Top Level Name
⌊ Superfamily (extended) Radical SAM 3-amino-3-carboxypropyl Radical Forming
Family known  |
|||||||
| Total |
100%  |
<100%  |
Family unknown  |
||||
| Functional domains | 2420 | 1024 | 180 | 1216 | |||
| UniProtKB | 2306 | 823 | 172 | 1311 | |||
| GI | 4318 | 1497 | 426 | 2395 | |||
| Structures | 2 | ||||||
| Reactions | 0 | ||||||
| Functional domains of this superfamily were last updated on June 10, 2017 | |||||||
| New functional domains were last added to this superfamily on March 23, 2015 | |||||||
Diphthamide, the target of diphtheria toxin, is a unique posttranslational modification on translation elongation factor 2 (EF2) in archaea and eukaryotes. The biosynthesis of diphthamide is proposed to involve three steps. The first step of which is the transfer of the 3-amino-3-carboxypropyl group from S-adenosyl-l-methionine (SAM) to the histidine residue of EF2, forming a C–C bond. This step requires four proteins Dph1-Dph4. Dph2 is is radical SAM-like protein which forms a homodimer in Pyrococcus horikoshii and a complex with Dph1 in yeast that is responsible for the first step. Dph3 has been demonstrated to act as the electron donor for the radical SAM FeS reduction in this system.
Uses SAM and a 4Fe-4S cluster to form a 3-amino-3-carboxypropyl radical intermediate. No obvious structural or sequence similarity to the main Radical SAM superfamily. The reference cited for this superfamily describes the function of the only defined family identified to date.
Zhang Y, Zhu X, Torelli AT, Lee M, Dzikovski B, Koralewski RM, Wang E, Freed J, Krebs C, Ealick SE, Lin H
Diphthamide biosynthesis requires an organic radical generated by an iron-sulphur enzyme
▸ Abstract
Nature 2010;465(7300):891-896 | PubMed ID: 20559380
Dong M, Su X, Dzikovski B, Dando EE, Zhu X, Du J, Freed JH, Lin H
Dph3 is an electron donor for Dph1-Dph2 in the first step of eukaryotic diphthamide biosynthesis
▸ Abstract
J Am Chem Soc 2014;136(5):1754-1757 | PubMed ID: 24422557
Dong M, Horitani M, Dzikovski B, Pandelia ME, Krebs C, Freed JH, Hoffman BM, Lin H
Organometallic Complex Formed by an Unconventional Radical S-Adenosylmethionine Enzyme
▸ Abstract
J Am Chem Soc 2016;None(None):None-None | PubMed ID: 27465315
Previous genetic studies showed this step requires four proteins in eukaryotes, Dph1–Dph4. However, the exact molecular functions for the four proteins are unknown. Previous study showed that Pyrococcus horikoshii Dph2 (PhDph2), a novel iron-sulfur cluster-containing enzyme, forms a homodimer and is sufficient for the first step of diphthamide biosynthesis in vitro. Here we demonstrate by in vitro reconstitution that yeast Dph1 and Dph2 form a complex (Dph1-Dph2) that is equivalent to the homodimer of PhDph2 and is sufficient to catalyze the first step in vitro in the presence of dithionite as the reductant. We further demonstrate that yeast Dph3 (also known as KTI11), a CSL-type zinc finger protein, can bind iron and in the reduced state can serve as an electron donor to reduce the Fe-S cluster in Dph1-Dph2. Our study thus firmly establishes the functions for three of the proteins involved in eukaryotic diphthamide biosynthesis. For most radical SAM enzymes in bacteria, flavodoxins and flavodoxin reductases are believed to serve as electron donors for the Fe-S clusters. The finding that Dph3 is an electron donor for the Fe-S clusters in Dph1-Dph2 is thus interesting and opens up new avenues of research on electron transfer to Fe-S proteins in eukaryotic cells.
Cofactor(s)
[4Fe-4S]+ Cluster (complex)

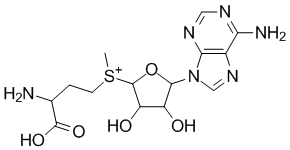
S-Adenosylmethionine (coenzyme)
Static File Downloads
| File Name | Description | Parameters | Stats |
|---|---|---|---|
| repnet.sf32.th50.pE20.mek250.xgmml | Representative network: each node is a group of similar sequences | node similarity threshold = 50 max edge count = 250 min -log10 E = 20 |
size = 6.7M num_edges = 19250 num_nodes = 350 |
| sfld_superfamily_32.tsv | Annotation data table, tab separated columns | size=737K #rows=2421 |
|
| sfld_superfamily_32.fasta | Protein sequences, fasta format | size=1.1M #seqs=2420 |
|
| sfldAlignmentSF32.msa | Annotated Sequence Alignment, Stockholm format | 163 sequences size: 658K |
| Subgroup ▸ Legend | T  |
K  |
C  |
U  |
S  |
||
|---|---|---|---|---|---|---|---|
| Diphthamide biosynthesis | 1204 | 1204 | 1024 | 0 | 2 | ||
| ┗ Diphthamide biosynthesis family (Dph2) | 1204 | 1204 | 1024 | 2 |