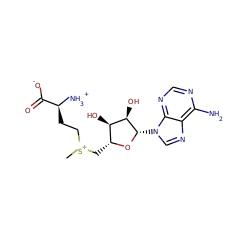
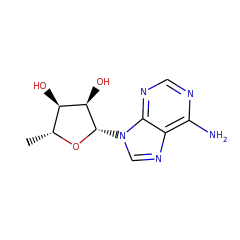
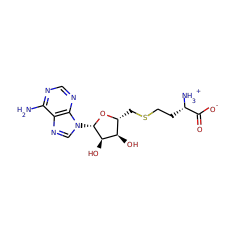
Catalyses the methylthiolation of N6-(dimethylallyl)adenosine (i6A), leading to the formation of 2-methylthio-N6-(dimethylallyl)adenosine (ms2i6A) at position 37 in tRNAs that read codons beginning with uridine. Binds 2 4Fe-4S clusters. One cluster is coordinated with 3 cysteines and an exchangeable S-adenosyl-L-methionine, the other is thought to be the sulfur donor. The second cluster has a polysulfide group bound to it, which is methylated in the first reaction step. Concurrently, the SAM-[4Fe-4S] cluster forms the 5'-dAdo radical, which abstracts a hydrogen atom from the substrate, which is then methylthiolated by the methylated polysulfide group.
Pierrel F, Hernandez HL, Johnson MK, Fontecave M, Atta M
MiaB protein from Thermotoga maritima. Characterization of an extremely thermophilic tRNA-methylthiotransferase
▸ Abstract
In Escherichia coli, the MiaB protein catalyzes the methylthiolation of N-6-isopentenyl adenosine in tRNAs, the last reaction step during biosynthesis of 2-methylthio-N-6-isopentenyl adenosine (ms2i6A-37). For the first time the thermophilic bacterium Thermotoga maritima is shown here to contain such a MiaB tRNA-modifying enzyme, named MiaBTm, and to synthesize ms2i6A-37 as demonstrated by an analysis of modified nucleosides from tRNA hydrolysates. The corresponding gene (TM0653) was identified by sequence similarity to the miaB gene cloned and expressed in E. coli. MiaBTm was purified to homogeneity and thoroughly characterized by biochemical and spectroscopic methods. It is a monomer of 443 residues with a molecular mass of 50,710 kilodaltons. Its amino acid sequence shares the CysXXX-CysXXCys sequence with MiaB from E. coli as well as with biotin synthase and lipoate synthase. This sequence was shown to be essential for chelation of an iron-sulfur center and for activity in these enzymes. As isolated, MiaBTm contains both iron and sulfide and an apoprotein form can coordinate up to 4 iron and 4 sulfur atoms per polypeptide chain. UV-visible absorption, resonance Raman, variable temperature magnetic circular dichroism, and EPR spectroscopy of MiaBTm indicate the presence of a [4Fe-4S]+2/+1 cluster under reducing and anaerobic conditions, whereas [3Fe-4S]+1 and [2Fe-2S]+2 forms are generated under aerobic conditions. The redox potential of the [4Fe-4S]+2/+1 transition is -495 +/- 10 mV (versus the normal hydrogen electrode). Finally, the expression of MiaBTm from T. maritima in an E. coli mutant strain lacking functional miaB gene allowed production of ms2i6A-37. These results provide further information on the enzymes involved in methylthiolation of tRNAs.
J Biol Chem.
