URP |
| Escherichia coli PA3 Taxon ID: 1005488 | 390688499 | EIN63557.1 (Genbank) | URP |
| Escherichia coli PA31 Taxon ID: 1005502 | 390751803 | EIO21676.1 (Genbank) | URP |
| Escherichia coli PA32 Taxon ID: 1005503 | 390752174 | EIO22023.1 (Genbank) | URP |
| Escherichia coli PA34 Taxon ID: 1005505 | 408085873 | EKH19443.1 (Genbank) | URP |
| Escherichia coli PA38 Taxon ID: 1005506 | 408232666 | EKI55835.1 (Genbank) | URP |
| Escherichia coli PA39 Taxon ID: 1005507 | 390783394 | EIO51000.1 (Genbank) | URP |
| Escherichia coli PA4 Taxon ID: 1005489 | 408141277 | EKH70751.1 (Genbank) | URP |
| Escherichia coli PA40 Taxon ID: 1005508 | 390762605 | EIO31863.1 (Genbank) | URP |
| Escherichia coli PA41 Taxon ID: 1005509 | 390775971 | EIO43950.1 (Genbank) | URP |
| Escherichia coli PA42 Taxon ID: 1005510 | 390777775 | EIO45553.1 (Genbank) | URP |
| Escherichia coli PA45 Taxon ID: 1005511 | 408157655 | EKH85798.1 (Genbank) | URP |
| Escherichia coli PA47 Taxon ID: 1005512 | 444632394 | ELW05965.1 (Genbank) | URP |
| Escherichia coli PA48 Taxon ID: 1005513 | 444632092 | ELW05669.1 (Genbank) | URP |
| Escherichia coli PA49 Taxon ID: 1005514 | 408152712 | EKH81140.1 (Genbank) | URP |
| Escherichia coli PA7 Taxon ID: 1005491 | 408072069 | EKH06394.1 (Genbank) | URP |
| Escherichia coli PA8 Taxon ID: 1005492 | 444636867 | ELW10251.1 (Genbank) | URP |
| Escherichia coli PA9 Taxon ID: 1005493 | 390691908 | EIN66630.1 (Genbank) | URP |
| Escherichia coli PCN033 Taxon ID: 1001989 | 828387410 | AKK47240.1 (Genbank) | URP |
| Escherichia coli PCN061 Taxon ID: 1358422 | 834763733 | AKM34205.1 (Genbank) | URP |
| Escherichia coli RS218 Taxon ID: 439184 | 756768499 | AJM72770.1 (Genbank) | URP |
| Escherichia coli RS218 Taxon ID: 439184 | 746692757 | KIE83872.1 (Genbank) | URP |
| Escherichia coli S17 Taxon ID: 1227813 | 449324728 | EMD14652.1 (Genbank) | URP |
| Escherichia coli SCD1 Taxon ID: 1337384 | 553020544 | ESA27431.1 (Genbank) | URP |
| Escherichia coli SCD2 Taxon ID: 1337385 | 553023953 | ESA30780.1 (Genbank) | URP |
| Escherichia coli SCI-07 Taxon ID: 1144302 | 380349526 | EIA37798.1 (Genbank) | URP |
| Escherichia coli SEPT362 Taxon ID: 1196033 | 449324654 | EMD14579.1 (Genbank) | URP |
| Escherichia coli SMS-3-5 Taxon ID: 439855 | 170519018 | ACB17196.1 (Genbank) | URP |
| Escherichia coli STEC_94C Taxon ID: 754083 | 345353453 | EGW85686.1 (Genbank) | URP |
| Escherichia coli STEC_C165-02 Taxon ID: 754085 | 345340944 | EGW73360.1 (Genbank) | URP |
| Escherichia coli STEC_EH250 Taxon ID: 754087 | 345366134 | EGW98232.1 (Genbank) | URP |
| Escherichia coli STEC_H.1.8 Taxon ID: 754088 | 345385652 | EGX15491.1 (Genbank) | URP |
| Escherichia coli STEC_MHI813 Taxon ID: 754089 | 345377862 | EGX09793.1 (Genbank) | URP |
| Escherichia coli STEC_O31 Taxon ID: 754081 | 397786674 | EJK97507.1 (Genbank) | URP |
| Escherichia coli STEC_S1191 Taxon ID: 754090 | 345390767 | EGX20564.1 (Genbank) | URP |
| Escherichia coli SWW33 Taxon ID: 1235805 | 476641411 | EMZ47832.1 (Genbank) | URP |
| Escherichia coli T1282_01 Taxon ID: 1081891 | 537056189 | ERE26306.1 (Genbank) | URP |
| Escherichia coli T1840_97 Taxon ID: 1081896 | 536895426 | ERC76450.1 (Genbank) | URP |
| Escherichia coli T234_00 Taxon ID: 1081892 | 536905764 | ERC86230.1 (Genbank) | URP |
| Escherichia coli T924_01 Taxon ID: 1081893 | 536909467 | ERC89543.1 (Genbank) | URP |
| Escherichia coli TA007 Taxon ID: 656429 | 323972083 | EGB67297.1 (Genbank) | URP |
| Escherichia coli TA124 Taxon ID: 656435 | 371595014 | EHN83868.1 (Genbank) | URP |
| Escherichia coli TA143 Taxon ID: 656437 | 331060414 | EGI32378.1 (Genbank) | URP |
| Escherichia coli TA206 Taxon ID: 656440 | 331054903 | EGI26912.1 (Genbank) | URP |
| Escherichia coli TA271 Taxon ID: 656443 | 331066204 | EGI38088.1 (Genbank) | URP |
| Escherichia coli TA280 Taxon ID: 656444 | 331070635 | EGI41998.1 (Genbank) | URP |
| Escherichia coli TT12B Taxon ID: 1005555 | 408166670 | EKH94221.1 (Genbank) | URP |
| Escherichia coli TW00353 Taxon ID: 1005564 | 408205754 | EKI30589.1 (Genbank) | URP |
| Escherichia coli TW06591 Taxon ID: 1005515 | 390785937 | EIO53479.1 (Genbank) | URP |
| Escherichia coli TW07509 Taxon ID: 1105330 | 536880666 | ERC62737.1 (Genbank) | URP |
| Escherichia coli TW07793 Taxon ID: 869693 | 386247566 | EII93739.1 (Genbank) | URP |
| Escherichia coli TW07945 Taxon ID: 1005516 | 390809419 | EIO76213.1 (Genbank) | URP |
| Escherichia coli TW09098 Taxon ID: 1005519 | 390822771 | EIO88867.1 (Genbank) | URP |
| Escherichia coli TW10119 Taxon ID: 1005522 | 390819170 | EIO85519.1 (Genbank) | URP |
| Escherichia coli TW10246 Taxon ID: 1005517 | 390791931 | EIO59294.1 (Genbank) | URP |
| Escherichia coli TW10509 Taxon ID: 656449 | 323976390 | EGB71480.1 (Genbank) | URP |
| Escherichia coli TW11039 Taxon ID: 1005518 | 390801994 | EIO69045.1 (Genbank) | URP |
| Escherichia coli TW14301 Taxon ID: 1005526 | 390855365 | EIP18085.1 (Genbank) | URP |
| Escherichia coli TW14313 Taxon ID: 1005525 | 390859655 | EIP21999.1 (Genbank) | URP |
| Escherichia coli TW15901 Taxon ID: 1005562 | 408198371 | EKI23601.1 (Genbank) | URP |
| Escherichia coli TX1999 Taxon ID: 670903 | 345395244 | EGX24993.1 (Genbank) | URP |
| Escherichia coli Tx1686 Taxon ID: 1207037 | 537071725 | ERE40923.1 (Genbank) | URP |
| Escherichia coli Tx3800 Taxon ID: 1207036 | 537076181 | ERE44741.1 (Genbank) | URP |
| Escherichia coli UCI 51 Taxon ID: 1438689 | 635895438 | KDG75997.1 (Genbank) | URP |
| Escherichia coli UCI 53 Taxon ID: 1438691 | 635900736 | KDG81242.1 (Genbank) | URP |
| Escherichia coli UCI 57 Taxon ID: 1438693 | 635907884 | KDG88337.1 (Genbank) | URP |
| Escherichia coli UCI 58 Taxon ID: 1438694 | 635911453 | KDG91876.1 (Genbank) | URP |
| Escherichia coli UCI 65 Taxon ID: 1438695 | 635921294 | KDH01658.1 (Genbank) | URP |
| Escherichia coli UCI 66 Taxon ID: 1438696 | 635921472 | KDH01834.1 (Genbank) | URP |
| Escherichia coli UM146 Taxon ID: 869729 | 307627934 | ADN72238.1 (Genbank) | URP |
| Escherichia coli UMEA 3014-1 Taxon ID: 1281173 | 535483287 | EQW22789.1 (Genbank) | URP |
| Escherichia coli UMEA 3022-1 Taxon ID: 1281174 | 535482895 | EQW22400.1 (Genbank) | URP |
| Escherichia coli UMEA 3033-1 Taxon ID: 1281175 | 535493144 | EQW32521.1 (Genbank) | URP |
| Escherichia coli UMEA 3041-1 Taxon ID: 1281176 | 535496697 | EQW36089.1 (Genbank) | URP |
| Escherichia coli UMEA 3052-1 Taxon ID: 1281177 | 535498573 | EQW37907.1 (Genbank) | URP |
| Escherichia coli UMEA 3053-1 Taxon ID: 1281178 | 535507610 | EQW46478.1 (Genbank) | URP |
| Escherichia coli UMEA 3065-1 Taxon ID: 1281179 | 535509413 | EQW48161.1 (Genbank) | URP |
| Escherichia coli UMEA 3087-1 Taxon ID: 1281180 | 535515910 | EQW54495.1 (Genbank) | URP |
| Escherichia coli UMEA 3088-1 Taxon ID: 1281181 | 535528592 | EQW66997.1 (Genbank) | URP |
| Escherichia coli UMEA 3097-1 Taxon ID: 1281182 | 535520121 | EQW58634.1 (Genbank) | URP |
| Escherichia coli UMEA 3108-1 Taxon ID: 1281183 | 535529880 | EQW68244.1 (Genbank) | URP |
| Escherichia coli UMEA 3113-1 Taxon ID: 1281184 | 535534296 | EQW72599.1 (Genbank) | URP |
| Escherichia coli UMEA 3117-1 Taxon ID: 1281185 | 535535850 | EQW74128.1 (Genbank) | URP |
| Escherichia coli UMEA 3121-1 Taxon ID: 1281186 | 535545090 | EQW83233.1 (Genbank) | URP |
| Escherichia coli UMEA 3122-1 Taxon ID: 1281187 | 535552047 | EQW90040.1 (Genbank) | URP |
| Escherichia coli UMEA 3124-1 Taxon ID: 1281188 | 535553735 | EQW91701.1 (Genbank) | URP |
| Escherichia coli UMEA 3139-1 Taxon ID: 1281189 | 535559641 | EQW97421.1 (Genbank) | URP |
| Escherichia coli UMEA 3140-1 Taxon ID: 1281190 | 535556615 | EQW94467.1 (Genbank) | URP |
| Escherichia coli UMEA 3144-1 Taxon ID: 1281191 | 536020406 | ERB19550.1 (Genbank) | URP |
| Escherichia coli UMEA 3148-1 Taxon ID: 1281192 | 556161435 | ESP49499.1 (Genbank) | URP |
| Escherichia coli UMEA 3150-1 Taxon ID: 1281193 | 536016405 | ERB15598.1 (Genbank) | URP |
| Escherichia coli UMEA 3151-1 Taxon ID: 1281194 | 536024776 | ERB23813.1 (Genbank) | URP |
| Escherichia coli UMEA 3152-1 Taxon ID: 1281195 | 535566105 | EQX03784.1 (Genbank) | URP |
| Escherichia coli UMEA 3155-1 Taxon ID: 1281196 | 535572601 | EQX10161.1 (Genbank) | URP |
