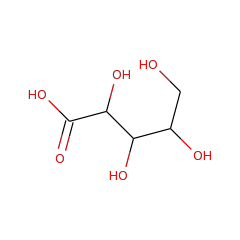
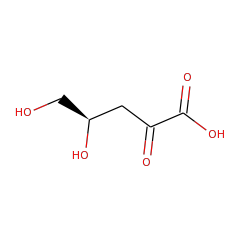
Top Level Name
⌊ Superfamily (core) Enolase
⌊ Subgroup mandelate racemase
⌊ Family L-lyxonate dehydratase
⌊ FunctionalDomain L-lyxonate dehydratase (ID 277983)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS |
| Family Assignment Evidence Code | IES PubMed:24831290 |
| This entry was last updated on | Nov. 21, 2017 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Dinoroseobacter shibae DFL 12 Taxon ID: 398580 | 345101064 | URP |
Length of Enzyme (full-length): 413 | Length of Functional Domain: 390
MHHHHHHSSGVDLGTENLYFQSMTKIKSVRTRVWNWTGPTVPPQGNFCTNASDALWIQGD
AMASFRFHQWLTCEVETEDGTIGIGNAALAPNVVKQAIDEWYAPLVIGEDPFDYAYLWEK
MYRRTHAWGRKGIGMTAISAIDIAIWDLMGKLVGKPVFKLLGGRTKEKIPVYYSKLYADS
IPAMQAEAEEAQKHGYQGYKTRFGYGPKDGPAGMRENLKRVEALREVLGYDVDLMLECYM
GWNLDYTKRMLPKLERFEPRWLEEPVIADDVAGYAELNAMGIVPISGGEHEFSVMGCAEL
INRKAVSVLQYDTNRVGGITAAQKINAIAEAAQIIVIPHAGQMHNYHLTMANMNCPISEY
FPVFDVEVGNELFYYIFDGDPEAVDGYLQLDDDTPGLGITISDAHLKHFEITE
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.