2003;278(32):29515-29524
| PubMed ID:
12766153
Pierrel F, Douki T, Fontecave M, Atta M
MiaB protein is a bifunctional radical-S-adenosylmethionine enzyme involved in thiolation and methylation of tRNA
▸ Abstract
The last biosynthetic step for 2-methylthio-N(6)-isopentenyl-adenosine (ms(2)i(6)A), present at position 37 in some tRNAs, consists of the methylthiolation of the isopentenyl-adenosine (i(6)A) precursor. In this work we have reconstituted in vitro the conversion of i(6)A to ms(2)i(6)A within a tRNA substrate using the iron-sulfur MiaB protein, S-adenosylmethionine (AdoMet), and a reducing agent. We show that a synthetic i(6)A-containing RNA corresponding to the anticodon stem loop of tRNA(Phe) is also a substrate. This study demonstrates that MiaB protein is a bifunctional system, involved in both thiolation and methylation of i(6)A. In this process, one molecule of AdoMet is converted to 5'-deoxyadenosine, probably through reductive cleavage and intermediate formation ofa5'-deoxyadenosyl radical as observed in other "Radical-AdoMet" enzymes, and a second molecule of AdoMet is used as a methyl donor as shown by labeling experiments. The origin of the sulfur atom is discussed.
J Biol Chem.
2004;279(46):47555-47563
| PubMed ID:
15339930
Hernández HL, Pierrel F, Elleingand E, García-Serres R, Huynh BH, Johnson MK, Fontecave M, Atta M
MiaB, a bifunctional radical-S-adenosylmethionine enzyme involved in the thiolation and methylation of tRNA, contains two essential [4Fe-4S] clusters
▸ Abstract
The radical-S-adenosylmethionine (radical-AdoMet) enzyme MiaB catalyzes the posttranscriptional methylthiolation of N-6-isopentenyladenosine in tRNAs. Spectroscopic and analytical studies of the reconstituted wild-type and C150/154/157A triple variant forms of Thermotoga maritima MiaB have revealed the presence of two distinct [4Fe-4S](2+,1+) clusters in the protein. One is coordinated by the three conserved cysteines in the radical-AdoMet motif (Cys150, Cys154, and Cys157) as previously reported, and the other, here observed for the first time, is proposed to be coordinated by the three N-terminal conserved cysteines (Cys10, Cys46, and Cys79). The two [4Fe-4S]2+ clusters have similar UV-visible absorption, resonance Raman, and Mössbauer properties but differ in terms of redox properties and the EPR properties of the reduced [4Fe-4S]1+ clusters. Reconstituted forms of MiaB containing two [4Fe-4S] clusters are more active than previously reported. Comparison of MiaB with other radical-AdoMet enzymes involved in thiolation reactions, such as biotin synthase and lipoate synthase, is discussed as well as a possible role of the second cluster as a sacrificial S-donor in the MiaB-catalyzed reaction.
Biochemistry
2007;46(174):5140-5147
| PubMed ID:
17407324
Landgraf BJ, Arcinas AJ, Lee KH, Booker SJ
Identification of an Intermediate Methyl Carrier in the Radical S-Adenosylmethionine Methylthiotransferases RimO and MiaB
▸ Abstract
RimO and MiaB are radical S-adenosylmethionine (SAM) enzymes that catalyze the attachment of methylthio (-SCH3) groups to macromolecular substrates. RimO attaches a methylthio group at C3 of aspartate 89 of protein S12, a component of the 30S subunit of the bacterial ribosome. MiaB attaches a methylthio group at C2 of N(6)-(isopentenyl)adenosine, found at nucleotide 37 in several prokaryotic tRNAs. These two enzymes are prototypical members of a subclass of radical SAM enzymes called methylthiotransferases (MTTases). It had been assumed that the sequence of steps in MTTase reactions involves initial sulfur insertion into the organic substrate followed by capping of the inserted sulfur atom with a SAM-derived methyl group. In this work, however, we show that both RimO and MiaB from Thermotoga maritima catalyze methyl transfer from SAM to an acid/base labile acceptor on the protein in the absence of their respective macromolecular substrates. Consistent with the assignment of the acceptor as an iron-sulfur cluster, denaturation of the SAM-treated protein with acid results in production of methanethiol. When RimO or MiaB is first incubated with SAM in the absence of substrate and reductant and then incubated with excess S-adenosyl-l-[methyl-d3]methionine in the presence of substrate and reductant, production of the unlabeled product precedes production of the deuterated product, showing that the methylated species is chemically and kinetically competent to be an intermediate.
J Am Chem Soc
2013;135(41):15404-15416
| PubMed ID:
23991893