| Escherichia coli UMEA 3159-1 Taxon ID: 1281197 | 535576981 | EQX14473.1 (Genbank) | URP |
| Escherichia coli UMEA 3160-1 Taxon ID: 1281198 | 535584900 | EQX22273.1 (Genbank) | URP |
| Escherichia coli UMEA 3161-1 Taxon ID: 1281199 | 535586029 | EQX23396.1 (Genbank) | URP |
| Escherichia coli UMEA 3162-1 Taxon ID: 1281200 | 535594487 | EQX31726.1 (Genbank) | URP |
| Escherichia coli UMEA 3163-1 Taxon ID: 1281201 | 535599702 | EQX36877.1 (Genbank) | URP |
| Escherichia coli UMEA 3172-1 Taxon ID: 1281202 | 535601537 | EQX38682.1 (Genbank) | URP |
| Escherichia coli UMEA 3173-1 Taxon ID: 1281203 | 535611372 | EQX48270.1 (Genbank) | URP |
| Escherichia coli UMEA 3174-1 Taxon ID: 1281204 | 535623100 | EQX59813.1 (Genbank) | URP |
| Escherichia coli UMEA 3175-1 Taxon ID: 1281205 | 535609869 | EQX46795.1 (Genbank) | URP |
| Escherichia coli UMEA 3176-1 Taxon ID: 1281206 | 535622797 | EQX59511.1 (Genbank) | URP |
| Escherichia coli UMEA 3178-1 Taxon ID: 1281207 | 535624792 | EQX61459.1 (Genbank) | URP |
| Escherichia coli UMEA 3180-1 Taxon ID: 1281208 | 535632738 | EQX69243.1 (Genbank) | URP |
| Escherichia coli UMEA 3185-1 Taxon ID: 1281209 | 535633099 | EQX69602.1 (Genbank) | URP |
| Escherichia coli UMEA 3190-1 Taxon ID: 1281210 | 535631837 | EQX68358.1 (Genbank) | URP |
| Escherichia coli UMEA 3193-1 Taxon ID: 1281211 | 535642576 | EQX78878.1 (Genbank) | URP |
| Escherichia coli UMEA 3199-1 Taxon ID: 1281212 | 535651021 | EQX87143.1 (Genbank) | URP |
| Escherichia coli UMEA 3200-1 Taxon ID: 1281213 | 535654321 | EQX90385.1 (Genbank) | URP |
| Escherichia coli UMEA 3201-1 Taxon ID: 1281214 | 535665876 | EQY01693.1 (Genbank) | URP |
| Escherichia coli UMEA 3203-1 Taxon ID: 1281215 | 535668989 | EQY04762.1 (Genbank) | URP |
| Escherichia coli UMEA 3206-1 Taxon ID: 1281216 | 535670676 | EQY06374.1 (Genbank) | URP |
| Escherichia coli UMEA 3208-1 Taxon ID: 1281217 | 535678087 | EQY13608.1 (Genbank) | URP |
| Escherichia coli UMEA 3212-1 Taxon ID: 1281218 | 535697016 | EQY32184.1 (Genbank) | URP |
| Escherichia coli UMEA 3215-1 Taxon ID: 1281219 | 535682798 | EQY18200.1 (Genbank) | URP |
| Escherichia coli UMEA 3216-1 Taxon ID: 1281220 | 535688738 | EQY24052.1 (Genbank) | URP |
| Escherichia coli UMEA 3217-1 Taxon ID: 1281221 | 535692950 | EQY28175.1 (Genbank) | URP |
| Escherichia coli UMEA 3220-1 Taxon ID: 1281222 | 535700157 | EQY35241.1 (Genbank) | URP |
| Escherichia coli UMEA 3221-1 Taxon ID: 1281223 | 535712402 | EQY47252.1 (Genbank) | URP |
| Escherichia coli UMEA 3222-1 Taxon ID: 1281224 | 535713961 | EQY48757.1 (Genbank) | URP |
| Escherichia coli UMEA 3230-1 Taxon ID: 1281225 | 535713149 | EQY47965.1 (Genbank) | URP |
| Escherichia coli UMEA 3233-1 Taxon ID: 1281226 | 535727703 | EQY62280.1 (Genbank) | URP |
| Escherichia coli UMEA 3240-1 Taxon ID: 1281227 | 535733910 | EQY68309.1 (Genbank) | URP |
| Escherichia coli UMEA 3244-1 Taxon ID: 1281228 | 535726150 | EQY60746.1 (Genbank) | URP |
| Escherichia coli UMEA 3257-1 Taxon ID: 1281230 | 535741518 | EQY75730.1 (Genbank) | URP |
| Escherichia coli UMEA 3264-1 Taxon ID: 1281231 | 535737695 | EQY72005.1 (Genbank) | URP |
| Escherichia coli UMEA 3268-1 Taxon ID: 1281232 | 535745625 | EQY79679.1 (Genbank) | URP |
| Escherichia coli UMEA 3271-1 Taxon ID: 1281233 | 536033519 | ERB32385.1 (Genbank) | URP |
| Escherichia coli UMEA 3290-1 Taxon ID: 1281234 | 554715101 | ESK20485.1 (Genbank) | URP |
| Escherichia coli UMEA 3292-1 Taxon ID: 1281235 | 536040352 | ERB39038.1 (Genbank) | URP |
| Escherichia coli UMEA 3298-1 Taxon ID: 1281236 | 536039569 | ERB38285.1 (Genbank) | URP |
| Escherichia coli UMEA 3304-1 Taxon ID: 1281237 | 535741911 | EQY76107.1 (Genbank) | URP |
| Escherichia coli UMEA 3314-1 Taxon ID: 1281238 | 535758939 | EQY92602.1 (Genbank) | URP |
| Escherichia coli UMEA 3318-1 Taxon ID: 1281240 | 535770898 | EQZ04369.1 (Genbank) | URP |
| Escherichia coli UMEA 3323-1 Taxon ID: 1281241 | 554736230 | ESK41372.1 (Genbank) | URP |
| Escherichia coli UMEA 3329-1 Taxon ID: 1281242 | 535771372 | EQZ04820.1 (Genbank) | URP |
| Escherichia coli UMEA 3336-1 Taxon ID: 1281243 | 554703716 | ESK09213.1 (Genbank) | URP |
| Escherichia coli UMEA 3337-1 Taxon ID: 1281244 | 535774070 | EQZ07432.1 (Genbank) | URP |
| Escherichia coli UMEA 3341-1 Taxon ID: 1281245 | 535783478 | EQZ16715.1 (Genbank) | URP |
| Escherichia coli UMEA 3342-1 Taxon ID: 1281246 | 554727194 | ESK32460.1 (Genbank) | URP |
| Escherichia coli UMEA 3355-1 Taxon ID: 1281247 | 535787001 | EQZ20108.1 (Genbank) | URP |
| Escherichia coli UMEA 3391-1 Taxon ID: 1281248 | 535789888 | EQZ23009.1 (Genbank) | URP |
| Escherichia coli UMEA 3426-1 Taxon ID: 1281249 | 554715817 | ESK21199.1 (Genbank) | URP |
| Escherichia coli UMEA 3489-1 Taxon ID: 1281250 | 565521344 | ETF33183.1 (Genbank) | URP |
| Escherichia coli UMEA 3490-1 Taxon ID: 1281251 | 535798363 | EQZ31284.1 (Genbank) | URP |
| Escherichia coli UMEA 3585-1 Taxon ID: 1281252 | 535791780 | EQZ24834.1 (Genbank) | URP |
| Escherichia coli UMEA 3592-1 Taxon ID: 1281253 | 535810553 | EQZ43216.1 (Genbank) | URP |
| Escherichia coli UMEA 3609-1 Taxon ID: 1281254 | 535813417 | EQZ46003.1 (Genbank) | URP |
| Escherichia coli UMEA 3617-1 Taxon ID: 1281255 | 535810953 | EQZ43613.1 (Genbank) | URP |
| Escherichia coli UMEA 3632-1 Taxon ID: 1281256 | 535824148 | EQZ56611.1 (Genbank) | URP |
| Escherichia coli UMEA 3652-1 Taxon ID: 1281257 | 537730418 | ERF50924.1 (Genbank) | URP |
| Escherichia coli UMEA 3656-1 Taxon ID: 1281258 | 535820956 | EQZ53441.1 (Genbank) | URP |
| Escherichia coli UMEA 3662-1 Taxon ID: 1281259 | 535828975 | EQZ61375.1 (Genbank) | URP |
| Escherichia coli UMEA 3671-1 Taxon ID: 1281260 | 535837499 | EQZ69776.1 (Genbank) | URP |
| Escherichia coli UMEA 3682-1 Taxon ID: 1281261 | 535837887 | EQZ70154.1 (Genbank) | URP |
| Escherichia coli UMEA 3687-1 Taxon ID: 1281262 | 535849261 | EQZ81353.1 (Genbank) | URP |
| Escherichia coli UMEA 3693-1 Taxon ID: 1281263 | 554726415 | ESK31692.1 (Genbank) | URP |
| Escherichia coli UMEA 3694-1 Taxon ID: 1281264 | 535849663 | EQZ81754.1 (Genbank) | URP |
| Escherichia coli UMEA 3702-1 Taxon ID: 1281265 | 535850930 | EQZ83012.1 (Genbank) | URP |
| Escherichia coli UMEA 3703-1 Taxon ID: 1281266 | 535865846 | EQZ97589.1 (Genbank) | URP |
| Escherichia coli UMEA 3705-1 Taxon ID: 1281267 | 535852214 | EQZ84261.1 (Genbank) | URP |
| Escherichia coli UMEA 3707-1 Taxon ID: 1281268 | 535861280 | EQZ93148.1 (Genbank) | URP |
| Escherichia coli UMEA 3718-1 Taxon ID: 1281269 | 535878427 | ERA09928.1 (Genbank) | URP |
| Escherichia coli UMEA 3805-1 Taxon ID: 1281270 | 535878264 | ERA09766.1 (Genbank) | URP |
| Escherichia coli UMEA 3821-1 Taxon ID: 1281271 | 535881058 | ERA12525.1 (Genbank) | URP |
| Escherichia coli UMEA 3834-1 Taxon ID: 1281272 | 535895235 | ERA26465.1 (Genbank) | URP |
| Escherichia coli UMEA 3889-1 Taxon ID: 1281273 | 535891109 | ERA22389.1 (Genbank) | URP |
| Escherichia coli UMEA 3893-1 Taxon ID: 1281274 | 535894092 | ERA25340.1 (Genbank) | URP |
| Escherichia coli UMEA 3899-1 Taxon ID: 1281275 | 535897380 | ERA28543.1 (Genbank) | URP |
| Escherichia coli UMEA 3955-1 Taxon ID: 1281276 | 535904130 | ERA35090.1 (Genbank) | URP |
| Escherichia coli UMEA 4075-1 Taxon ID: 1281277 | 535909248 | ERA40096.1 (Genbank) | URP |
| Escherichia coli UMEA 4076-1 Taxon ID: 1281278 | 535920860 | ERA51351.1 (Genbank) | URP |
| Escherichia coli UMEA 4207-1 Taxon ID: 1281279 | 535917900 | ERA48491.1 (Genbank) | URP |
| Escherichia coli UMNF18 Taxon ID: 1050617 | 339413588 | AEJ55260.1 (Genbank) | URP |
| Escherichia coli UMNK88 Taxon ID: 696406 | 332341974 | AEE55308.1 (Genbank) | URP |
| Escherichia coli UTI89 Taxon ID: 364106 | 91071250 | ABE06131.1 (Genbank) | URP |
| Escherichia coli VR50 Taxon ID: 941323 | 801067085 | AKA89582.1 (Genbank) | URP |
| Escherichia coli W Taxon ID: 566546 | 315059883 | ADT74210.1 (Genbank) | URP |
| Escherichia coli W Taxon ID: 566546 | 383404103 | AFH10346.1 (Genbank) | URP |
| Escherichia coli W Taxon ID: 566546 | 306907358 | EFN37863.1 (Genbank) | URP |
| Escherichia coli WV_060327 Taxon ID: 945433 | 320194190 | EFW68822.1 (Genbank) | URP |
| Escherichia coli XH001 Taxon ID: 1069495 | 344192806 | EGV46893.1 (Genbank) | URP |
