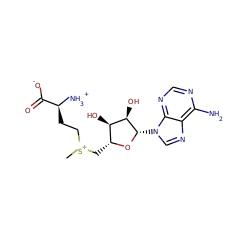
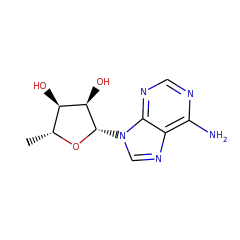
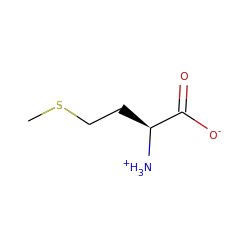
Top Level Name
⌊ Superfamily (core) Radical SAM
⌊ Subgroup SPASM/twitch domain containing
⌊ main SPASM domain-containing
⌊ Family coenzyme PQQ synthesis protein E (PqqE-like)
⌊ FunctionalDomain Coenzyme PQQ synthesis protein E (ID 381007)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS |
| Family Assignment Evidence Code | CFM ISS PubMed:18371220 |
| This entry was last updated on | Dec. 10, 2016 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Taxon ID: 570 | 490288534 | WP_004184158.1 (RefSeq) | |
| Klebsiella pneumoniae Taxon ID: 573 | 846738473 | KMK49591.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839787686 | KMI60428.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839757825 | KMI30799.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839747225 | KMI20320.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839739123 | KMI12324.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839733848 | KMI07086.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839681897 | KMH55750.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839646633 | KMH21197.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839627790 | KMH02528.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839625857 | KMH00800.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839609458 | KMG84406.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839598243 | KMG73570.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839580252 | KMG55623.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839420352 | KME97085.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839402533 | KME79425.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839246797 | KMD25180.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839239367 | KMD17828.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839156796 | KMB36099.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 839048098 | KMA28402.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 838990141 | KLZ70934.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 820950707 | AKG99217.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 770614017 | KJL11815.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 770611883 | KJL09694.1 (Genbank) | URP |
| Klebsiella pneumoniae 1158 Taxon ID: 1392499 | 743571881 | AJC04610.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 727676071 | KHF71093.1 (Genbank) | URP |
| Klebsiella pneumoniae subsp. pneumoniae Taxon ID: 72407 | 723251907 | AIX84531.1 (Genbank) | URP |
| Klebsiella pneumoniae subsp. pneumoniae Taxon ID: 72407 | 723236941 | AIX69580.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 721490721 | KGY34282.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 719630324 | AIW99607.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 694260904 | KGJ46334.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 694253180 | KGJ38691.1 (Genbank) | URP |
| Klebsiella pneumoniae Taxon ID: 573 | 673512788 | KFJ77026.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 17 Taxon ID: 1400170 | 655431648 | KED01823.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 68 Taxon ID: 1438811 | 636472725 | KDM49174.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 56 Taxon ID: 1438803 | 636443171 | KDM19930.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH-74 Taxon ID: 1438797 | 636416788 | KDL93765.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH-70 Taxon ID: 1438793 | 636393951 | KDL71101.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 66 Taxon ID: 1438789 | 636372689 | KDL49993.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 60 Taxon ID: 1438785 | 636358435 | KDL35870.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 52 Taxon ID: 1438783 | 636349342 | KDL26836.1 (Genbank) | URP |
