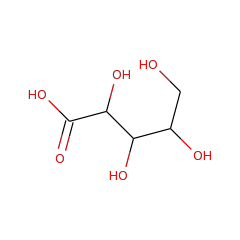
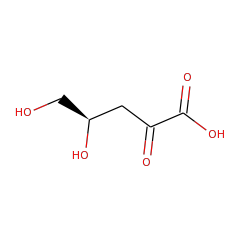
Top Level Name
⌊ Superfamily (core) Enolase
⌊ Subgroup mandelate racemase
⌊ Family L-lyxonate dehydratase
⌊ FunctionalDomain L-lyxonate dehydratase (ID 282)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS PubMed:24831290 |
| Family Assignment Evidence Code | IGS PubMed:24831290 |
| This entry was last updated on | Nov. 21, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Pseudomonas aeruginosa PAO1 Taxon ID: 208964 | 15597411 | NP_250905.1 (RefSeq) | PRP URP |
| Pseudomonas aeruginosa PAO1-VE13 Taxon ID: 1367494 | 651879623 | YP_008706369.1 (RefSeq) | URP |
| Pseudomonas aeruginosa PAO1-VE2 Taxon ID: 1367493 | 651874250 | YP_008694088.1 (RefSeq) | URP |
| Pseudomonas aeruginosa PAO581 Taxon ID: 1352354 | 651861828 | YP_008558115.1 (RefSeq) | URP |
| Pseudomonas aeruginosa c7447m Taxon ID: 1352355 | 651854610 | YP_008552447.1 (RefSeq) | URP |
| Taxon ID: 286 | 489204637 | WP_003113703.1 (RefSeq) | |
| Pseudomonas aeruginosa DSM 50071 Taxon ID: 1123015 | 861673605 | AKO86889.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 861347510 | KMQ55778.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 856782681 | KMN00965.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 823574176 | KLI80168.1 (Genbank) | URP |
| Pseudomonas aeruginosa MRSN 321 Taxon ID: 1410038 | 816098633 | KKJ46029.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 815971859 | AKE70018.1 (Genbank) | URP |
| Pseudomonas sp. HMSC05H02 Taxon ID: 1581118 | 768693156 | KJJ22446.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 763856529 | KJC16041.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 751373820 | AJF49648.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 745844030 | AJD64274.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 725805309 | KHE59554.1 (Genbank) | URP |
| Pseudomonas sp. YS-1p Taxon ID: 1534978 | 687283655 | KGD88931.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 674738992 | KFL05974.1 (Genbank) | URP |
| Pseudomonas aeruginosa PGPR2 Taxon ID: 1328315 | 667999364 | KFB18878.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO1H2O Taxon ID: 1147787 | 660506822 | AID73431.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 658113815 | KEI28528.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 656994546 | KEF91190.1 (Genbank) | URP |
| Pseudomonas aeruginosa C2773C Taxon ID: 1441459 | 653204529 | KEA25173.1 (Genbank) | URP |
| Pseudomonas aeruginosa C0324C Taxon ID: 1441461 | 653202323 | KEA23185.1 (Genbank) | URP |
| Pseudomonas aeruginosa C2159M Taxon ID: 1441462 | 653195452 | KEA16561.1 (Genbank) | URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 647089763 | KDR49016.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAK Taxon ID: 1009714 | 619448325 | KAJ90560.1 (Genbank) | URP |
| Pseudomonas aeruginosa M10 Taxon ID: 1339266 | 618787768 | KAJ24195.1 (Genbank) | URP |
| Pseudomonas aeruginosa IGB83 Taxon ID: 1339265 | 618770772 | KAJ07985.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH051 Taxon ID: 1418237 | 611940461 | EZP16428.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH050 Taxon ID: 1418236 | 611935101 | EZP11082.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH055 Taxon ID: 1418241 | 611905943 | EZO82059.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH056 Taxon ID: 1418242 | 611893242 | EZO69419.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH059 Taxon ID: 1418245 | 611892455 | EZO68640.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH057 Taxon ID: 1418243 | 611881538 | EZO57750.1 (Genbank) | URP |