| Escherichia coli XH140A Taxon ID: 1068608 | 342361833 | EGU25962.1 (Genbank) | URP |
| Escherichia coli Xuzhou21 Taxon ID: 741093 | 386794665 | AFJ27699.1 (Genbank) | URP |
| Escherichia coli p0305293.1 Taxon ID: 1116089 | 476682354 | EMZ87892.1 (Genbank) | URP |
| Escherichia coli p0305293.11 Taxon ID: 1116099 | 477379399 | ENG47158.1 (Genbank) | URP |
| Escherichia coli p0305293.2 Taxon ID: 1116090 | 477392849 | ENG59955.1 (Genbank) | URP |
| Escherichia coli p0305293.4 Taxon ID: 1116092 | 477402324 | ENG68993.1 (Genbank) | URP |
| Escherichia coli p0305293.7 Taxon ID: 1116095 | 477493175 | ENH54320.1 (Genbank) | URP |
| Escherichia coli p0305293.9 Taxon ID: 1116097 | 477413732 | ENG79524.1 (Genbank) | URP |
| Escherichia coli str. 'clone D i14' Taxon ID: 885275 | 355423998 | AER88194.1 (Genbank) | URP |
| Escherichia coli str. 'clone D i2' Taxon ID: 885276 | 355419078 | AER83275.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. DH10B Taxon ID: 316385 | 169888042 | ACB01749.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. DH10B Taxon ID: 316385 | 169888143 | ACB01850.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. MG1655 Taxon ID: 511145 | 1786846 | AAC73729.1 (Genbank) | URP |
| Escherichia coli str. K-12 substr. MG1655 Taxon ID: 511145 | 732685424 | AIZ92229.1 (Genbank) | URP |
| Escherichia sp. 1_1_43 Taxon ID: 457400 | 404292564 | EJZ49382.1 (Genbank) | URP |
| Escherichia sp. 3_2_53FAA Taxon ID: 469598 | 226898610 | EEH84869.1 (Genbank) | URP |
| Escherichia sp. 4_1_40B Taxon ID: 457401 | 386123203 | EIG71802.1 (Genbank) | URP |
| Escherichia coli KTE172 Taxon ID: 1279005 | 508500068 | EOW69138.1 (Genbank) | URP |
| Shigella boydii Sb227 Taxon ID: 300268 | 81244485 | ABB65193.1 (Genbank) | URP |
| Shigella dysenteriae 1617 Taxon ID: 754093 | 558571404 | AHA63542.1 (Genbank) | URP |
| Shigella dysenteriae 1617 Taxon ID: 754093 | 308926052 | EFP71530.1 (Genbank) | URP |
| Shigella dysenteriae Sd197 Taxon ID: 300267 | 81240045 | ABB60755.1 (Genbank) | URP |
| Shigella dysenteriae WRSd3 Taxon ID: 1401327 | 559655748 | ESU78350.1 (Genbank) | URP |
| Shigella dysenteriae WRSd5 Taxon ID: 1401259 | 559660787 | ESU82569.1 (Genbank) | URP |
| Shigella flexneri Taxon ID: 623 | 684114921 | KFZ96613.1 (Genbank) | URP |
| Shigella flexneri Taxon ID: 623 | 721539482 | KGY82838.1 (Genbank) | URP |
| Shigella flexneri 2002017 Taxon ID: 591020 | 281600036 | ADA73020.1 (Genbank) | URP |
| Shigella flexneri 2003036 Taxon ID: 1282357 | 675090661 | AIL34629.1 (Genbank) | URP |
| Shigella flexneri 2747-71 Taxon ID: 766157 | 332761266 | EGJ91552.1 (Genbank) | URP |
| Shigella flexneri 2850-71 Taxon ID: 766158 | 391254342 | EIQ13504.1 (Genbank) | URP |
| Shigella flexneri 2930-71 Taxon ID: 766159 | 332768270 | EGJ98455.1 (Genbank) | URP |
| Shigella flexneri 2a str. 2457T Taxon ID: 198215 | 30040428 | AAP16160.1 (Genbank) | URP |
| Shigella flexneri 2a str. 301 Taxon ID: 198214 | 24050899 | AAN42289.1 (Genbank) | |
| Shigella flexneri 4343-70 Taxon ID: 766160 | 332761002 | EGJ91290.1 (Genbank) | URP |
| Shigella flexneri 6603-63 Taxon ID: 766161 | 397900821 | EJL17177.1 (Genbank) | URP |
| Shigella flexneri G1663 Taxon ID: 1435046 | 828440762 | AKK53244.1 (Genbank) | URP |
| Shigella flexneri J1713 Taxon ID: 754092 | 335576937 | EGM63175.1 (Genbank) | URP |
| Shigella flexneri K-1770 Taxon ID: 766153 | 391258819 | EIQ17906.1 (Genbank) | URP |
| Shigella flexneri K-218 Taxon ID: 766146 | 333008098 | EGK27574.1 (Genbank) | URP |
| Shigella flexneri K-227 Taxon ID: 766147 | 333020872 | EGK40132.1 (Genbank) | URP |
| Shigella flexneri K-272 Taxon ID: 766148 | 333010039 | EGK29474.1 (Genbank) | URP |
| Shigella flexneri K-304 Taxon ID: 766149 | 333021560 | EGK40810.1 (Genbank) | URP |
| Shigella flexneri K-404 Taxon ID: 766151 | 391273527 | EIQ32352.1 (Genbank) | URP |
| Shigella flexneri K-671 Taxon ID: 766152 | 332763373 | EGJ93613.1 (Genbank) | URP |
| Shigella flexneri Shi06HN006 Taxon ID: 1282358 | 675097074 | AIL39546.1 (Genbank) | URP |
| Shigella sonnei Taxon ID: 624 | 721552801 | KGY96139.1 (Genbank) | URP |
| Shigella sonnei 3226-85 Taxon ID: 766162 | 391288378 | EIQ46883.1 (Genbank) | URP |
| Shigella sonnei 3233-85 Taxon ID: 766163 | 391289461 | EIQ47956.1 (Genbank) | URP |
| Shigella sonnei 53G Taxon ID: 216599 | 323164083 | EFZ49891.1 (Genbank) | URP |
| Shigella sonnei Ss046 Taxon ID: 300269 | 73854641 | AAZ87348.1 (Genbank) | URP |
| Shigella sonnei str. Moseley Taxon ID: 754077 | 397903195 | EJL19502.1 (Genbank) | URP |
| Escherichia coli C321.deltaA Taxon ID: 1385755 | 549812147 | AGX32765.1 (Genbank) | URP |
| uncultured bacterium Contig365 Taxon ID: 1393560 | 571052987 | AHF26588.1 (Genbank) | |
| n/a | 915273908 | AKW64267.1 (Genbank) | |
| Escherichia coli 536 Taxon ID: 362663 | 122958495 | URP | |
| Escherichia coli 55989 Taxon ID: 585055 | 254809187 | URP | |
| Escherichia coli APEC O1 Taxon ID: 405955 | 166230416 | URP | |
| Escherichia coli ATCC 8739 Taxon ID: 481805 | 189046601 | URP | |
| Escherichia coli BW2952 Taxon ID: 595496 | 259494931 | URP | |
| Escherichia coli CFT073 Taxon ID: 199310 | 46397471 | URP | |
| Escherichia coli ED1a Taxon ID: 585397 | 254809188 | URP | |
| Escherichia coli HS Taxon ID: 331112 | 166990681 | URP | |
| Escherichia coli IAI1 Taxon ID: 585034 | 226737637 | URP | |
| Escherichia coli IAI39 Taxon ID: 585057 | 226737636 | URP | |
| Escherichia coli K-12 Taxon ID: 83333 | 46397470 | PRP URP | |
| Escherichia coli O127:H6 str. E2348/69 Taxon ID: 574521 | 254809186 | URP | |
| Escherichia coli E24377A Taxon ID: 331111 | 166990680 | URP | |
| Escherichia coli O157:H7 Taxon ID: 83334 | 46397472 | URP | |
| Escherichia coli O157:H7 str. EC4115 Taxon ID: 444450 | 226781790 | URP | |
| Escherichia coli S88 Taxon ID: 585035 | 226737635 | URP | |
| Escherichia coli SE11 Taxon ID: 409438 | 226781791 | URP | |
| Escherichia coli SMS-3-5 Taxon ID: 439855 | 226737639 | URP | |
| Escherichia coli UMN026 Taxon ID: 585056 | 226737638 | URP | |
| Escherichia coli UTI89 Taxon ID: 364106 | 123266135 | URP | |
| Shigella boydii Sb227 Taxon ID: 300268 | 123755643 | URP | |
| Shigella dysenteriae Sd197 Taxon ID: 300267 | 123742174 | URP | |
| Shigella flexneri Taxon ID: 623 | 46397473 | URP | |
| Shigella flexneri 5 str. 8401 Taxon ID: 373384 | 172045859 | URP | |
| Shigella sonnei Ss046 Taxon ID: 300269 | 123773559 | URP | |
| Escherichia coli Taxon ID: 562 | 412968470 | URP | |
| Escherichia coli Taxon ID: 562 | 745370402 | URP | |
| Escherichia coli Taxon ID: 562 | 633875549 | URP | |
| Escherichia coli Taxon ID: 562 | 665443802 | URP | |
| Escherichia coli Taxon ID: 562 | 687671556 | URP | |
| Escherichia coli Taxon ID: 562 | 687675817 | URP | |
| Escherichia coli Taxon ID: 562 | 678293566 | URP | |
| Escherichia coli Taxon ID: 562 | 777223824 | URP | |
| Escherichia coli 55989 Taxon ID: 585055 | 218350800 | URP | |
| Escherichia coli BL21(DE3) Taxon ID: 469008 | 242376404 | URP | |
| Escherichia coli CAG:4 Taxon ID: 1263075 | 524546836 | URP | |
| Escherichia coli D6-113.11 Taxon ID: 1400022 | 633880736 | URP | |
| Escherichia coli D6-113.11 Taxon ID: 1400022 | 665438104 | URP | |
| Escherichia coli D6-117.29 Taxon ID: 1400023 | 633887482 | URP | |
| Escherichia coli DH1 Taxon ID: 536056 | 315135294 | URP | |
| Escherichia coli ED1a Taxon ID: 585397 | 218426015 | URP | |
| Escherichia coli ETEC H10407 Taxon ID: 316401 | 309700866 | URP | |
| Escherichia coli IAI1 Taxon ID: 585034 | 218359938 | URP | |
| Escherichia coli IAI39 Taxon ID: 585057 | 218368986 | URP | |
| Escherichia coli IS1 Taxon ID: 1433131 | 568387313 | URP | |
| Escherichia coli IS29 Taxon ID: 1432549 | 572158840 | URP | |
| Escherichia coli IS35 Taxon ID: 1432551 | 571236470 | URP | |
| Escherichia coli IS5 Taxon ID: 1433129 | 568393094 | URP | |
| Escherichia coli IS9 Taxon ID: 1411645 | 571222714 | URP | |
| Escherichia coli ISC7 Taxon ID: 1432555 | 571184719 | URP | |
| Escherichia coli K-12 Taxon ID: 83333 | 802134205 | PRP URP | |
| Escherichia coli LF82 Taxon ID: 591946 | 222032388 | URP | |
| Escherichia coli Nissle 1917 Taxon ID: 316435 | 441712168 | URP | |
| Escherichia coli O103:H2 str. 12009 Taxon ID: 585395 | 257758001 | URP | |
| Escherichia coli O10:K5(L):H4 str. ATCC 23506 Taxon ID: 1246623 | 441604201 | URP | |
