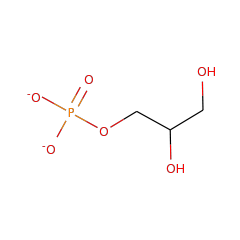
Top Level Name
⌊ Superfamily (core) Haloacid Dehalogenase
⌊ Subgroup C1.5: HAD, Beta-PGM, Phosphatase Like
⌊ C1.5.6: HAD, Beta-PGM, Phosphatase Like
⌊ Family glycerol-3-phosphate phosphatase
⌊ FunctionalDomain glycerol-3-phosphate phosphatase (ID 2207)
No Notes.
| Superfamily Assignment Evidence Code(s) | FSM PubMed:9649311 |
| Family Assignment Evidence Code | CFM PubMed:8662716 |
| This entry was last updated on | Nov. 22, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Saccharomyces cerevisiae S288c Taxon ID: 559292 | 86558907 | NP_012211.2 (RefSeq) | PRP URP |
| Saccharomyces cerevisiae YJM1592 Taxon ID: 1294387 | 766282274 | AJR53688.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1573 Taxon ID: 1294385 | 766281890 | AJR53306.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1549 Taxon ID: 1294384 | 766281698 | AJR53115.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1527 Taxon ID: 1294383 | 766281501 | AJR52919.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1526 Taxon ID: 1294382 | 766268305 | AJR52725.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1479 Taxon ID: 1294381 | 766238403 | AJR52608.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1478 Taxon ID: 1294380 | 766205724 | AJR52416.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1477 Taxon ID: 1294379 | 766175860 | AJR52228.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1463 Taxon ID: 1294378 | 766143799 | AJR52035.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1460 Taxon ID: 1294377 | 766121920 | AJR51848.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1450 Taxon ID: 1294376 | 766121728 | AJR51657.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1447 Taxon ID: 1294375 | 766121538 | AJR51468.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1444 Taxon ID: 1294374 | 766121355 | AJR51286.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1443 Taxon ID: 1294373 | 766120608 | AJR51095.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1434 Taxon ID: 1294371 | 766060462 | AJR50714.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1433 Taxon ID: 1294370 | 766032798 | AJR50521.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1419 Taxon ID: 1294369 | 766003542 | AJR50328.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1418 Taxon ID: 1294368 | 765977427 | AJR50141.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1417 Taxon ID: 1294367 | 765955702 | AJR49952.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1415 Taxon ID: 1294366 | 765939323 | AJR49759.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1402 Taxon ID: 1294365 | 765939129 | AJR49566.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1401 Taxon ID: 1294364 | 765938938 | AJR49376.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1400 Taxon ID: 1294363 | 765938751 | AJR49190.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1399 Taxon ID: 1294362 | 765938614 | AJR49054.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1389 Taxon ID: 1294361 | 765938420 | AJR48861.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1388 Taxon ID: 1294360 | 765923586 | AJR48669.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1387 Taxon ID: 1294359 | 765911019 | AJR48476.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1386 Taxon ID: 1294358 | 765893407 | AJR48283.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1385 Taxon ID: 1294357 | 765874911 | AJR48092.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1383 Taxon ID: 1294356 | 765851978 | AJR47921.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1381 Taxon ID: 1294355 | 765834045 | AJR47733.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1356 Taxon ID: 1294354 | 765827068 | AJR47542.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1355 Taxon ID: 1294353 | 765826873 | AJR47348.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1342 Taxon ID: 1294352 | 765826684 | AJR47160.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1341 Taxon ID: 1294351 | 765826276 | AJR46969.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1338 Taxon ID: 1294350 | 765816316 | AJR46775.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1336 Taxon ID: 1294349 | 765802060 | AJR46582.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1332 Taxon ID: 1294348 | 765783660 | AJR46390.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1326 Taxon ID: 1294347 | 765747907 | AJR46201.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1311 Taxon ID: 1294346 | 765717241 | AJR46009.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1304 Taxon ID: 1294344 | 765706860 | AJR45632.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1273 Taxon ID: 1294343 | 765706667 | AJR45440.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1252 Taxon ID: 1294342 | 765706474 | AJR45248.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1615 Taxon ID: 1294388 | 765706280 | AJR45055.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1250 Taxon ID: 1294341 | 765706091 | AJR44867.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1244 Taxon ID: 1294339 | 765705704 | AJR44482.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1242 Taxon ID: 1294338 | 765705510 | AJR44289.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1208 Taxon ID: 1294337 | 765705314 | AJR44094.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1202 Taxon ID: 1294336 | 765705122 | AJR43903.