| Pseudomonas aeruginosa PS75 Taxon ID: 1418247 | 611874376 | EZO50629.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3573 Taxon ID: 1447528 | 611869513 | EZO45776.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3574 Taxon ID: 1447529 | 611854936 | EZO31270.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO1-GFP Taxon ID: 1418248 | 611854149 | EZO30490.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3575 Taxon ID: 1447530 | 611845328 | EZO21784.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3578 Taxon ID: 1447533 | 611835451 | EZO12069.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3577 Taxon ID: 1447532 | 611831156 | EZO07830.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3579 Taxon ID: 1447534 | 611821768 | EZN98559.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3581 Taxon ID: 1447536 | 611813368 | EZN90275.1 (Genbank) | URP |
| Pseudomonas aeruginosa 3580 Taxon ID: 1447535 | 611812669 | EZN89580.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH029 Taxon ID: 1447537 | 611802436 | EZN79525.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH031 Taxon ID: 1447539 | 611794568 | EZN71703.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH032 Taxon ID: 1447540 | 611783443 | EZN60716.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH033 Taxon ID: 1447541 | 611776527 | EZN53876.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH035 Taxon ID: 1447543 | 611775934 | EZN53289.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWH036 Taxon ID: 1447544 | 611769191 | EZN46584.1 (Genbank) | URP |
| Pseudomonas aeruginosa PA96 Taxon ID: 1457392 | 610416110 | AHW71301.1 (Genbank) | URP |
| Pseudomonas aeruginosa LESlike4 Taxon ID: 1408275 | 589341166 | AHL13703.1 (Genbank) | URP |
| Pseudomonas aeruginosa LESlike1 Taxon ID: 1408274 | 589335236 | AHL07774.1 (Genbank) | URP |
| Pseudomonas aeruginosa LESB65 Taxon ID: 1408273 | 589329288 | AHL01827.1 (Genbank) | URP |
| Pseudomonas aeruginosa LES400 Taxon ID: 1408271 | 589323327 | AHK95866.1 (Genbank) | URP |
| Pseudomonas aeruginosa LESlike7 Taxon ID: 1408277 | 589317345 | AHK89885.1 (Genbank) | URP |
| Pseudomonas aeruginosa LESlike5 Taxon ID: 1408276 | 589311442 | AHK83983.1 (Genbank) | URP |
| Pseudomonas aeruginosa SG17M Taxon ID: 1443105 | 584588277 | EWH27623.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA038 Taxon ID: 1402530 | 574078139 | ETV54634.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA040 Taxon ID: 1402532 | 574065322 | ETV41897.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA042 Taxon ID: 1402534 | 574051670 | ETV28295.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA045 Taxon ID: 1402537 | 574040961 | ETV17629.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA047 Taxon ID: 1402539 | 574028052 | ETV04777.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA048 Taxon ID: 1402540 | 574011860 | ETU88660.1 (Genbank) | URP |
| Pseudomonas aeruginosa Z61 Taxon ID: 1402541 | 574010023 | ETU86825.1 (Genbank) | URP |