| Escherichia coli O111:H- str. 11128 Taxon ID: 585396 | 257763132 | URP | |
| Escherichia coli O127:H6 str. E2348/69 Taxon ID: 574521 | 215263741 | URP | |
| Escherichia coli O157:H7 str. Sakai Taxon ID: 386585 | 13360124 | ||
| Escherichia coli O26:H11 Taxon ID: 244319 | 740529497 | ||
| Escherichia coli O26:H11 str. 11368 Taxon ID: 573235 | 257752529 | URP | |
| Escherichia coli O5:K4(L):H4 str. ATCC 23502 Taxon ID: 1268238 | 441654532 | URP | |
| Escherichia coli PMV-1 Taxon ID: 1382700 | 544341567 | URP | |
| Escherichia coli S88 Taxon ID: 585035 | 218364336 | URP | |
| Escherichia coli SE11 Taxon ID: 409438 | 209911145 | URP | |
| Escherichia coli SE15 Taxon ID: 431946 | 281177777 | URP | |
| Escherichia coli ST131 Taxon ID: 1359206 | 641683187 | ||
| Escherichia coli UMN026 Taxon ID: 585056 | 218431058 | URP | |
| Escherichia coli c7122 Taxon ID: 475609 | 412961882 | URP | |
| Escherichia coli str. K-12 substr. MC4100 Taxon ID: 1403831 | 557271019 | URP | |
| Escherichia coli str. K-12 substr. MDS42 Taxon ID: 1110693 | 359331345 | URP | |
| Escherichia coli str. K-12 substr. W3110 Taxon ID: 316407 | 1651257 | ||
| Klebsiella pneumoniae IS22 Taxon ID: 1432547 | 571206603 | ||
| Shigella flexneri Taxon ID: 623 | 713635045 | URP | |
| Shigella flexneri 2a Taxon ID: 42897 | 874341251 | URP | |
| Shigella sonnei Taxon ID: 624 | 903697633 | URP | |
| Shigella sonnei Taxon ID: 624 | 904355985 | URP | |
| Shigella sonnei Taxon ID: 624 | 903984100 | URP | |
| Shigella sonnei Taxon ID: 624 | 903632473 | URP | |
| Shigella sonnei Taxon ID: 624 | 903710891 | URP | |
| Shigella sonnei Taxon ID: 624 | 904022586 | URP | |
| Shigella sonnei Taxon ID: 624 | 904738274 | URP | |
| Shigella sonnei Taxon ID: 624 | 903794487 | URP | |
| Shigella sonnei Taxon ID: 624 | 903693565 | URP | |
| Shigella sonnei Taxon ID: 624 | 903702786 | URP | |
| Shigella sonnei Taxon ID: 624 | 903624163 | URP | |
| Shigella sonnei Taxon ID: 624 | 903870448 | URP | |
| Shigella sonnei Taxon ID: 624 | 903831466 | URP | |
| Shigella sonnei Taxon ID: 624 | 903884498 | URP | |
| Shigella sonnei Taxon ID: 624 | 904028540 | URP | |
| Shigella sonnei Taxon ID: 624 | 904165758 | URP | |
| Shigella sonnei Taxon ID: 624 | 903841156 | URP | |
| Shigella sonnei Taxon ID: 624 | 903803572 | URP | |
| Shigella sonnei Taxon ID: 624 | 903763194 | URP | |
| Shigella sonnei Taxon ID: 624 | 905187730 | URP | |
| Shigella sonnei Taxon ID: 624 | 905164112 | URP | |
| Shigella sonnei Taxon ID: 624 | 904175821 | URP | |
| Shigella sonnei Taxon ID: 624 | 903861490 | URP | |
| Shigella sonnei Taxon ID: 624 | 903748647 | URP | |
| Shigella sonnei Taxon ID: 624 | 904370343 | URP | |
| Shigella sonnei Taxon ID: 624 | 905004006 | URP | |
| Shigella sonnei Taxon ID: 624 | 904596489 | URP | |
| Shigella sonnei Taxon ID: 624 | 904219436 | URP | |
| Shigella sonnei Taxon ID: 624 | 904317713 | URP | |
| Shigella sonnei Taxon ID: 624 | 904704595 | URP | |
| Shigella sonnei Taxon ID: 624 | 904287267 | URP | |
| Shigella sonnei Taxon ID: 624 | 904312707 | URP | |
| Shigella sonnei Taxon ID: 624 | 903752990 | URP | |
| Shigella sonnei Taxon ID: 624 | 904185418 | URP | |
| Shigella sonnei Taxon ID: 624 | 904214207 | URP | |
| Shigella sonnei Taxon ID: 624 | 905068446 | URP | |
| Shigella sonnei Taxon ID: 624 | 904453378 | URP | |
| Shigella sonnei Taxon ID: 624 | 904385080 | URP | |
| Shigella sonnei Taxon ID: 624 | 904433946 | URP | |
| Shigella sonnei Taxon ID: 624 | 903837124 | URP | |
| Shigella sonnei Taxon ID: 624 | 904099877 | URP | |
| Shigella sonnei Taxon ID: 624 | 904939460 | URP | |
| Shigella sonnei Taxon ID: 624 | 903845900 | URP | |
| Shigella sonnei Taxon ID: 624 | 903913724 | URP | |
| Shigella sonnei Taxon ID: 624 | 903614492 | URP | |
| Shigella sonnei Taxon ID: 624 | 903743869 | URP | |
| Shigella sonnei Taxon ID: 624 | 903927432 | URP | |
| Shigella sonnei Taxon ID: 624 | 903600790 | URP | |
| Shigella sonnei Taxon ID: 624 | 905839806 | URP | |
| Shigella sonnei Taxon ID: 624 | 905073417 | URP | |
| Shigella sonnei Taxon ID: 624 | 904322404 | URP | |
| Shigella sonnei Taxon ID: 624 | 905936978 | URP | |
| Shigella sonnei Taxon ID: 624 | 905931817 | URP | |
| Shigella sonnei Taxon ID: 624 | 905890844 | URP | |
| Shigella sonnei Taxon ID: 624 | 905819341 | URP | |
| Shigella sonnei Taxon ID: 624 | 905232554 | URP | |
| Shigella sonnei Taxon ID: 624 | 904502083 | URP | |
| Shigella sonnei Taxon ID: 624 | 904658937 | URP | |
| Shigella sonnei Taxon ID: 624 | 905346940 | URP | |
| Shigella sonnei Taxon ID: 624 | 905829456 | URP | |
| Shigella sonnei Taxon ID: 624 | 905926791 | URP | |
| Shigella sonnei Taxon ID: 624 | 905788952 | URP | |
| Shigella sonnei Taxon ID: 624 | 905172758 | URP | |
| Shigella sonnei Taxon ID: 624 | 905692519 | URP | |
| Shigella sonnei Taxon ID: 624 | 905596987 | URP | |
| Shigella sonnei Taxon ID: 624 | 905814224 | URP | |
| Shigella sonnei Taxon ID: 624 | 906009185 | URP | |
| Shigella sonnei Taxon ID: 624 | 906175278 | URP | |
| Shigella sonnei Taxon ID: 624 | 906201488 | URP | |
| Shigella sonnei Taxon ID: 624 | 905993255 | URP | |
| Shigella sonnei Taxon ID: 624 | 904959035 | URP | |
| Shigella sonnei Taxon ID: 624 | 906133533 | URP | |
| Shigella sonnei Taxon ID: 624 | 905431626 | URP | |
| Shigella sonnei Taxon ID: 624 | 906076416 | URP | |
| Shigella sonnei Taxon ID: 624 | 905466634 | URP | |
| Shigella sonnei Taxon ID: 624 | 905906178 | URP | |
| Shigella sonnei Taxon ID: 624 | 906055390 | URP | |
| Shigella sonnei Taxon ID: 624 | 905421698 | URP | |
| Shigella sonnei Taxon ID: 624 | 905973014 | URP | |
| Shigella sonnei Taxon ID: 624 | 905586995 | URP | |
| Shigella sonnei Taxon ID: 624 | 905491653 | URP | |
| Shigella sonnei Taxon ID: 624 | 905546746 | URP | |
| Shigella sonnei Taxon ID: 624 | 905277249 | URP | |
| Shigella sonnei Taxon ID: 624 | 904802781 | URP | |
| Shigella sonnei Taxon ID: 624 | 905978165 | URP | |
| Shigella sonnei Taxon ID: 624 | 905441743 | URP | |
| Shigella sonnei Taxon ID: 624 | 905901372 | URP | |
| Shigella sonnei Taxon ID: 624 | 906206858 | URP | |
| Shigella sonnei Taxon ID: 624 | 905576988 | URP | |
| Shigella sonnei Taxon ID: 624 | 905038518 | URP | |
| Shigella sonnei Taxon ID: 624 | 905237479 | URP | |
| Shigella sonnei Taxon ID: 624 | 904865433 | URP | |
| Shigella sonnei Taxon ID: 624 | 904644260 | URP | |
| Shigella sonnei Taxon ID: 624 | 905824520 | URP | |
| Shigella sonnei Taxon ID: 624 | 905192690 | URP | |
| Shigella sonnei Taxon ID: 624 | 904821179 | URP | |
| Shigella sonnei Taxon ID: 624 | 904345998 | URP | |
| Shigella sonnei Taxon ID: 624 | 905633564 | URP | |
| Shigella sonnei Taxon ID: 624 | 904531761 | URP | |
| Shigella sonnei Taxon ID: 624 | 905763889 | URP | |
| Shigella sonnei Taxon ID: 624 | 904911020 | URP | |
| Shigella sonnei Taxon ID: 624 | 905618344 | URP | |
| Shigella sonnei Taxon ID: 624 | 904994050 | URP | |
| Shigella sonnei Taxon ID: 624 | 906145061 | URP | |
| Shigella sonnei Taxon ID: 624 | 905218042 | URP | |
| Shigella sonnei Taxon ID: 624 | 905416616 | URP | |
| Shigella sonnei Taxon ID: 624 | 905592207 | URP | |
| Shigella sonnei Taxon ID: 624 | 904109155 | URP | |
| Shigella sonnei Taxon ID: 624 | 904609866 | URP | |
| Shigella sonnei Taxon ID: 624 | 905865476 | URP | |
| Shigella sonnei Taxon ID: 624 | 904999567 | URP | |
| Shigella sonnei Taxon ID: 624 | 904079523 | URP | |
| Shigella sonnei Taxon ID: 624 | 905875637 | URP | |
| Shigella sonnei Taxon ID: 624 | 904772349 | URP | |
| Shigella sonnei Taxon ID: 624 | 904477865 | URP | |
| Shigella sonnei Taxon ID: 624 | 906123280 | URP | |
| Shigella sonnei Taxon ID: 624 | 906249960 | URP | |
| Shigella sonnei Taxon ID: 624 | 905048478 | URP | |
| Shigella sonnei Taxon ID: 624 | 906128439 | URP | |
| Shigella sonnei Taxon ID: 624 | 904915893 | URP | |
| Shigella sonnei Taxon ID: 624 | 904122591 | URP | |
| Shigella sonnei Taxon ID: 624 | 904816294 | URP | |
| Shigella sonnei Taxon ID: 624 | 904786820 | URP | |
| Shigella sonnei Taxon ID: 624 | 905506211 | URP | |
| Shigella sonnei Taxon ID: 624 | 905044639 | URP | |
| Shigella sonnei Taxon ID: 624 | 905622406 | URP | |
| Shigella sonnei Taxon ID: 624 | 905009015 | URP | |