| Klebsiella pneumoniae BWH 48 Taxon ID: 1438706 | 635976366 | KDH56396.1 (Genbank) | URP |
| Klebsiella pneumoniae BIDMC 69 Taxon ID: 1438702 | 635956673 | KDH36838.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 64 Taxon ID: 1438809 | 612321710 | EZR14046.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 60 Taxon ID: 1438805 | 612319628 | EZR12015.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 35 Taxon ID: 1328371 | 583711983 | EWF74063.1 (Genbank) | URP |
| Klebsiella pneumoniae BWH 2 Taxon ID: 1328381 | 583678630 | EWF40959.1 (Genbank) | URP |
| Klebsiella pneumoniae BIDMC 35 Taxon ID: 1328424 | 583578383 | EWE41526.1 (Genbank) | URP |
| Klebsiella pneumoniae BIDMC 46b Taxon ID: 1400164 | 583554857 | EWE18207.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 25 Taxon ID: 1400176 | 583494330 | EWD58103.1 (Genbank) | URP |
| Klebsiella pneumoniae UCI 26 Taxon ID: 1400177 | 583488467 | EWD52306.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 17 Taxon ID: 1328362 | 555217094 | ESN59394.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 18 Taxon ID: 1328363 | 555209710 | ESN52040.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 19 Taxon ID: 1328364 | 555196423 | ESN38864.1 (Genbank) | URP |
| Klebsiella pneumoniae MGH 48 Taxon ID: 1328380 | 555114362 | ESM57327.1 (Genbank) | URP |
| Klebsiella pneumoniae BWH 30 Taxon ID: 1328385 | 555096494 | ESM39610.1 (Genbank) | URP |
| Klebsiella pneumoniae UCICRE 8 Taxon ID: 1328394 | 555060531 | ESM04755.1 (Genbank) | URP |
| Klebsiella pneumoniae UCICRE 7 Taxon ID: 1328393 | 555059226 | ESM03452.1 (Genbank) | URP |
| Klebsiella pneumoniae BIDMC 22 Taxon ID: 1328417 | 555019600 | ESL64029.1 (Genbank) | URP |
| Escherichia coli 909957 Taxon ID: 1269006 | 553376045 | ESB02151.1 (Genbank) | URP |
| Klebsiella pneumoniae KP-1 Taxon ID: 1365186 | 542317595 | ERI58583.1 (Genbank) | URP |
| Klebsiella pneumoniae subsp. pneumoniae WGLW5 Taxon ID: 1203547 | 405612680 | EKB85431.1 (Genbank) | URP |
| Klebsiella pneumoniae subsp. pneumoniae 1084 Taxon ID: 1193292 | 402541575 | AFQ65724.1 (Genbank) | URP |
| Klebsiella sp. 4_1_44FAA Taxon ID: 665944 | 363655190 | EHL94048.1 (Genbank) | URP |
| Klebsiella pneumoniae KCTC 2242 Taxon ID: 1049565 | 339762073 | AEJ98293.1 (Genbank) | URP |
| Klebsiella pneumoniae BJ1-GA Taxon ID: 1379687 | 573072868 | URP | |
| Klebsiella pneumoniae T69 Taxon ID: 1379689 | 573046237 | URP | |
| Klebsiella pneumoniae subsp. pneumoniae Ecl8 Taxon ID: 1226680 | 414708173 | URP | |
| Klebsiella pneumoniae NTUH-K2044 Taxon ID: 484021 | 238547164 | URP | |
| Klebsiella pneumoniae Taxon ID: 573 | 809708 | URP | |
| Klebsiella pneumoniae Taxon ID: 573 | 130802 | URP | |
| obsolete GIs = 425091621, 424830742, 365138212, 402780672, 386034945, 238894848 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| Coenzyme PQQ synthesis protein E | P27507 | PQQE_KLEPN (Swiss-Prot) |
Length of Enzyme (full-length): 380 | Length of Functional Domain: 378
MSQSKPTVNPPLWLLAELTYRCPLQCPYCSNPLDFARQDKELTTEQWIEVFRQARAMGSV
QLGFSGGEPLTRKDLPELIRAARDLGFYTNLITSGIGLTESKLDAFSEAGLDHIQISFQA
SDEVLNAALAGNKKAFQQKLAMAKAVKARDYPMVLNFVLHRHNIDQLDKIIELCIELEAD
DVELATCQFYGWAFLNREGLLPTREQIARAEQVVADYRQKMAASGNLTNLLFVTPDYYEE
RPKGCMGGWGSIFLSVTPEGTALPCHSARQLPVAFPSVLEQSLESIWYDSFGFNRYRGYD
WMPEPCRSCDEKEKDFGGCRCQAFMLTGSADNADPVCSKSPHHHKILEARREAACSDIKV
SQLQFRNRTRSQLIYQTRDL
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.