Top Level Name
⌊ Superfamily (core) Haloacid Dehalogenase
⌊ Subgroup C1.5: HAD, Beta-PGM, Phosphatase Like
⌊ C1.5.1: Epoxide Hydrolase Phosphatase Like
⌊ FunctionalDomain C1.5.1: Epoxide Hydrolase Phosphatase Like (ID 248326)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS |
| This entry was last updated on | Nov. 22, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Bacteroides thetaiotaomicron VPI-5482 Taxon ID: 226186 | 29346380 | NP_809883.1 (RefSeq) | PRP URP |
| Taxon ID: 816 | 496037100 | WP_008761607.1 (RefSeq) | |
| Bacteroides sp. 1_1_14 Taxon ID: 469585 | 298261849 | EFI04715.1 (Genbank) | URP |
| Bacteroides sp. 1_1_6 Taxon ID: 469586 | 251842295 | EES70375.1 (Genbank) | URP |
| Bacteroides thetaiotaomicron VPI-5482 Taxon ID: 226186 | 29338275 | AAO76077.1 (Genbank) | PRP URP |
| Bacteroides thetaiotaomicron VPI-5482 Taxon ID: 226186 | 81445097 | PRP URP | |
| obsolete GIs = 253568222, 383122608, 298385708 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
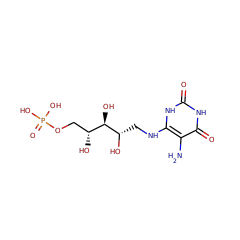
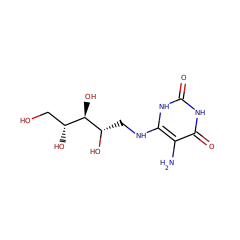
| D-ribitol-5-phosphate phosphatase {ECO:0000305|PubMed:25513739} | Q8A947 | RIBX_BACTN (Swiss-Prot) |
Length of Enzyme (full-length): 206 | Length of Functional Domain: 206
MIKNIVFDFGGVIVDIDRDKAVQAFIKLGLADADTRLDKYHQTGIFQELEEGKLSADEFR
KQLGDLCGRELTMEETKQAWLGFFNEVDLRKLDYILGLRKSYHVYLLSNTNPFVMSWACS
PEFSSEGKPLNDYCDKLYLSYQLGHTKPAPEIFDFMIKDSHVIPSETLFVDDGSSNIHIG
KELGFETFQPENGADWRQELTVILNS
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.