| Shigella sonnei Taxon ID: 624 | 905845877 | URP | |
| Shigella sonnei Taxon ID: 624 | 906243682 | URP | |
| Shigella sonnei Taxon ID: 624 | 904117778 | URP | |
| Shigella sonnei Taxon ID: 624 | 906275389 | URP | |
| Shigella sonnei Taxon ID: 624 | 905501564 | URP | |
| Shigella sonnei Taxon ID: 624 | 904855776 | URP | |
| Shigella sonnei Taxon ID: 624 | 906264794 | URP | |
| Shigella sonnei Taxon ID: 624 | 904614865 | URP | |
| Shigella sonnei Taxon ID: 624 | 906270186 | URP | |
| Shigella sonnei Taxon ID: 624 | 906254312 | URP | |
| Shigella sonnei Taxon ID: 624 | 904880308 | URP | |
| Shigella sonnei Taxon ID: 624 | 904896130 | URP | |
| Shigella sonnei Taxon ID: 624 | 904732916 | URP | |
| Shigella sonnei Taxon ID: 624 | 904668771 | URP | |
| Shigella sonnei Taxon ID: 624 | 904791705 | URP | |
| Shigella sonnei Taxon ID: 624 | 904008197 | URP | |
| Shigella sonnei Taxon ID: 624 | 904875407 | URP | |
| Shigella sonnei Taxon ID: 624 | 904826225 | URP | |
| Shigella sonnei Taxon ID: 624 | 904969224 | URP | |
| Shigella sonnei Taxon ID: 624 | 904718916 | URP | |
| Shigella sonnei Taxon ID: 624 | 904683383 | URP | |
| Shigella sonnei Taxon ID: 624 | 904885170 | URP | |
| Shigella sonnei Taxon ID: 624 | 904565905 | URP | |
| Shigella sonnei Taxon ID: 624 | 904629548 | URP | |
| Shigella sonnei Taxon ID: 624 | 904404526 | URP | |
| Shigella sonnei Taxon ID: 624 | 904233791 | URP | |
| Shigella sonnei Taxon ID: 624 | 904209346 | URP | |
| Shigella sonnei Taxon ID: 624 | 904974081 | URP | |
| Shigella sonnei Taxon ID: 624 | 904482659 | URP | |
| Shigella sonnei Taxon ID: 624 | 904929557 | URP | |
| Shigella sonnei Taxon ID: 624 | 905612059 | URP | |
| Shigella sonnei Taxon ID: 624 | 904170853 | URP | |
| Shigella sonnei Taxon ID: 624 | 904521824 | URP | |
| Shigella sonnei Taxon ID: 624 | 905672648 | URP | |
| Shigella sonnei Taxon ID: 624 | 905207648 | URP | |
| Shigella sonnei Taxon ID: 624 | 906061608 | URP | |
| Shigella sonnei Taxon ID: 624 | 905197730 | URP | |
| Shigella sonnei Taxon ID: 624 | 906014358 | URP | |
| Shigella sonnei Taxon ID: 624 | 904752560 | URP | |
| Shigella sonnei Taxon ID: 624 | 905739200 | URP | |
| Shigella sonnei Taxon ID: 624 | 904458179 | URP | |
| Shigella sonnei Taxon ID: 624 | 905337091 | URP | |
| Shigella sonnei Taxon ID: 624 | 906138945 | URP | |
| Shigella sonnei Taxon ID: 624 | 905272417 | URP | |
| Shigella sonnei Taxon ID: 624 | 906045294 | URP | |
| Shigella sonnei Taxon ID: 624 | 904341206 | URP | |
| Shigella sonnei Taxon ID: 624 | 906154656 | URP | |
| Shigella sonnei Taxon ID: 624 | 905582202 | URP | |
| Shigella sonnei Taxon ID: 624 | 904845822 | URP | |
| Shigella sonnei Taxon ID: 624 | 906024638 | URP | |
| Shigella sonnei Taxon ID: 624 | 906097218 | URP | |
| Shigella sonnei Taxon ID: 624 | 906118055 | URP | |
| Shigella sonnei Taxon ID: 624 | 905147873 | URP | |
| Shigella sonnei Taxon ID: 624 | 903956902 | URP | |
| Shigella sonnei Taxon ID: 624 | 905247433 | URP | |
| Shigella sonnei Taxon ID: 624 | 905482437 | URP | |
| Shigella sonnei Taxon ID: 624 | 906040186 | URP | |
| Shigella sonnei Taxon ID: 624 | 906071335 | URP | |
| Shigella sonnei Taxon ID: 624 | 905860346 | URP | |
| Shigella sonnei Taxon ID: 624 | 905607231 | URP | |
| Shigella sonnei Taxon ID: 624 | 906035088 | URP | |
| Shigella sonnei Taxon ID: 624 | 905083400 | URP | |
| Shigella sonnei Taxon ID: 624 | 905637553 | URP | |
| Shigella sonnei Taxon ID: 624 | 904160935 | URP | |
| Shigella sonnei Taxon ID: 624 | 906086730 | URP | |
| Shigella sonnei Taxon ID: 624 | 905983473 | URP | |
| Shigella sonnei Taxon ID: 624 | 903970009 | URP | |
| Shigella sonnei Taxon ID: 624 | 905999632 | URP | |
| Shigella sonnei Taxon ID: 624 | 904747585 | URP | |
| Shigella sonnei Taxon ID: 624 | 904092801 | URP | |
| Shigella sonnei Taxon ID: 624 | 904326675 | URP | |
| Shigella sonnei Taxon ID: 624 | 904473013 | URP | |
| Shigella sonnei Taxon ID: 624 | 904031786 | URP | |
| Shigella sonnei Taxon ID: 624 | 905255700 | URP | |
| Shigella sonnei Taxon ID: 624 | 904050733 | URP | |
| Shigella sonnei Taxon ID: 624 | 903932191 | URP | |
| Shigella sonnei Taxon ID: 624 | 903720050 | URP | |
| Shigella sonnei Taxon ID: 624 | 903706345 | URP | |
| Shigella sonnei Taxon ID: 624 | 903654787 | URP | |
| Shigella sonnei Taxon ID: 624 | 903989188 | URP | |
| Shigella sonnei Taxon ID: 624 | 904228876 | URP | |
| Shigella sonnei Taxon ID: 624 | 903730759 | URP | |
| Shigella sonnei Taxon ID: 624 | 904649005 | URP | |
| Shigella sonnei Taxon ID: 624 | 904003314 | URP | |
| Shigella sonnei Taxon ID: 624 | 903757770 | URP | |
| Shigella sonnei Taxon ID: 624 | 904590339 | URP | |
| Shigella sonnei Taxon ID: 624 | 905396977 | URP | |
| Shigella sonnei Taxon ID: 624 | 903678795 | URP | |
| Shigella sonnei Taxon ID: 624 | 903965296 | URP | |
| Shigella sonnei Taxon ID: 624 | 903993932 | URP | |
| Shigella sonnei Taxon ID: 624 | 904292221 | URP | |
| Shigella sonnei Taxon ID: 624 | 903899000 | URP | |
| Shigella sonnei Taxon ID: 624 | 904036833 | URP | |
| Shigella sonnei Taxon ID: 624 | 904253007 | URP | |
| Shigella sonnei Taxon ID: 624 | 905322085 | URP | |
| Shigella sonnei Taxon ID: 624 | 903865950 | URP | |
| Shigella sonnei Taxon ID: 624 | 904070205 | URP | |
| Shigella sonnei Taxon ID: 624 | 903908077 | URP | |
| Shigella sonnei Taxon ID: 624 | 904156084 | URP | |
| Shigella sonnei Taxon ID: 624 | 904046586 | URP | |
| Shigella sonnei Taxon ID: 624 | 904074812 | URP | |
| Shigella sonnei Taxon ID: 624 | 904238314 | URP | |
| Shigella sonnei Taxon ID: 624 | 903974886 | URP | |
| Shigella sonnei Taxon ID: 624 | 904336296 | URP | |
| Shigella sonnei Taxon ID: 624 | 903780456 | URP | |
| Shigella sonnei Taxon ID: 624 | 904056091 | URP | |
| Shigella sonnei Taxon ID: 624 | 905312133 | URP | |
| Shigella sonnei Taxon ID: 624 | 903998831 | URP | |
| Shigella sonnei Taxon ID: 624 | 903738863 | URP | |
| Shigella sonnei Taxon ID: 624 | 903979553 | URP | |
| Shigella sonnei Taxon ID: 624 | 903775587 | URP | |
| Shigella sonnei Taxon ID: 624 | 903922784 | URP | |
| Shigella sonnei Taxon ID: 624 | 903669614 | URP | |
| Shigella sonnei Taxon ID: 624 | 904041524 | URP | |
| Shigella sonnei Taxon ID: 624 | 903610033 | URP | |
| Shigella sonnei Taxon ID: 624 | 903889096 | URP | |
| Shigella sonnei Taxon ID: 624 | 904089214 | URP | |
| Shigella sonnei Taxon ID: 624 | 904331269 | URP | |
| Shigella sonnei Taxon ID: 624 | 903937177 | URP | |
| Shigella sonnei Taxon ID: 624 | 904112720 | URP | |
| Shigella sonnei Taxon ID: 624 | 904264409 | URP | |
| Shigella sonnei Taxon ID: 624 | 903531962 | URP | |
| Shigella sonnei Taxon ID: 624 | 903721549 | URP | |
| Shigella sonnei Taxon ID: 624 | 903714057 | URP | |
| Shigella sonnei Taxon ID: 624 | 904103566 | URP | |
| Shigella sonnei Taxon ID: 624 | 904141499 | URP | |
| Shigella sonnei Taxon ID: 624 | 903798527 | URP | |
| Shigella sonnei Taxon ID: 624 | 904248034 | URP | |
| Shigella sonnei Taxon ID: 624 | 904721291 | URP | |
| Shigella sonnei Taxon ID: 624 | 903826369 | URP | |
| Shigella sonnei Taxon ID: 624 | 903644157 | URP | |
| Shigella sonnei Taxon ID: 624 | 904064006 | URP | |
| Shigella sonnei Taxon ID: 624 | 903618741 | URP | |
| Shigella sonnei Taxon ID: 624 | 903581721 | URP | |
| Shigella sonnei Taxon ID: 624 | 903647187 | URP | |
| Shigella sonnei Taxon ID: 624 | 903892876 | URP | |
| Shigella sonnei Taxon ID: 624 | 903783986 | URP | |
| Shigella sonnei Taxon ID: 624 | 904782339 | URP | |
| Shigella sonnei Taxon ID: 624 | 903585830 | URP | |
| Shigella sonnei Taxon ID: 624 | 903960544 | URP | |
| Shigella sonnei Taxon ID: 624 | 904388552 | URP | |
| Shigella sonnei Taxon ID: 624 | 903766998 | URP | |
| Shigella sonnei Taxon ID: 624 | 903683305 | URP | |
| Shigella sonnei Taxon ID: 624 | 903789809 | URP | |
| Shigella sonnei Taxon ID: 624 | 903916659 | URP | |
| Shigella sonnei Taxon ID: 624 | 903822012 | URP | |
| Shigella sonnei Taxon ID: 624 | 905411327 | URP | |
| Shigella sonnei Taxon ID: 624 | 905088475 | URP | |
| Shigella sonnei Taxon ID: 624 | 903687988 | URP | |
| Shigella sonnei Taxon ID: 624 | 904560746 | URP | |
| Shigella sonnei Taxon ID: 624 | 905401351 | URP | |
| Shigella sonnei Taxon ID: 624 | 903946347 | URP | |
