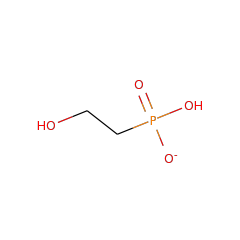
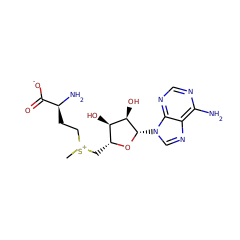
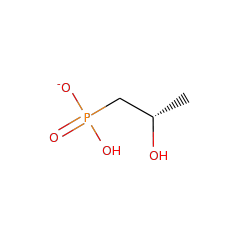
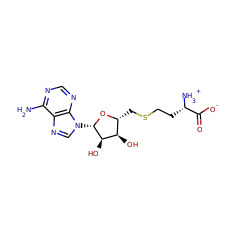
Top Level Name
⌊ Superfamily (core) Radical SAM
⌊ Subgroup B12-binding domain containing
⌊ Family phosphonoacetaldehyde methylase (fom3-like)
⌊ FunctionalDomain Phosphonoacetaldehyde methylase (ID 391556)
No Notes.
| Superfamily Assignment Evidence Code(s) | ISS |
| Family Assignment Evidence Code | CFM PubMed:19489722 |
| This entry was last updated on | April 9, 2016 |
References to Other Databases
Genbank
| Species | GI | Accession | Proteome |
|---|---|---|---|
| Streptomyces wedmorensis Taxon ID: 43759 | 1061002 |
Uniprot
| Protein Name | Accession | EC Number
 |
Identifier |
|---|---|---|---|
| 2-hydroxyethylphosphonate methyltransferase {ECO:0000305} | Q56184 | FOM3_STRWE (Swiss-Prot) |
Length of Enzyme (full-length): 534 | Length of Functional Domain: 534
MTIGSLGSTEFALHGKPAIRWGDLPQRVGKPETRRYQKVLLLNPSATLFRHDLPRCTYPL
GLGYIAAVLEKYGYEVKILDVFAEGYYNAQPVDGDDQFLRYGLSDDDIVKVMKEFGPDVV
GISSIFSNQADNVHHLLKLADLVTPEAVTAIGGAHARYFPKACLDDPNLDAVFLGEGEMT
FLLWMEHLNGNVSDDEVHGIAWRDRDGKVQIKPELPLISSMRPEGPETGKSSPMLSMAGE
LDHIPFPAWHHYNMEKYFEIKAYQSPYTVGSRVGQLYTSRGCTAHCTFCTTTHFWGQKLR
RRSVQDVVDEVLRLRDEYGIDEFHIQDDNITNDMDHARELFRAFKEVGLPWATPQGTALW
RMDEELLDLMAESGAYQVTFAIESGVQRVLKELIKKPLNLERTSHLIKYARSLGMHVHGF
FIIGMPPMCGNAGESIEEMQASYDYAEEAGFSSASFFAASPIVGSELLRECIRQGFVDPE
ESLYRMTYKQGIINVPGLWDGEEIAELAAKFNRDFNARRDRAYTPQKQWNANQY
Conserved catalytic residues (as determined by automated alignment to family, subgroup, or superfamily HMMs) are shown with teal highlighting . Conserved catalytic residues which do not matched the Conserved Alignment Residue are shown with maroon highlighting . Information regarding their function can be found in the Conserved Residues section below.