| Pseudomonas aeruginosa PS42 Taxon ID: 1402581 | 574005971 | ETU82800.1 (Genbank) | URP |
| Pseudomonas aeruginosa SCV20265 Taxon ID: 1427342 | 567366328 | AHC77273.1 (Genbank) | URP |
| Pseudomonas aeruginosa LES431 Taxon ID: 1408272 | 566564234 | AHC65755.1 (Genbank) | URP |
| Pseudomonas aeruginosa VRFPA06 Taxon ID: 1423141 | 564871637 | ETD73540.1 (Genbank) | URP |
| Pseudomonas aeruginosa VRFPA07 Taxon ID: 1431713 | 564834889 | ETD46641.1 (Genbank) | URP |
| Pseudomonas aeruginosa MTB-1 Taxon ID: 1415629 | 563411657 | AHB56079.1 (Genbank) | URP |
| Pseudomonas aeruginosa DHS29 Taxon ID: 1411701 | 563278204 | ESZ82887.1 (Genbank) | URP |
| Pseudomonas aeruginosa DHS01 Taxon ID: 1411700 | 557590396 | ESR98047.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO1-VE13 Taxon ID: 1367494 | 553900035 | AGY73567.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO1-VE2 Taxon ID: 1367493 | 553891751 | AGY68445.1 (Genbank) | URP |
| Pseudomonas aeruginosa MSH10 Taxon ID: 1402487 | 552727508 | ERZ40879.1 (Genbank) | URP |
| Pseudomonas aeruginosa CF127 Taxon ID: 1402488 | 552717602 | ERZ31014.1 (Genbank) | URP |
| Pseudomonas aeruginosa CF27 Taxon ID: 1402490 | 552714134 | ERZ27563.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA009 Taxon ID: 1402509 | 552685388 | ERY98954.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA010 Taxon ID: 1402510 | 552679875 | ERY93464.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA015 Taxon ID: 1402515 | 552672595 | ERY86230.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA016 Taxon ID: 1402516 | 552665608 | ERY79280.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL01 Taxon ID: 1402542 | 552651541 | ERY66006.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL02 Taxon ID: 1402543 | 552648808 | ERY63283.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL03 Taxon ID: 1402544 | 552637122 | ERY51642.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL05 Taxon ID: 1402546 | 552635555 | ERY50081.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL06 Taxon ID: 1402547 | 552628134 | ERY44570.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL12 Taxon ID: 1402553 | 552617968 | ERY34468.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL24 Taxon ID: 1402565 | 552590816 | ERY07471.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL25 Taxon ID: 1402566 | 552580529 | ERX97239.1 (Genbank) | URP |
| Pseudomonas aeruginosa M8A.4 Taxon ID: 1402570 | 552572405 | ERX89177.1 (Genbank) | URP |
| Pseudomonas aeruginosa 62 Taxon ID: 1402485 | 552524047 | ERX80833.1 (Genbank) | URP |
| Pseudomonas aeruginosa E2 Taxon ID: 1163395 | 552517303 | ERX74188.1 (Genbank) | URP |
| Pseudomonas aeruginosa MSH3 Taxon ID: 1402486 | 552512054 | ERX68983.1 (Genbank) | URP |
| Pseudomonas aeruginosa UDL Taxon ID: 1402493 | 552503238 | ERX60242.1 (Genbank) | URP |
| Pseudomonas aeruginosa X24509 Taxon ID: 1402492 | 552500488 | ERX57528.1 (Genbank) | URP |
| Pseudomonas aeruginosa CF18 Taxon ID: 1402489 | 552499758 | ERX56844.1 (Genbank) | URP |