| Shigella sonnei Taxon ID: 624 | 903817344 | URP | |
| Shigella sonnei Taxon ID: 624 | 903674327 | URP | |
| Shigella sonnei Taxon ID: 624 | 904443664 | URP | |
| Shigella sonnei Taxon ID: 624 | 903808261 | URP | |
| Shigella sonnei Taxon ID: 624 | 904302151 | URP | |
| Shigella sonnei Taxon ID: 624 | 904127494 | URP | |
| Shigella sonnei Taxon ID: 624 | 904964285 | URP | |
| Shigella sonnei Taxon ID: 624 | 903729878 | URP | |
| Shigella sonnei Taxon ID: 624 | 904841048 | URP | |
| Shigella sonnei Taxon ID: 624 | 904296942 | URP | |
| Shigella sonnei Taxon ID: 624 | 904409563 | URP | |
| Shigella sonnei Taxon ID: 624 | 904017844 | URP | |
| Shigella sonnei Taxon ID: 624 | 904954333 | URP | |
| Shigella sonnei Taxon ID: 624 | 903951729 | URP | |
| Shigella sonnei Taxon ID: 624 | 903660083 | URP | |
| Shigella sonnei Taxon ID: 624 | 904742912 | URP | |
| Shigella sonnei Taxon ID: 624 | 904575729 | URP | |
| Shigella sonnei Taxon ID: 624 | 904438717 | URP | |
| Shigella sonnei Taxon ID: 624 | 904546449 | URP | |
| Shigella sonnei Taxon ID: 624 | 904272861 | URP | |
| Shigella sonnei Taxon ID: 624 | 904136992 | URP | |
| Shigella sonnei Taxon ID: 624 | 906019298 | URP | |
| Shigella sonnei Taxon ID: 624 | 904468208 | URP | |
| Shigella sonnei Taxon ID: 624 | 905561982 | URP | |
| Shigella sonnei Taxon ID: 624 | 903879923 | URP | |
| Shigella sonnei Taxon ID: 624 | 905551987 | URP | |
| Shigella sonnei Taxon ID: 624 | 904847376 | URP | |
| Shigella sonnei Taxon ID: 624 | 904757452 | URP | |
| Shigella sonnei Taxon ID: 624 | 904448354 | URP | |
| Shigella sonnei Taxon ID: 624 | 903938312 | URP | |
| Shigella sonnei Taxon ID: 624 | 905222672 | URP | |
| Shigella sonnei Taxon ID: 624 | 905262390 | URP | |
| Shigella sonnei Taxon ID: 624 | 904890066 | URP | |
| Shigella sonnei Taxon ID: 624 | 904673215 | URP | |
| Shigella sonnei Taxon ID: 624 | 905988645 | URP | |
| Shigella sonnei Taxon ID: 624 | 904619751 | URP | |
| Shigella sonnei Taxon ID: 624 | 905728253 | URP | |
| Shigella sonnei Taxon ID: 624 | 906081637 | URP | |
| Shigella sonnei Taxon ID: 624 | 904132180 | URP | |
| Shigella sonnei Taxon ID: 624 | 904199954 | URP | |
| Shigella sonnei Taxon ID: 624 | 904277511 | URP | |
| Shigella sonnei Taxon ID: 624 | 904243366 | URP | |
| Shigella sonnei Taxon ID: 624 | 903856172 | URP | |
| Shigella sonnei Taxon ID: 624 | 904146438 | URP | |
| Shigella sonnei Taxon ID: 624 | 904536649 | URP | |
| Shigella sonnei Taxon ID: 624 | 904419165 | URP | |
| Shigella sonnei Taxon ID: 624 | 904195112 | URP | |
| Shigella sonnei Taxon ID: 624 | 904084382 | URP | |
| Shigella sonnei Taxon ID: 624 | 905733287 | URP | |
| Shigella sonnei Taxon ID: 624 | 904012844 | URP | |
| Shigella sonnei Taxon ID: 624 | 903903524 | URP | |
| Shigella sonnei Taxon ID: 624 | 904463278 | URP | |
| Shigella sonnei Taxon ID: 624 | 904497364 | URP | |
| Shigella sonnei Taxon ID: 624 | 904979137 | URP | |
| Shigella sonnei Taxon ID: 624 | 905372071 | URP | |
| Shigella sonnei Taxon ID: 624 | 905182572 | URP | |
| Shigella sonnei Taxon ID: 624 | 905774116 | URP | |
| Shigella sonnei Taxon ID: 624 | 904282070 | URP | |
| Shigella sonnei Taxon ID: 624 | 904870391 | URP | |
| Shigella sonnei Taxon ID: 624 | 904904887 | URP | |
| Shigella sonnei Taxon ID: 624 | 904060516 | URP | |
| Shigella sonnei Taxon ID: 624 | 904257751 | URP | |
| Shigella sonnei Taxon ID: 624 | 904634507 | URP | |
| Shigella sonnei Taxon ID: 624 | 905794177 | URP | |
| Shigella sonnei Taxon ID: 624 | 904708312 | URP | |
| Shigella sonnei Taxon ID: 624 | 904836018 | URP | |
| Shigella sonnei Taxon ID: 624 | 904860696 | URP | |
| Shigella sonnei Taxon ID: 624 | 904556025 | URP | |
| Shigella sonnei Taxon ID: 624 | 905496694 | URP | |
| Shigella sonnei Taxon ID: 624 | 905748335 | URP | |
| Shigella sonnei Taxon ID: 624 | 905881103 | URP | |
| Shigella sonnei Taxon ID: 624 | 905381866 | URP | |
| Shigella sonnei Taxon ID: 624 | 905708055 | URP | |
| Shigella sonnei Taxon ID: 624 | 905896578 | URP | |
| Shigella sonnei Taxon ID: 624 | 905013816 | URP | |
| Shigella sonnei Taxon ID: 624 | 905753372 | URP | |
| Shigella sonnei Taxon ID: 624 | 905834907 | URP | |
| Shigella sonnei Taxon ID: 624 | 905531774 | URP | |
| Shigella sonnei Taxon ID: 624 | 905516721 | URP | |
| Shigella sonnei Taxon ID: 624 | 904423780 | URP | |
| Shigella sonnei Taxon ID: 624 | 904492326 | URP | |
| Shigella sonnei Taxon ID: 624 | 904693341 | URP | |
| Shigella sonnei Taxon ID: 624 | 905282359 | URP | |
| Shigella sonnei Taxon ID: 624 | 905107896 | URP | |
| Shigella sonnei Taxon ID: 624 | 904949268 | URP | |
| Shigella sonnei Taxon ID: 624 | 905242515 | URP | |
| Shigella sonnei Taxon ID: 624 | 905252488 | URP | |
| Shigella sonnei Taxon ID: 624 | 905366972 | URP | |
| Shigella sonnei Taxon ID: 624 | 904190141 | URP | |
| Shigella sonnei Taxon ID: 624 | 905911547 | URP | |
| Shigella sonnei Taxon ID: 624 | 904811313 | URP | |
| Shigella sonnei Taxon ID: 624 | 904517057 | URP | |
| Shigella sonnei Taxon ID: 624 | 905886236 | URP | |
| Shigella sonnei Taxon ID: 624 | 904624624 | URP | |
| Shigella sonnei Taxon ID: 624 | 904360580 | URP | |
| Shigella sonnei Taxon ID: 624 | 904762408 | URP | |
| Shigella sonnei Taxon ID: 624 | 905078358 | URP | |
| Shigella sonnei Taxon ID: 624 | 904605163 | URP | |
| Shigella sonnei Taxon ID: 624 | 904507127 | URP | |
| Shigella sonnei Taxon ID: 624 | 904512724 | URP | |
| Shigella sonnei Taxon ID: 624 | 904984006 | URP | |
| Shigella sonnei Taxon ID: 624 | 904924540 | URP | |
| Shigella sonnei Taxon ID: 624 | 905028558 | URP | |
| Shigella sonnei Taxon ID: 624 | 905212388 | URP | |
| Shigella sonnei Taxon ID: 624 | 904767068 | URP | |
| Shigella sonnei Taxon ID: 624 | 905267361 | URP | |
| Shigella sonnei Taxon ID: 624 | 904306562 | URP | |
| Shigella sonnei Taxon ID: 624 | 904224262 | URP | |
| Shigella sonnei Taxon ID: 624 | 905391862 | URP | |
| Shigella sonnei Taxon ID: 624 | 905132930 | URP | |
| Shigella sonnei Taxon ID: 624 | 904204202 | URP | |
| Shigella sonnei Taxon ID: 624 | 904688268 | URP | |
| Shigella sonnei Taxon ID: 624 | 905142798 | URP | |
| Shigella sonnei Taxon ID: 624 | 905564860 | URP | |
| Shigella sonnei Taxon ID: 624 | 905018800 | URP | |
| Shigella sonnei Taxon ID: 624 | 904570779 | URP | |
| Shigella sonnei Taxon ID: 624 | 904151084 | URP | |
| Shigella sonnei Taxon ID: 624 | 904399771 | URP | |
| Shigella sonnei Taxon ID: 624 | 904350861 | URP | |
| Shigella sonnei Taxon ID: 624 | 905342091 | URP | |
| Shigella sonnei Taxon ID: 624 | 904653935 | URP | |
| Shigella sonnei Taxon ID: 624 | 904526744 | URP | |
| Shigella sonnei Taxon ID: 624 | 905916597 | URP | |
| Shigella sonnei Taxon ID: 624 | 904541477 | URP | |
| Shigella sonnei Taxon ID: 624 | 905376974 | URP | |
| Shigella sonnei Taxon ID: 624 | 905436747 | URP | |
| Shigella sonnei Taxon ID: 624 | 905471514 | URP | |
| Shigella sonnei Taxon ID: 624 | 905809577 | URP | |
| Shigella sonnei Taxon ID: 624 | 904600082 | URP | |
| Shigella sonnei Taxon ID: 624 | 905541791 | URP | |
| Shigella sonnei Taxon ID: 624 | 904727198 | URP | |
| Shigella sonnei Taxon ID: 624 | 905406813 | URP | |
| Shigella sonnei Taxon ID: 624 | 905702910 | URP | |
| Shigella sonnei Taxon ID: 624 | 905476592 | URP | |
| Shigella sonnei Taxon ID: 624 | 904380038 | URP | |
| Shigella sonnei Taxon ID: 624 | 904919711 | URP | |
| Shigella sonnei Taxon ID: 624 | 905446619 | URP | |
| Shigella sonnei Taxon ID: 624 | 904899823 | URP | |
| Shigella sonnei Taxon ID: 624 | 905302304 | URP | |
| Shigella sonnei Taxon ID: 624 | 904934368 | URP | |
| Shigella sonnei Taxon ID: 624 | 905033704 | URP | |
| Shigella sonnei Taxon ID: 624 | 905921739 | URP | |
| Shigella sonnei Taxon ID: 624 | 904713046 | URP | |
| Shigella sonnei Taxon ID: 624 | 905332153 | URP | |
| Shigella sonnei Taxon ID: 624 | 905536825 | URP | |
| Shigella sonnei Taxon ID: 624 | 905287506 | URP | |
| Shigella sonnei Taxon ID: 624 | 905677651 | URP | |
| Shigella sonnei Taxon ID: 624 | 905063900 | URP | |
| Shigella sonnei Taxon ID: 624 | 904663756 | URP | |
| Shigella sonnei Taxon ID: 624 | 905023834 | URP | |
| Shigella sonnei Taxon ID: 624 | 905357095 | URP | |
| Shigella sonnei Taxon ID: 624 | 905157731 | URP | |
| Shigella sonnei Taxon ID: 624 | 905804471 | URP | |