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1199 Taxon ID: 1294335 | 765704934 | AJR43716.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1190 Taxon ID: 1294334 | 765704741 | AJR43524.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1133 Taxon ID: 1294333 | 765704545 | AJR43329.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1129 Taxon ID: 1293430 | 765704351 | AJR43136.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1083 Taxon ID: 1292971 | 765704157 | AJR42943.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM996 Taxon ID: 1294332 | 765703968 | AJR42755.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM990 Taxon ID: 1294330 | 765703773 | AJR42561.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM987 Taxon ID: 1294329 | 765703578 | AJR42367.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM984 Taxon ID: 1294328 | 765703387 | AJR42177.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM981 Taxon ID: 1294327 | 765703192 | AJR41983.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM978 Taxon ID: 1294326 | 765702998 | AJR41790.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM975 Taxon ID: 1294325 | 765702803 | AJR41596.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM972 Taxon ID: 1294324 | 765702398 | AJR41403.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM969 Taxon ID: 1294323 | 765689049 | AJR41209.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM693 Taxon ID: 1294322 | 765688625 | AJR41037.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM689 Taxon ID: 1294321 | 765688346 | AJR40843.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM683 Taxon ID: 1294320 | 765685253 | AJR40663.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM682 Taxon ID: 1294319 | 765685058 | AJR40469.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM681 Taxon ID: 1294318 | 765684853 | AJR40277.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM627 Taxon ID: 1294317 | 765684664 | AJR40089.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM555 Taxon ID: 1294316 | 765684471 | AJR39897.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM554 Taxon ID: 1294315 | 765683615 | AJR39706.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM541 Taxon ID: 1294314 | 765680430 | AJR39527.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM470 Taxon ID: 1294313 | 765680246 | AJR39344.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM456 Taxon ID: 1294312 | 765669938 | AJR39160.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM453 Taxon ID: 1294311 | 765669400 | AJR38967.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM451 Taxon ID: 502869 | 765669204 | AJR38772.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM450 Taxon ID: 1294310 | 765665288 | AJR38582.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM428 Taxon ID: 947044 | 765647314 | AJR38390.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM326 Taxon ID: 468558 | 765615727 | AJR38196.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM320 Taxon ID: 947042 | 765597455 | AJR38006.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM271 Taxon ID: 1294309 | 765576378 | AJR37824.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM248 Taxon ID: 1294307 | 765568661 | AJR37434.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM244 Taxon ID: 1294306 | 765546362 | AJR37242.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM195 Taxon ID: 1294305 | 765543141 | AJR37094.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM193 Taxon ID: 1294304 | 765542949 | AJR36903.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM189 Taxon ID: 1294303 | 765531053 | AJR36708.1 (Genbank) | URP |
| Saccharomyces cerevisiae YJM1078 Taxon ID: 1296266 | 761460103 | AJP39415.1 (Genbank) | URP |
| n/a | 756729206 | AJM56145.1 (Genbank) | |
| n/a | 756729034 | AJM55973.1 (Genbank) | |
| Saccharomyces cerevisiae YJM993 Taxon ID: 1294331 | 628175566 | AHY75947.1 (Genbank) | URP |
| Saccharomyces cerevisiae P283 Taxon ID: 1177187 | 584476164 | EWH17915.1 (Genbank) | URP |
| Saccharomyces cerevisiae R103 Taxon ID: 1182967 | 584375601 | EWG95482.1 (Genbank) | URP |
| Saccharomyces cerevisiae P301 Taxon ID: 1182968 | 584370564 | EWG90509.1 (Genbank) | URP |
| Saccharomyces cerevisiae R008 Taxon ID: 1182966 | 584365343 | EWG85345.1 (Genbank) | URP |
| Saccharomyces cerevisiae CEN.PK113-7D Taxon ID: 889517 | 392298665 | EIW09761.1 (Genbank) | URP |
| Saccharomyces cerevisiae JAY291 Taxon ID: 574961 | 256269766 | EEU05032.1 (Genbank) | URP |
| Saccharomyces cerevisiae Taxon ID: 4932 | 311816304 | ||
| Saccharomyces cerevisiae S288c Taxon ID: 559292 | 285812595 | PRP URP | |
| Saccharomyces cerevisiae Taxon ID: 4932 | 227483892 | ||
| Saccharomyces cerevisiae Taxon ID: 4932 | 227467896 | ||
| Saccharomyces cerevisiae Taxon ID: 4932 | 206986851 | ||
| Saccharomyces cerevisiae S288c Taxon ID: 559292 | 2507038 | PRP URP | |
| Saccharomyces cerevisiae Taxon ID: 4932 | 1236750 | ||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| Glycerol-1-phosphate phosphohydrolase 1 {ECO:0000305} | P41277 | GPP1_YEAST (Swiss-Prot) |
Length of Enzyme (full-length): 250 | Length of Functional Domain: 250
MPLTTKPLSLKINAALFDVDGTIIISQPAIAAFWRDFGKDKPYFDAEHVIHISHGWRTYD
AIAKFAPDFADEEYVNKLEGEIPEKYGEHSIEVPGAVKLCNALNALPKEKWAVATSGTRD
MAKKWFDILKIKRPEYFITANDVKQGKPHPEPYLKGRNGLGFPINEQDPSKSKVVVFEDA
PAGIAAGKAAGCKIVGIATTFDLDFLKEKGCDIIVKNHESIRVGEYNAETDEVELIFDDY
LYAKDDLLKW
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.