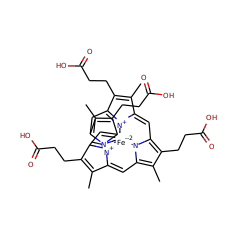
An alternative route for haem b biosynthesis is operative in sulphate-reducing bacteria of the Desulfovibrio genus and in methanogenic Archaea. This pathway diverges from the canonical one at the level of uroporphyrinogen III and progresses via a distinct branch, where sirohaem acts as an intermediate precursor being converted into haem b by a set of novel enzymes, named the alternative haem biosynthetic proteins (Ahb). AhbD is the fourth enzyme in this gene and converts iron-coproporphyrin III into haem b in two steps and contains two iron-sulfur clusters.
Kühner M, Haufschildt K, Neumann A, Storbeck S, Streif J, Layer G
The Alternative Route to Heme in the Methanogenic Archaeon Methanosarcina barkeri
▸ Abstract
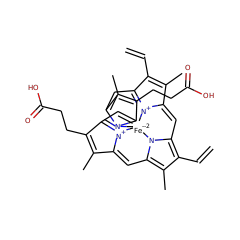
In living organisms heme is formed from the common precursor uroporphyrinogen III by either one of two substantially different pathways. In contrast to eukaryotes and most bacteria which employ the so-called "classical" heme biosynthesis pathway, the archaea use an alternative route. In this pathway, heme is formed from uroporphyrinogen III via the intermediates precorrin-2, sirohydrochlorin, siroheme, 12,18-didecarboxysiroheme, and iron-coproporphyrin III. In this study the heme biosynthesis proteins AhbAB, AhbC, and AhbD from Methanosarcina barkeri were functionally characterized. Using an in vivo enzyme activity assay it was shown that AhbA and AhbB (Mbar_A1459 and Mbar_A1460) together catalyze the conversion of siroheme into 12,18-didecarboxysiroheme. The two proteins form a heterodimeric complex which might be subject to feedback regulation by the pathway end-product heme. Further, AhbC (Mbar_A1793) was shown to catalyze the formation of iron-coproporphyrin III in vivo. Finally, recombinant AhbD (Mbar_A1458) was produced in E. coli and purified indicating that this protein most likely contains two [4Fe-4S] clusters. Using an in vitro enzyme activity assay it was demonstrated that AhbD catalyzes the conversion of iron-coproporphyrin III into heme.
Archaea
2014;2014(None):32763-327637
| PubMed ID:
24669201
Lobo SA, Lawrence AD, Romão CV, Warren MJ, Teixeira M, Saraiva LM
Characterisation of Desulfovibrio vulgaris haem b synthase, a radical SAM family member
▸ Abstract
An alternative route for haem b biosynthesis is operative in sulphate-reducing bacteria of the Desulfovibrio genus and in methanogenic Archaea. This pathway diverges from the canonical one at the level of uroporphyrinogen III and progresses via a distinct branch, where sirohaem acts as an intermediate precursor being converted into haem b by a set of novel enzymes, named the alternative haem biosynthetic proteins (Ahb). In this work, we report the biochemical characterisation of the Desulfovibrio vulgaris AhbD enzyme that catalyses the last step of the pathway. Mass spectrometry analysis showed that AhbD promotes the cleavage of S-adenosylmethionine (SAM) and converts iron-coproporphyrin III via two oxidative decarboxylations to yield haem b, methionine and the 5'-deoxyadenosyl radical. Electron paramagnetic resonance spectroscopy studies demonstrated that AhbD contains two [4Fe-4S](2+/1+) centres and that binding of the substrates S-adenosylmethionine and iron-coproporphyrin III induces conformational modifications in both centres. Amino acid sequence comparisons indicated that D. vulgaris AhbD belongs to the radical SAM protein superfamily, with a GGE-like motif and two cysteine-rich sequences typical for ligation of SAM molecules and iron-sulphur clusters, respectively. A structural model of D. vulgaris AhbD with putative binding pockets for the iron-sulphur centres and the substrates SAM and iron-coproporphyrin III is discussed.
Biochim Biophys Acta
2014;None(None):None-None
| PubMed ID:
24713144
Bali S, Palmer DJ, Schroeder S, Ferguson SJ, Warren MJ
Recent advances in the biosynthesis of modified tetrapyrroles: the discovery of an alternative pathway for the formation of heme and heme d 1
▸ Abstract
Hemes (a, b, c, and o) and heme d 1 belong to the group of modified tetrapyrroles, which also includes chlorophylls, cobalamins, coenzyme F430, and siroheme. These compounds are found throughout all domains of life and are involved in a variety of essential biological processes ranging from photosynthesis to methanogenesis. The biosynthesis of heme b has been well studied in many organisms, but in sulfate-reducing bacteria and archaea, the pathway has remained a mystery, as many of the enzymes involved in these characterized steps are absent. The heme pathway in most organisms proceeds from the cyclic precursor of all modified tetrapyrroles uroporphyrinogen III, to coproporphyrinogen III, which is followed by oxidation of the ring and finally iron insertion. Sulfate-reducing bacteria and some archaea lack the genetic information necessary to convert uroporphyrinogen III to heme along the "classical" route and instead use an "alternative" pathway. Biosynthesis of the isobacteriochlorin heme d 1, a cofactor of the dissimilatory nitrite reductase cytochrome cd 1, has also been a subject of much research, although the biosynthetic pathway and its intermediates have evaded discovery for quite some time. This review focuses on the recent advances in the understanding of these two pathways and their surprisingly close relationship via the unlikely intermediate siroheme, which is also a cofactor of sulfite and nitrite reductases in many organisms. The evolutionary questions raised by this discovery will also be discussed along with the potential regulation required by organisms with overlapping tetrapyrrole biosynthesis pathways.
Cell Mol Life Sci
2014;None(None):None-None
| PubMed ID:
24515122
Kuhner, M; Schweyen, P; Hoffmann, M; Ramos, JV; Reijerse, EJ; Lubitz, W; Broring, M; Layer, G
The auxiliary [4Fe–4S] cluster of the Radical SAM heme synthase from Methanosarcina barkeri is involved in electron transfer
▸ Abstract
The heme synthase AhbD catalyzes the oxidative decarboxylation of two propionate side chains of iron-coproporphyrin III to the corresponding vinyl groups of heme during the alternative heme biosynthesis pathway occurring in sulfate-reducing bacteria and archaea. AhbD belongs to the family of Radical SAM enzymes and contains two [4Fe–4S] clusters. Whereas one of these clusters is required for substrate radical formation, the role of the second iron–sulfur cluster is not known. In this study, the function of the auxiliary cluster during AhbD catalysis was investigated. Two single cluster variants of AhbD from M. barkeri carrying either one of the two clusters were created. Using these enzyme variants it was shown that the auxiliary cluster is not required for substrate binding and formation of the substrate radical. Instead, the auxiliary cluster is involved in a late step of AhbD catalysis most likely in electron transfer from the reaction intermediate to a final electron acceptor. Moreover, by using alternative substrates such as coproporphyrin III, Cu-coproporphyrin III and Zn-coproporphyrin III for the AhbD activity assay it was observed that the central iron ion of the porphyrin substrate also participates in the electron transfer from the reaction intermediate to the auxiliary [4Fe–4S] cluster. Altogether, new insights concerning the completely uncharacterized late steps of AhbD catalysis were obtained.
CHEMICAL SCIENCE
2016;7(7):4633-4643
| PubMed ID:
None