| Shigella sonnei Taxon ID: 624 | 904777117 | URP | |
| Shigella sonnei Taxon ID: 624 | 905768950 | URP | |
| Shigella sonnei Taxon ID: 624 | 904580605 | URP | |
| Shigella sonnei Taxon ID: 624 | 905292302 | URP | |
| Shigella sonnei Taxon ID: 624 | 905137785 | URP | |
| Shigella sonnei Taxon ID: 624 | 906004069 | URP | |
| Shigella sonnei Taxon ID: 624 | 905687733 | URP | |
| Shigella sonnei Taxon ID: 624 | 906280565 | URP | |
| Shigella sonnei Taxon ID: 624 | 904796551 | URP | |
| Shigella sonnei Taxon ID: 624 | 905870472 | URP | |
| Shigella sonnei Taxon ID: 624 | 905297260 | URP | |
| Shigella sonnei Taxon ID: 624 | 905451738 | URP | |
| Shigella sonnei Taxon ID: 624 | 905053465 | URP | |
| Shigella sonnei Taxon ID: 624 | 905227633 | URP | |
| Shigella sonnei Taxon ID: 624 | 904487644 | URP | |
| Shigella sonnei Taxon ID: 624 | 905118016 | URP | |
| Shigella sonnei Taxon ID: 624 | 905849948 | URP | |
| Shigella sonnei Taxon ID: 624 | 904549473 | URP | |
| Shigella sonnei Taxon ID: 624 | 905757629 | URP | |
| Shigella sonnei Taxon ID: 624 | 904806828 | URP | |
| Shigella sonnei Taxon ID: 624 | 903875282 | URP | |
| Shigella sonnei Taxon ID: 624 | 905784076 | URP | |
| Shigella sonnei Taxon ID: 624 | 905521836 | URP | |
| Shigella sonnei Taxon ID: 624 | 905461709 | URP | |
| Shigella sonnei Taxon ID: 624 | 905167750 | URP | |
| Shigella sonnei Taxon ID: 624 | 905307167 | URP | |
| Shigella sonnei Taxon ID: 624 | 905327180 | URP | |
| Shigella sonnei Taxon ID: 624 | 905627304 | URP | |
| Shigella sonnei Taxon ID: 624 | 905712982 | URP | |
| Shigella sonnei Taxon ID: 624 | 904585630 | URP | |
| Shigella sonnei Taxon ID: 624 | 905056644 | URP | |
| Shigella sonnei Taxon ID: 624 | 905177846 | URP | |
| Shigella sonnei Taxon ID: 624 | 905152665 | URP | |
| Shigella sonnei Taxon ID: 624 | 905682738 | URP | |
| Shigella sonnei Taxon ID: 624 | 904697950 | URP | |
| Shigella sonnei Taxon ID: 624 | 904678689 | URP | |
| Shigella sonnei Taxon ID: 624 | 905112950 | URP | |
| Shigella sonnei Taxon ID: 624 | 905952430 | URP | |
| Shigella sonnei Taxon ID: 624 | 905386943 | URP | |
| Shigella sonnei Taxon ID: 624 | 905602097 | URP | |
| Shigella sonnei Taxon ID: 624 | 904394755 | URP | |
| Shigella sonnei Taxon ID: 624 | 906091838 | URP | |
| Shigella sonnei Taxon ID: 624 | 906112741 | URP | |
| Shigella sonnei Taxon ID: 624 | 905968024 | URP | |
| Shigella sonnei Taxon ID: 624 | 906212016 | URP | |
| Shigella sonnei Taxon ID: 624 | 905962835 | URP | |
| Shigella sonnei Taxon ID: 624 | 906227761 | URP | |
| Shigella sonnei Taxon ID: 624 | 906029765 | URP | |
| Shigella sonnei Taxon ID: 624 | 906180483 | URP | |
| Shigella sonnei Taxon ID: 624 | 906238284 | URP | |
| Shigella sonnei Taxon ID: 624 | 906191129 | URP | |
| Shigella sonnei Taxon ID: 624 | 906164781 | URP | |
| Shigella sonnei Taxon ID: 624 | 906102371 | URP | |
| Shigella sonnei Taxon ID: 624 | 906217368 | URP | |
| Shigella sonnei Taxon ID: 624 | 904944377 | URP | |
| Shigella sonnei Taxon ID: 624 | 905957510 | URP | |
| Shigella sonnei Taxon ID: 624 | 906170073 | URP | |
| Shigella sonnei Taxon ID: 624 | 905947431 | URP | |
| Shigella sonnei Taxon ID: 624 | 905942267 | URP | |
| Shigella sonnei Taxon ID: 624 | 906222529 | URP | |
| Shigella sonnei Taxon ID: 624 | 906065967 | URP | |
| Shigella sonnei Taxon ID: 624 | 906050573 | URP | |
| Shigella sonnei Taxon ID: 624 | 904831060 | URP | |
| Shigella sonnei Taxon ID: 624 | 906185857 | URP | |
| Shigella sonnei Taxon ID: 624 | 905558017 | URP | |
| Shigella sonnei Taxon ID: 624 | 906107559 | URP | |
| Shigella sonnei Taxon ID: 624 | 906149289 | URP | |
| Shigella sonnei Taxon ID: 624 | 905663317 | URP | |
| Shigella sonnei Taxon ID: 624 | 905122930 | URP | |
| Shigella sonnei Taxon ID: 624 | 905526897 | URP | |
| Shigella sonnei Taxon ID: 624 | 905647416 | URP | |
| Shigella sonnei Taxon ID: 624 | 906159692 | URP | |
| Shigella sonnei Taxon ID: 624 | 906233097 | URP | |
| Shigella sonnei Taxon ID: 624 | 905426645 | URP | |
| Shigella sonnei Taxon ID: 624 | 905351961 | URP | |
| Shigella sonnei Taxon ID: 624 | 905778958 | URP | |
| Shigella sonnei Taxon ID: 624 | 904988910 | URP | |
| Shigella sonnei Taxon ID: 624 | 905572032 | URP | |
| Shigella sonnei Taxon ID: 624 | 905456642 | URP | |
| Shigella sonnei Taxon ID: 624 | 905093233 | URP | |
| Shigella sonnei Taxon ID: 624 | 905317115 | URP | |
| Shigella sonnei Taxon ID: 624 | 906259460 | URP | |
| Shigella sonnei Taxon ID: 624 | 905642364 | URP | |
| Shigella sonnei Taxon ID: 624 | 905697754 | URP | |
| Shigella sonnei Taxon ID: 624 | 905717980 | URP | |
| Shigella sonnei Taxon ID: 624 | 905855130 | URP | |
| Shigella sonnei Taxon ID: 624 | 906196223 | URP | |
| Shigella sonnei Taxon ID: 624 | 905099349 | URP | |
| Shigella sonnei Taxon ID: 624 | 905667505 | URP | |
| Shigella sonnei Taxon ID: 624 | 905657498 | URP | |
| Shigella sonnei Taxon ID: 624 | 904365467 | URP | |
| Shigella sonnei Taxon ID: 624 | 904375284 | URP | |
| Shigella sonnei Taxon ID: 624 | 905743339 | URP | |
| Shigella sonnei Taxon ID: 624 | 905799223 | URP | |
| Shigella sonnei Taxon ID: 624 | 903812531 | URP | |
| Shigella sonnei Taxon ID: 624 | 905511772 | URP | |
| Shigella sonnei Taxon ID: 624 | 903641310 | URP | |
| Shigella sonnei Taxon ID: 624 | 903604748 | URP | |
| Shigella sonnei Taxon ID: 624 | 903567561 | URP | |
| Shigella sonnei Taxon ID: 624 | 903550163 | URP | |
| Shigella sonnei Taxon ID: 624 | 903146439 | URP | |
| Shigella sonnei Taxon ID: 624 | 903106041 | URP | |
| Shigella sonnei Taxon ID: 624 | 903022971 | URP | |
| Shigella sonnei Taxon ID: 624 | 905361811 | URP | |
| Shigella sonnei Taxon ID: 624 | 905202743 | URP | |
| Shigella sonnei Taxon ID: 624 | 905652300 | URP | |
| Shigella sonnei Taxon ID: 624 | 904639476 | URP | |
| Shigella sonnei Taxon ID: 624 | 904180392 | URP | |
| Shigella sonnei Taxon ID: 624 | 904428811 | URP | |
| Trichuris trichiura Taxon ID: 36087 | 669219720 | URP | |
| Escherichia coli Taxon ID: 562 | 45237908 | URP | |
| Escherichia coli K-12 Taxon ID: 83333 | 298551671 | PRP URP | |
| obsolete GIs = 256023761, 193069052, 193063458, 191172329, 191167344, 188493168, 168786476, 168779119, 168770231, 168754730, 332281764, 331682051, 331676286, 331672168, 331666980, 331661992, 331656655, 331651636, 331645785, 331641130, 312970708, 312965074, 309795505, 309785941, 307312656, 306812940, 301643961, 301302106, 301049822, 301028897, 301025243, 300996742, 300990082, 300939617, 300927274, 300920562, 300907802, 300901132, 300823058, 300817855, 298379666, 293418739, 293413924, 293408753, 293403890, 261257789, 261224095, 237707400, 227884393, 217324391, 208823110, 208815279, 195936177, 194437446, 194427988, 386279651, 375321607, 375320259, 424126187, 424113871, 424107882, 424101069, 424094672, 424088449, 424081830, 423708566, 423701381, 423658897, 423058974, 423052008, 423048425, 423041737, 423036618, 423033751, 423027916, 423022765, 423008576, 423006933, 422998469, 422993259, 422991319, 422975482, 422960508, 422839120, 422834692, 422827832, 422816613, 422799988, 422791415, 422785235, 422782586, 422777308, 422769830, 422765169, 422763456, 422753254, 422748339, 422382466, 422378240, 422369089, 422365190, 422359127, 422355958, 422330896, 421829063, 421822332, 421816943, 421777021, 421459888, 420384256, 420357297, 420340338, 420329919, 420319049, 420314776, 420312706, 420302500, 420290763, 420286285, 420278963, 420273687, 420267855, 420130596, 420125851, 420119345, 420115784, 420108899, 420099957, 420093139, 420087810, 419948743, 419945387, 419936503, 419928993, 419924397, 419917156, 419915316, 419906220, 419900355, 419895871, 419889415, 419880232, 419875863, 419868789, 419812461, 419805023, 419699511, 419411219, 419405728, 419395204, 419390028, 419384780, 419379573, 419374297, 419368898, 419358940, 419353610, 419348696, 419344258, 419338408, 419333005, 419327568, 419321348, 419315505, 419310228, 419305174, 419298908, 419293428, 419288092, 419282549, 419276824, 419271018, 419265306, 419259348, 419253292, 419247542, 419242096, 419236519, 419231048, 419225353, 419219889, 419214236, 419201620, 419195739, 419190410, 419185163, 419179601, 419173987, 419168607, 419162875, 419146407, 419141151, 