| Pseudomonas aeruginosa S35004 Taxon ID: 1402499 | 552463531 | ERX21133.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA003 Taxon ID: 1402503 | 552445043 | ERX02331.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA001 Taxon ID: 1402501 | 552444052 | ERX01953.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA004 Taxon ID: 1402504 | 552435764 | ERW93728.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA002 Taxon ID: 1402502 | 552434695 | ERW92662.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA005 Taxon ID: 1402505 | 552427776 | ERW85876.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA012 Taxon ID: 1402512 | 552408433 | ERW66815.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA017 Taxon ID: 1402517 | 552399875 | ERW58331.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA018 Taxon ID: 1402518 | 552396762 | ERW55296.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA014 Taxon ID: 1402514 | 552396497 | ERW55037.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA020 Taxon ID: 1402520 | 552379235 | ERW37861.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA019 Taxon ID: 1402519 | 552378336 | ERW36970.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA022 Taxon ID: 1402522 | 552367405 | ERW26244.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA023 Taxon ID: 1402523 | 552366785 | ERW25625.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA021 Taxon ID: 1402521 | 552362532 | ERW21386.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA025 Taxon ID: 1402525 | 552348684 | ERW07724.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA024 Taxon ID: 1402524 | 552346469 | ERW05522.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL07 Taxon ID: 1402548 | 552314130 | ERV73559.1 (Genbank) | URP |
| Pseudomonas aeruginosa BWHPSA028 Taxon ID: 1402528 | 552312446 | ERV72130.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL10 Taxon ID: 1402551 | 552282939 | ERV42981.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL11 Taxon ID: 1402552 | 552277563 | ERV37639.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL19 Taxon ID: 1402560 | 552254514 | ERV14837.1 (Genbank) | URP |
| Pseudomonas aeruginosa BL18 Taxon ID: 1402559 | 552253288 | ERV13613.1 (Genbank) | URP |
| Pseudomonas aeruginosa M9A.1 Taxon ID: 1402571 | 552239767 | ERV00330.1 (Genbank) | URP |
| Pseudomonas aeruginosa M8A.1 Taxon ID: 1402567 | 552233085 | ERU93218.1 (Genbank) | URP |
| Pseudomonas aeruginosa C20 Taxon ID: 1402572 | 552222755 | ERU83011.1 (Genbank) | URP |
| Pseudomonas aeruginosa C23 Taxon ID: 1402573 | 552220749 | ERU81107.1 (Genbank) | URP |
| Pseudomonas aeruginosa C40 Taxon ID: 1402574 | 552206968 | ERU67570.1 (Genbank) | URP |
| Pseudomonas aeruginosa C41 Taxon ID: 1402575 | 552206598 | ERU67201.1 (Genbank) | URP |
| Pseudomonas aeruginosa C48 Taxon ID: 1402576 | 552198858 | ERU59631.1 (Genbank) | URP |
| Pseudomonas aeruginosa C51 Taxon ID: 1402577 | 552197410 | ERU58188.1 (Genbank) | URP |
| Pseudomonas aeruginosa C52 Taxon ID: 1402578 | 552191020 | ERU51884.1 (Genbank) | URP |
| Pseudomonas aeruginosa CF614 Taxon ID: 1402580 | 552175499 | ERU35978.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO581 Taxon ID: 1352354 | 543881663 | AGV59664.1 (Genbank) | URP |
| Pseudomonas aeruginosa c7447m Taxon ID: 1352355 | 543874688 | AGV63847.1 (Genbank) | URP |
| Pseudomonas aeruginosa WC55 Taxon ID: 1333855 | 534382827 | EQM85098.1 (Genbank) | URP |
| Pseudomonas aeruginosa RP73 Taxon ID: 1340851 | 514247535 | AGO39916.1 (Genbank) | URP |