419135104, 419130366, 419124741, 419119125, 419113535, 419107718, 419102365, 419096512, 419084600, 419078979, 419073718, 419066787, 419060882, 419055289, 419049234, 419043718, 419038348, 419033485, 419027568, 419022761, 419017068, 419012165, 419006296, 419000785, 418995431, 418944429, 418943077, 418301490, 418262658, 418254068, 417977530, 417946395, 417863863, 417831713, 417826737, 417803961, 417754397, 417742190, 417737532, 417732215, 417727097, 417721685, 417716128, 417711170, 417700721, 417665750, 417661171, 417637951, 417633097, 417627558, 417621943, 417617022, 417611649, 417600968, 417595560, 417585427, 417307113, 417294391, 417289939, 417288774, 417275246, 417274122, 417267120, 417263756, 417253529, 417247128, 417229473, 417225119, 417208910, 417193211, 417179418, 417176092, 417167600, 417153275, 417144564, 417139730, 417134662, 417128660, 417118408, 416835746, 416824854, 416814816, 416803899, 416792440, 416781341, 416346285, 416337854, 416325443, 416316989, 416313170, 415876272, 415864188, 415852560, 415828011, 415819004, 415804774, 415789954, 415776974, 414574833, 404373966, 403499280, 425259203, 425252891, 425247156, 425241035, 425235037, 425228737, 425222495, 425215921, 425209877, 425204122, 425197464, 425191136, 425178234, 425172351, 425166063, 425160549, 425154099, 425148480, 425142169, 425136268, 425129927, 425123889, 425118737, 425113975, 425108060, 425102270, 425096124, 424768258, 424761491, 424750294, 424579447, 424561279, 424554935, 424548670, 424542347, 424530461, 424524257, 424518430, 424510863, 424504602, 424498372, 424485155, 424479094, 424473146, 424466588, 424460111, 424423045, 424268121, 424185145, 424145262, 424132281, 425304114, 425298798, 425292763, 425287184, 425281986, 425271308, 425265307, 425421274, 425409043, 425402510, 425396517, 425390394, 425383704, 425377471, 425364460, 425358360, 425352363, 425346145, 425340265, 425333853, 425327670, 425321477, 425315418, 425309495, 429957809, 429949527, 429946895, 429939252, 429933596, 429931992, 429928062, 429922246, 429917208, 429911369, 429907173, 429824266, 429821500, 429815912, 429810152, 429805341, 429801709, 429795783, 429789553, 429784215, 429780165, 429774900, 429722803, 429077071, 429071755, 429059722, 429048680, 429042905, 429036968, 429030785, 429018777, 429012572, 429006245, 428999996, 428993891, 428988074, 428981776, 428976128, 428969645, 428963461, 428957147, 428951291, 428945141, 427808291, 427803700, 474592487, 474559871, 474503552, 474484628, 459125034, 459092088, 459082824, 459058548, 459018837, 459011842, 458986599, 458979619, 458969893, 458954567, 458943634, 458914602, 458884280, 458808433, 458687607, 458626682, 458612391, 458584339, 458572229, 458455209, 458393318, 458372868, 458321181, 458192962, 458180106, 458174197, 458138630, 458094009, 458077863, 458062861, 458053213, 458024067, 458020983, 457975714, 452969754, 450240108, 450211601, 450186074, 445054992, 445049330, 445043766, 445032861, 445027363, 445016670, 445010889, 445005727, 445000268, 444994764, 444989411, 444984171, 444978881, 444973335, 444967831, 444962103, 444956799, 444951345, 444945821, 444940199, 444934621, 444929270, 444922948, 442606861, 442596961, 442595650, 433323330, 433211517, 433206771, 433202121, 433197220, 433192585, 433187440, 433182191, 433177109, 433172469, 433167456, 433162443, 433157610, 433152745, 433148045, 433143232, 433138217, 433133811, 433128995, 433124156, 433119204, 433114539, 433109924, 433104742, 433100165, 433095533, 433090965, 433086261, 433081539, 433076789, 433071703, 433066873, 433061879, 433056948, 433051951, 433046764, 433042138, 433037588, 433032120, 433027636, 433022451, 433017601, 433012827, 433011331, 433004122, 432998802, 432994383, 432989702, 432984285, 432980110, 432977312, 432970769, 432966742, 432960127, 432953763, 432945790, 432942603, 432932973, 432925090, 432917784, 432910914, 432902988, 432897466, 432893372, 432884909, 432880097, 432873263, 432848952, 432842875, 432838197, 432833660, 432830594, 432826128, 432819914, 432814178, 432812744, 432804717, 432797824, 432791859, 432786322, 432782495, 432777533, 432773815, 432769484, 432763966, 432758397, 432753392, 432749074, 432744582, 432740390, 432736141, 432731319, 432726704, 432722161, 432717680, 432712313, 432709438, 432703174, 432697962, 432693392, 432690529, 432684441, 432679066, 432673604, 432669543, 432664869, 432659866, 432654883, 432650116, 432645085, 432639802, 432635913, 432630259, 432626183, 432620713, 432615454, 432610327, 432606472, 432601177, 432596537, 432591706, 432586894, 432582714, 432579289, 432572590, 432567427, 432562511, 432557622, 432552603, 432547329, 432541985, 432536643, 432532786, 432529966, 432522741, 432521330, 432512851, 432503296, 432498851, 432494457, 432488180, 432484335, 432480020, 432474694, 432470025, 432464616, 432459656, 432455517, 432453396, 432445087, 432439974, 432435232, 432430704, 432424834, 432420716, 432415556, 432410656, 432405463, 432400781, 432396531, 432390625, 432386050, 432380279, 432375748, 432361413, 432356941, 432352543, 514489851, 557584861, 557581057, 557567712, 557564238, 548764787, 548757459, 560155434, 618836878, 608100814, 662002850, 618831663, 723106832, 556550738, 544389421, 471332708, 443616664, 410483767, 407468055, 387881207, 387828620, 387620371, 387611130, 387505690, 386708411, 386703810, 386638006, 386633099, 386628179, 386623021, 386618083, 386612805, 386607966, 386605450, 386598371, 386596516, 384542251, 383177202, 378713992, 291281579, 260866776, 260853880, 260842854, 254791794, 254287498, 254160710, 253774386, 251784131, 238899905, 209917886, 209399163, 170681300, 170021015, 157156344, 117622844, 110640857, 91209676, 82543074, 74311164, 480325699, 30062183, 26246608, 388476731, 222155383, 218694068, 218688451, 218557567, 218553170, 215485669, 170080308, 170080207, 157160123, 544575043, 828405798, 828397371, 828395788, 827503912, 338771290 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | A7ZJ15 | LIPA_ECO24 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7UKR9 | LIPA_ECO27 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7MFQ1 | LIPA_ECO45 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7L9H2 | LIPA_ECO55 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | P60718 | LIPA_ECO57 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B5YQH9 | LIPA_ECO5E (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7NLY7 | LIPA_ECO7I (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7MRR5 | LIPA_ECO81 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7M5F5 | LIPA_ECO8A (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | C4ZWB5 | LIPA_ECOBW (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | A7ZXQ4 | LIPA_ECOHS (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | A1A8Q3 | LIPA_ECOK1 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | Q0TK45 | LIPA_ECOL5 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | P60717 | LIPA_ECOL6 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B1IYI0 | LIPA_ECOLC (Swiss-Prot) | |
| Lipoyl synthase | P60716 | LIPA_ECOLI (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B7N9N3 | LIPA_ECOLU (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B6I137 | LIPA_ECOSE (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | B1LKL9 | LIPA_ECOSM (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | Q1RET3 | LIPA_ECOUT (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | Q324R5 | LIPA_SHIBS (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | Q32IV2 | LIPA_SHIDS (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | Q0T6N8 | LIPA_SHIF8 (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | P60719 | LIPA_SHIFL (Swiss-Prot) | |
| Lipoyl synthase {ECO:0000255|HAMAP-Rule:MF_00206} | Q3Z4G4 | LIPA_SHISS (Swiss-Prot) | |
| Show All | |||
Length of Enzyme (full-length): 321 | Length of Functional Domain: 297
MSKPIVMERGVKYRDADKMALIPVKNVATEREALLRKPEWMKIKLPADSTRIQGIKAAMR
KNGLHSVCEEASCPNLAECFNHGTATFMILGAICTRRCPFCDVAHGRPVAPDANEPVKLA
QTIADMALRYVVITSVDRDDLRDGGAQHFADCITAIREKSPQIKIETLVPDFRGRMDRAL
DILTATPPDVFNHNLENVPRIYRQVRPGADYNWSLKLLERFKEAHPEIPTKSGLMVGLGE
TNEEIIEVMRDLRRHGVTMLTLGQYLQPSRHHLPVQRYVSPDEFDEMKAEALAMGFTHAA
CGPFVRSSYHADLQAKGMEVK
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.