| Pseudomonas aeruginosa MSH-10 Taxon ID: 1331059 | 508139316 | EOT16178.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAK Taxon ID: 1009714 | 508138379 | EOT15251.1 (Genbank) | URP |
| Pseudomonas aeruginosa VRFPA02 Taxon ID: 1315978 | 500730456 | EOQ79565.1 (Genbank) | URP |
| Pseudomonas aeruginosa PA45 Taxon ID: 1295076 | 477560815 | ENH94268.1 (Genbank) | URP |
| Pseudomonas sp. P179 Taxon ID: 1125698 | 476640748 | EMZ47177.1 (Genbank) | URP |
| Pseudomonas aeruginosa PA21_ST175 Taxon ID: 1285581 | 453042202 | EME89951.1 (Genbank) | URP |
| Pseudomonas aeruginosa E2 Taxon ID: 1163395 | 404544984 | EKA54098.1 (Genbank) | URP |
| Pseudomonas aeruginosa ATCC 14886 Taxon ID: 1163392 | 404519323 | EKA30086.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO579 Taxon ID: 1191475 | 404350601 | EJZ76938.1 (Genbank) | URP |
| Pseudomonas aeruginosa DK2 Taxon ID: 1093787 | 392319815 | AFM65195.1 (Genbank) | URP |
| Pseudomonas aeruginosa MPAO1/P2 Taxon ID: 1131758 | 375048775 | EHS41290.1 (Genbank) | URP |
| Pseudomonas aeruginosa MPAO1/P1 Taxon ID: 1131757 | 375044062 | EHS36674.1 (Genbank) | URP |
| Pseudomonas aeruginosa 2192 Taxon ID: 350703 | 126194015 | EAZ58078.1 (Genbank) | URP |
| Pseudomonas aeruginosa C3719 Taxon ID: 350704 | 126167172 | EAZ52683.1 (Genbank) | URP |
| Pseudomonas aeruginosa PAO1 Taxon ID: 208964 | 9948239 | AAG05603.1 (Genbank) | PRP URP |
| Pseudomonas aeruginosa Taxon ID: 287 | 831199574 | URP | |
| Pseudomonas aeruginosa Taxon ID: 287 | 777185893 | URP | |
| Pseudomonas aeruginosa WS136 Taxon ID: 1341644 | 747645812 | URP | |
| Pseudomonas aeruginosa Taxon ID: 287 | 734477238 | URP | |
| Pseudomonas aeruginosa Taxon ID: 287 | 727037974 | URP | |
| Pseudomonas aeruginosa Taxon ID: 287 | 675289021 | URP | |
| Pseudomonas aeruginosa PA38182 Taxon ID: 1407059 | 575873307 | URP | |
| Pseudomonas aeruginosa MH38 Taxon ID: 1336203 | 571153380 | URP | |
| Pseudomonas aeruginosa 18A Taxon ID: 1286151 | 451756021 | URP | |
| Pseudomonas aeruginosa NCMG1179 Taxon ID: 1078464 | 346057892 | URP | |
| Pseudomonas aeruginosa LESB58 Taxon ID: 557722 | 218772058 | URP | |
| obsolete GIs = 458890779, 451986305, 424941446, 421516873, 421180683, 421156132, 418592349, 418585773, 254240637, 254235241, 107101642, 692153454, 568309898, 568154255, 564952723, 514409565, 392984310, 218891832 | |||
| Show All | |||
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| n/a | A0A1G5IR37 | A0A1G5IR37_ACIBA (TrEMBL) | |
| n/a | A0A1E9BUL2 | A0A1E9BUL2_9PSED (TrEMBL) | |
| n/a | A0A0P1DBK0 | A0A0P1DBK0_PSEAI (TrEMBL) | |
| n/a | A0A072ZMJ7 | A0A072ZMJ7_PSEAI (TrEMBL) | |
| n/a | A0A0E1ATH4 | A0A0E1ATH4_PSEAI (TrEMBL) | |
| n/a | Q9I1Q2 | Q9I1Q2_PSEAE (TrEMBL) | |
| n/a | A0A1D5BMS6 | A0A1D5BMS6_9PSED (TrEMBL) | |
| n/a | A0A1F0I4T0 | A0A1F0I4T0_9PSED (TrEMBL) | |
| Show All | |||
Length of Enzyme (full-length): 391 | Length of Functional Domain: 391
MKIKSVRTRVFEWKGKVVPPQAHFCTNASDILFEKGDAMGSFRFHGWLVVEIETDDGLVG
IGNCALAPRVAKEIVDLYLAPICIGEDPFDNEYIWQKMYRRTHAWGRKGIGMAAISAVDL
AIWDIMGKAVNKPVFKLLGGRTKEKIWTYASKLYANDNLDAFLEEAQGYLNQGFTALKMR
FGYGPKDGPTGMRRNIEQVRALRELAGPDIDIMLECYMGWTLEYARRMLPKLAEFEPRWL
EEPVIADDIEGYVELKKMGIMPISGGEHEFTGHGFKDLLERRAVDVIQYDTNRVGGITAA
RKINAMAEAWSVPVIPHAGQLHNYHLTMASTASPMAEFFPVFDVEVGNELFYYVFKGEPQ
PVDGYIQLDDHKPGLGLEISEEHLKDFIIIE
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.